Abstract
An increasing number of studies are highlighting gastrointestinal disorders associated with hypermobile Ehlers-Danlos syndrome (hEDS), particularly in cases where it is accompanied by postural orthostatic tachycardia syndrome and mast cell activation syndrome. The diagnosis of these conditions remains challenging. Gastrointestinal symptoms include chronic small intestinal dysmotility and visceral hypersensitivity. Here, the authors present the case of a young woman with hEDS, postural orthostatic tachycardia syndrome, and mast cell activation syndrome, whose quality of life improved significantly after she started receiving artificial nutrition. This case is of particular interest as, during the initial phase of parenteral nutrition, several central vascular accesses failed prematurely due to an abnormal subcutaneous fibrinoid reaction. Switching to enteral nutrition was crucial in resolving the complications associated with a central venous access, restoring intestinal function, and improving quality of life. This paper will examine the challenges presented by this case in detail, providing notes on the necessary pathophysiology to understand the gastrointestinal complications related to hEDS and evaluating the most effective and sustainable medical and nutritional strategies.
Key Points
1. The article describes a rare vascular complication in a patient with hypermobile Ehlers-Danlos syndrome receiving parenteral nutrition and her subsequent successful management with enteral feeding.2. This case report highlights the diagnostic and therapeutic challenges of managing severe gastrointestinal dysfunction in a patient with hypermobile Ehlers-Danlos syndrome complicated by postural orthostatic tachycardia syndrome and mast cell activation syndrome.
3. Enteral nutrition could resolve complications associated with central venous access, restore intestinal function, and improve quality of life.
INTRODUCTION
Ehlers-Danlos syndrome (EDS) hypermobility type (hEDS) is an inherited connective tissue disorder that is characterised by joint hypermobility and musculoskeletal pain. It frequently presents with gastrointestinal (GI) symptoms, including chronic small intestinal dysmotility and visceral hypersensitivity.
There are 13 recognised subtypes of EDS.1 Among these, Type 3, the hypermobile variant (hEDS), is the only one without an identified genetic cause or clearly defined pathophysiology. This subtype is most strongly linked to GI symptoms. The lack of a known genetic marker represents a major challenge for diagnosis, as hEDS is currently identified solely based on clinical criteria.
Although not extensively investigated, it can be hypothesised that alterations in the mechano-elastic properties of the GI tract, combined with changes in mechanosensory afferent signalling, may contribute to the development of visceral pain. Sensitisation, defined as a lowered threshold for perceiving sensations originating from visceral organs, could result from this enhanced afferent activity. Moreover, rectal hyposensitivity has been reported in individuals with EDS and is considered a key factor contributing to constipation.
Visceral hypersensitivity is widely recognised as a possible mechanism underlying disorders of gut–brain interaction and may represent a shared pathophysiological process between disorders of gut–brain interaction and EDS. In addition, studies examining fibroblasts from the skin of patients with EDS suggest that chronic inflammation may also play a contributory role.2
The coexistence of postural orthostatic tachycardia syndrome (POTS), mast cell activation syndrome (MCAS), and hEDS within the same individual is well documented. POTS has been identified as a strong predictor of GI dysmotility in EDS. As previously noted, individuals with hEDS frequently experience GI and orthostatic symptoms, suggesting that GI manifestations observed in POTS may, at least in part, arise from this overlap. Some authors have proposed that the combination of POTS and hEDS represents a distinct POTS subtype, supported by evidence indicating that affected individuals are typically younger, present with more frequent GI symptoms, demonstrate greater multisystem involvement, and show a higher prevalence of non-GI manifestations, such as fatigue, fibromyalgia, and depression, compared with those who have hEDS alone.
When eating-related symptoms result in reduced oral intake, nutritional support should be provided within a multidisciplinary framework.3 The initial strategy should focus on optimising oral intake through dietary adjustments and the use of oral nutritional supplements. Clinically assisted nutrition and hydration are primarily indicated in cases of objectively confirmed malnutrition or electrolyte imbalance and should not be employed solely for symptom management due to the potential iatrogenic risks. If necessary, nasogastric feeding may be initiated, with consideration of long-term percutaneous endoscopic gastrostomy feeding if tolerated. In cases of severe gastric dysfunction but preserved small intestinal motility and absorptive capacity, post-pyloric feeding may be started via a nasojejunal route, with possible transition to long-term feeding through a percutaneous endoscopic gastrostomy-jejunal tube or a direct jejunal route (such as percutaneous endoscopic jejunostomy, balloon jejunostomy, or surgical jejunostomy), if tolerated. However, insertion of percutaneous feeding tubes carries inherent risks of morbidity and mortality, as well as complications related to wound healing, infection, and overgranulation, issues to which this patient population may be particularly vulnerable.
