Abstract
Background: Carboplatin and paclitaxel are standard first-line therapies for advanced ovarian cancer, but hypersensitivity reactions (HSR) may compromise treatment, especially when involving both agents.
Case report: A 71-year-old woman with Stage IIIC ovarian carcinoma and no comorbidities developed sequential HSRs to carboplatin and paclitaxel. After carboplatin, she experienced a delayed generalised maculopapular rash at 24 hours. During paclitaxel infusion, she developed acute chest tightness and desaturation to 85%. Allergy testing (prick, intradermal, patch) was initially equivocal but subsequently negative. Management included corticosteroid/antihistamine premedication, prolonged carboplatin infusion, and a seven-step paclitaxel desensitisation protocol. The patient successfully completed six chemotherapy cycles.
Conclusion: This case emphasises the importance of multidisciplinary collaboration and personalised strategies to ensure continuity of effective oncologic treatment despite HSRs.
[/hl]Key Points[/hl]
1. Hypersensitivity reactions to carboplatin and paclitaxel are recognised complications in advanced ovarian cancer and may threaten continuation of first-line chemotherapy.2. This case report describes sequential hypersensitivity to both agents in a single patient and outlines the diagnostic evaluation and multidisciplinary management approach implemented. 3. Individualised premedication, infusion adjustment, and structured desensitisation protocols can enable safe chemotherapy continuation without compromising short-term oncologic control.
INTRODUCTION
Carboplatin and paclitaxel remain the cornerstone of first-line chemotherapy for advanced ovarian cancer, as recommended by international guidelines, with well-established efficacy and survival benefits.1 However, hypersensitivity reactions (HSR) to these agents present a major clinical challenge, sometimes leading to treatment discontinuation and compromising therapeutic efficacy.2
Carboplatin HSRs are typically delayed and may appear after several cycles, often manifesting as cutaneous eruptions or, less frequently, systemic reactions.3 Paclitaxel HSRs, in contrast, usually occur during the first or second infusion, are often non-IgE mediated, and are related to polyoxyl 35 castor oil.4 Dual hypersensitivity to both carboplatin and paclitaxel is uncommon, but represents a critical barrier to treatment continuity.
This case is unique in that it demonstrates sequential hypersensitivity to both carboplatin and paclitaxel in the same patient, managed successfully through a multidisciplinary collaboration between oncologists and allergologists. It highlights the importance of repeated allergy testing, premedication, infusion adjustment, and desensitisation protocols in enabling completion of therapy.
CASE PRESENTATION
A 71-year-old woman with no significant past medical history was diagnosed with Stage IIIC ovarian carcinoma. She was started on first-line chemotherapy with carboplatin and paclitaxel according to international guidelines.
Carboplatin Reaction
During the second cycle, approximately 24 hours after carboplatin infusion, the patient developed a diffuse maculopapular rash predominantly affecting the thighs and lower limbs. The eruption was pruritic but without mucosal involvement or systemic features, such as fever, dyspnoea, or haemodynamic instability. The lesions resolved with topical corticosteroid treatment within a few days (Figure 1).
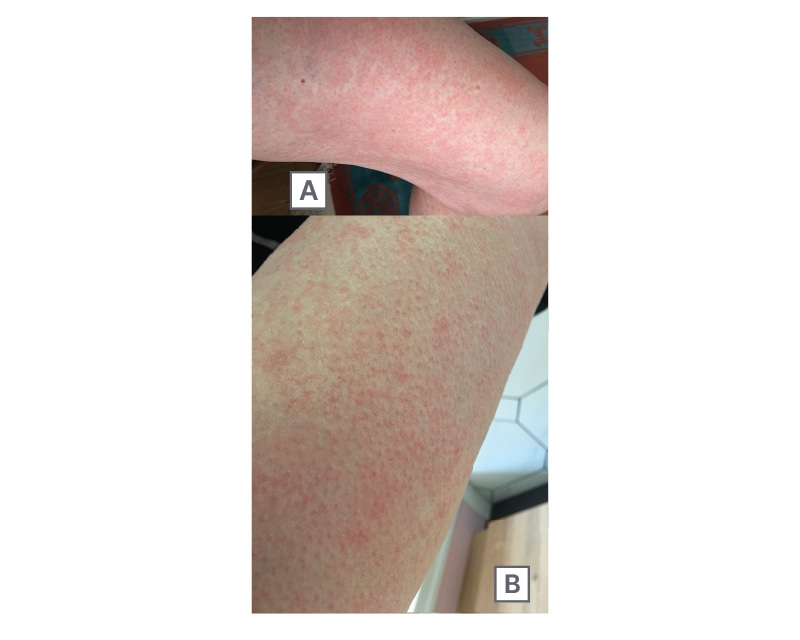
Figure 1: Delayed cutaneous hypersensitivity reaction after carboplatin infusion.
