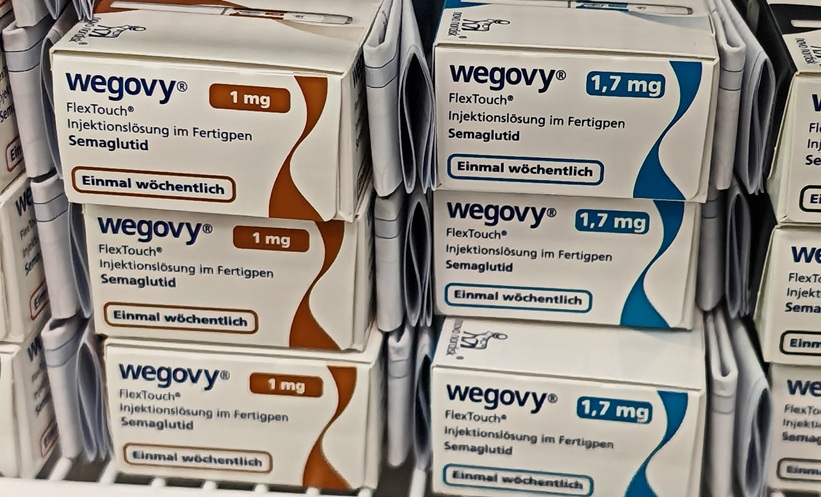
SEMAGLUTIDE (Wegovy) is set to be available on the NHS within months and 1.2 million people with cardiovascular disease are eligible to receive it, the NHS reported on 1st April 2026.
This follows approval by the National Institute for Health and Care Excellence (NICE) to prescribe the glucagon-like peptide-1 (GLP-1) receptor agonist, already available on the NHS for patients with Type 2 diabetes, to those at risk of heart attack and strokes.
The injection will be an additional treatment offered on top of, for example, statins and lifestyle interventions, offering extra protection against cardiovascular disease.
The news comes after the London Assembly Health Committee’s March report revealed that people in the capital seeking weight loss drugs are sourcing them privately and risking serious harm, due to a slow NHS rollout.
NICE Guidance
Draft NICE guidance on semaglutide, published on 1st April 2026, recommends the drug be available for those who have previously had a heart attack, stroke, peripheral arterial disease, or are overweight or obese with a BMI of 27 kg/m² or higher.
The NHS said it has struck a deal that makes the treatment cost-effective for a rollout on the health service.
Sarah Hodgson, Health Minister, London, UK, said: “Weight loss drugs are proving to be a game changer in tackling obesity. Extending their use for people who also suffer from cardiovascular disease will be a life saver.
“We are committed to ensuring the NHS makes these drugs available to those who need them the most.”
Supporting Evidence
Data from the SELECT large-scale trial has shown that taking semaglutide alongside existing cardiovascular medicines reduced the risk of major adverse cardiovascular events by 20% in people with heart and circulatory disease who are overweight.
NICE’s independent committee assessed the evidence, concluding that semaglutide “offers good value for the NHS”.
Similarly, other emerging evidence suggests that discontinuation of GLP-1 treatment, even for limited periods, could increase the risk of heart disease in adults with Type 2 diabetes.
Combatting Cardiovascular Disease in England
Heart and circulatory disease are one of the leading causes of early death and ill health in England.
Annually, there are 100,000 hospitalisations due to heart attacks, 100,000 people who experience a stroke, and around 350,000 people living with peripheral arterial disease.
Helen Williams, National Clinical Director for Cardiovascular Disease Prevention, NHS England, London, UK, said: “We know that people who have already had a heart attack or stroke face a much higher risk of having another.
“Used alongside a healthy diet and regular exercise, semaglutide could help prevent thousands of future major cardiovascular events and give many people the chance at a longer and healthier life.”
References
National Institute for Health and Care Excellence. Semaglutide for reducing the risk of major adverse cardiovascular events in people with cardiovascular disease and overweight or obesity [ID6441]. Available at: https://www.nice.org.uk/guidance/indevelopment/gid-ta11544. Last accessed: 4 April 2026.
London Assembly Health Committee. Weight loss medication in London. Available at: https://www.london.gov.uk/who-we-are/what-london-assembly-does/london-assembly-work/london-assembly-publications/weight-loss-medicines-london. Last accessed: 16 March 2026.
Deanfield J et al. Semaglutide and cardiovascular outcomes by baseline and changes in adiposity measurements: a prespecified analysis of the SELECT trial. Lancet. 2025;406(10516):2257-2268.
Xie Y et al. Glucagon-like peptide 1 receptor agonist discontinuation and risks of major adverse cardiovascular events in adults with type 2 diabetes: target trial emulation. BMJ med. 2026;5(1):DOI:10.1136/ bmjmed-2025-002150.
Featured image: Semi on Adobe Stock