A) Maculopapular rash on the thigh. B) Diffuse erythematous eruption with confluent macules and papules.
Given the suspicion of carboplatin hypersensitivity, the patient underwent allergological testing. The first series included skin prick, intradermal, and patch testing for carboplatin, paclitaxel, and premedication drugs. Results showed a doubtful intradermal reaction to carboplatin at 72 hours. A second series of tests, performed at a distance from the reaction, was completely negative.
Carboplatin was subsequently reintroduced under systematic premedication with intravenous corticosteroids and antihistamines, combined with a prolonged infusion protocol. Under these conditions, treatment was continued with only mild erythematous reactions, without recurrence of generalised rash.
Paclitaxel Reaction
At Cycle 3 (Day 15), during paclitaxel infusion, the patient developed acute chest tightness and oxygen desaturation to 85%. In addition, she presented with a diffuse erythematous rash involving the abdomen and thighs (Figure 2). The infusion was immediately interrupted, and intravenous corticosteroids and antihistamines were administered, resulting in rapid improvement.
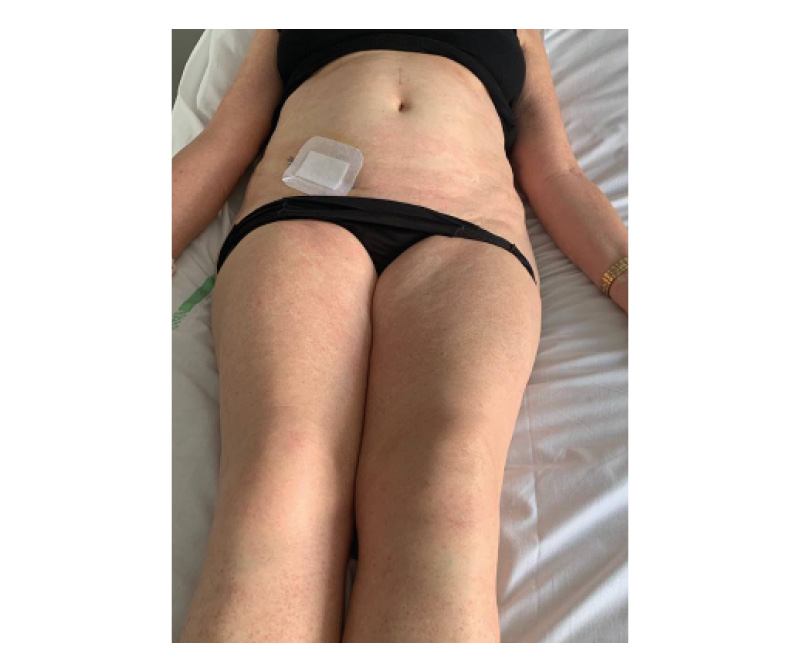
Figure 2: Diffuse erythematous rash involving the abdomen and thighs during paclitaxel infusion, consistent with acute hypersensitivity reaction.
Given the importance of maintaining paclitaxel in the regimen, a seven-step desensitisation protocol over 5 hours was initiated. The patient tolerated this approach well and was able to complete paclitaxel administration without recurrence of severe reactions.
Outcome
Through close multidisciplinary collaboration between oncologists and allergologists, the patient successfully completed six cycles of carboplatin–paclitaxel chemotherapy. No anaphylactic events occurred, and clinical and radiological follow-up during treatment did not raise concern for early disease progression, suggesting that continuation of chemotherapy through desensitisation did not appear to compromise short-term oncologic control.
A detailed summary of clinical events, diagnostic investigations, and management strategies is presented in Table 1.
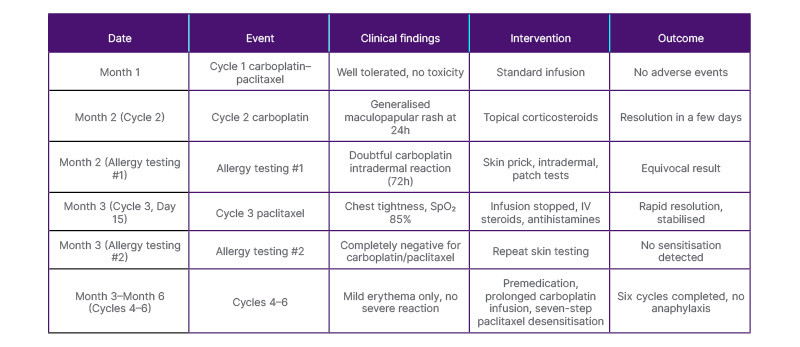
Table 1: Timeline of hypersensitivity management during first-line chemotherapy with carboplatin–paclitaxel in a 71-year-old woman with Stage IIIC ovarian carcinoma.
Clinical events, interventions, and outcomes are summarised. IV: intravenous; SpO₂: peripheral oxygen saturation.
DISCUSSION
HSRs to platinum compounds occur in up to 27% of patients with ovarian cancer, especially after multiple cycles.3 They are usually mediated by delayed-type hypersensitivity mechanisms, with clinical manifestations ranging from mild rash to severe systemic reactions.5 In the patient, carboplatin induced a generalised maculopapular eruption within 24 hours, consistent with a delayed reaction. Although allergy testing showed an equivocal intradermal result initially, a repeat test later returned negative, underlining the variability and limitations of diagnostic tools in non-IgE-mediated reactions.6
Several risk factors have been associated with HSRs to platinum agents and taxanes, including repeated exposure, cumulative dose, a history of atopy or drug allergy, and prolonged platinum-free intervals. Awareness of these factors may help identify patients at higher risk and guide closer monitoring during subsequent cycles. These observations have been highlighted in recent analyses of chemotherapy-associated HSRs.7
Paclitaxel-related HSRs are reported in 10–30% of patients, with severe reactions in 2–5%.4 These reactions are mostly attributed to polyoxyl 35 castor oil, which triggers mast cell activation through a non-IgE-mediated pathway.8 In this case, the patient developed acute chest tightness and desaturation during infusion, which was rapidly reversed after stopping the drug and administering corticosteroids.
Management of chemotherapy-induced HSRs requires a tailored, multidisciplinary approach. Premedication with corticosteroids and antihistamines reduces the incidence of paclitaxel HSRs, while prolonged infusion protocols can minimise carboplatin toxicity.9,10 Desensitisation, performed through gradual reintroduction in multiple steps, has shown high success rates for both carboplatin and paclitaxel.11 In the authors’ patient, a seven-step paclitaxel desensitisation protocol allowed treatment continuation without recurrence of severe events.
Experience from breast cancer treatment has significantly contributed to the development and validation of desensitisation protocols, particularly for taxane-based regimens. Structured multi-step protocols have enabled many patients with prior HSRs to safely continue first-line therapy without compromising treatment intensity. Lessons from these settings support the broader applicability of desensitisation strategies across solid tumours, including ovarian cancer.12
An important clinical consideration in patients requiring desensitisation is whether modification of infusion protocols may compromise oncologic efficacy. In this case, continuation of carboplatin and paclitaxel using premedication, prolonged infusion, and a structured desensitisation protocol allowed completion of the planned first-line regimen. Although a formal Response Evaluation Criteria in Solid Tumors (RECIST) response assessment was not the primary objective of this report, no early clinical or radiological signs of progression were observed during treatment. Long-term outcomes, such as progression-free and overall survival, could not be assessed due to the limited follow-up period, which represents an inherent limitation of single-patient case reports.
The strength of this case lies in the demonstration that, even in the presence of dual hypersensitivity, therapeutic efficacy can be preserved through multidisciplinary management. The main limitation is the absence of confirmatory biomarkers for predicting reactions, emphasising the need for clinical vigilance and individualised strategies.
Take-Away Lesson
Negative allergy tests do not exclude clinical hypersensitivity. Personalised premedication, infusion adjustment, and desensitisation, supported by close oncologist–allergologist collaboration, are essential to ensure both safety and oncologic efficacy in patients with complex chemotherapy-induced HSRs.
CONCLUSION
This case highlights the clinical challenges of managing hypersensitivity reactions to both carboplatin and paclitaxel in advanced ovarian cancer. Despite negative allergy tests, the patient developed reproducible reactions, emphasising that diagnostic tools may not fully predict clinical risk. Through a multidisciplinary approach involving oncologists and allergologists, the use of premedication, prolonged infusion, and desensitisation protocols enabled safe continuation and completion of six chemotherapy cycles. The main take-away message is that individualised strategies and close collaboration across specialties are essential to preserve oncologic efficacy, while ensuring patient safety in the context of complex chemotherapy-induced HSRs.