Intolerance to small intestinal enteral feeding is often driven by pain or discomfort rather than impaired intestinal motility or absorption. In patients with chronic visceral hypersensitivity, food intake may become associated with fear avoidance behaviours. Consequently, chronic pain management, focusing on neuropathic pain agents and psychological interventions with a rehabilitative approach, represents an essential component of multidisciplinary care.
The case the authors present is notable for two main features: (1) To their best knowledge, fibrinoid catheter reactions in hEDS necessitating discontinuation of parenteral nutrition (PN) have not been reported before; and (2) the patient has been successfully treated with enteral post-pyloric nutrition.
DETAILED CASE DESCRIPTION
A 24-year-old White woman with hEDS and myopathic features, POTS, and MCAS was referred to the authors’ ambulatory care centre for nutritional counselling. She presented with GI pain, dyspepsia, and hiccups after eating solid foods or oral nutritional supplements. The patient reported that she had experienced these symptoms for several years and had required PN support at another facility. PN was suspended due to intolerance to the administered mixtures, which manifested itself after several weeks of therapy (Table 1).
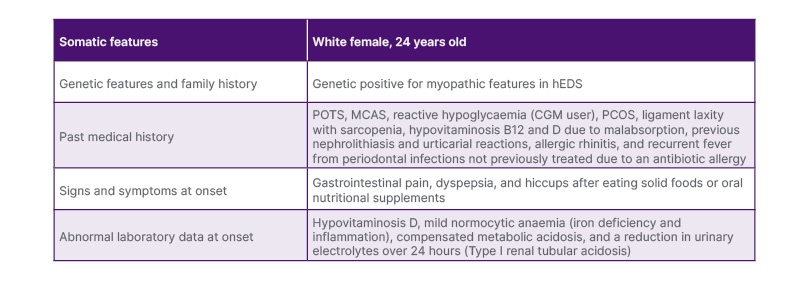
Table 1: Patient and disease characteristics at onset.
CGM: continuous glucose monitoring; hEDS: hypermobile Ehlers-Danlos syndrome; MCAS: mast cell activation syndrome; PCOS: polycystic ovary syndrome; POTS: postural orthostatic tachycardia syndrome.
The patient’s medical history also included reactive hypoglycaemia (she was a continuous glucose monitoring user), polycystic ovary syndrome, ligament laxity with sarcopenia, and hypovitaminosis B12 and D due to malabsorption. She had a history of previous nephrolithiasis and urticarial reactions, which ceased with the start of chronic antihistamine therapy (ebastine 10 mg + cetirizine 10 mg/mL oral drops, 20 drops in the evening, increasable to 30 drops). The patient also had allergic rhinitis and recurrent fever from periodontal infections that had not been previously treated due to antibiotic polyallergies.
During the suspension phase of parenteral therapy, the young woman experienced a progressive reduction in lean body mass, which impaired her mobility. She required a wheelchair due to ligament laxity and frequent joint subluxations. She was subsequently referred to Ancona University Hospital, Italy, for further nutritional evaluation.
During the authors’ initial assessment (Figure 1), she reported experiencing debilitating abdominal pain after consuming solid and liquid foods. She was constipated and bloated. Urine output was consistently low at <500 mL. She consumed 2.5 L of water per day, divided into smaller amounts, due to epigastric pain after drinking liquids. She also underwent daily infusion therapy, receiving 1,000 mL/day of saline solution and 500 mL/day of rehydrating electrolyte solution.
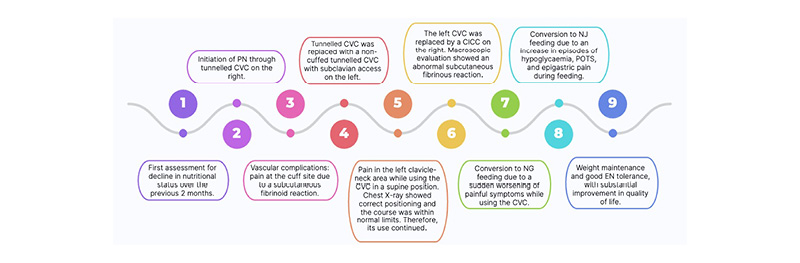
Figure 1: Timeline and sequence of events.
CICC: centrally inserted central catheter; CVC: central venous catheter; EN: enteral nutrition; NJ: nasojejunal; PN: parenteral nutrition; POTS: postural orthostatic tachycardia syndrome.
Her extremely variable dietary history was approximately 356 kcal (mainly dairy products to promote GI tolerance), providing 14.5 g of protein. After discontinuing parenteral support, she began taking four nutritional supplements per day, each containing 300 kcal and 12 g of protein. Ten days before the initial assessment, she stopped taking these supplements due to increased epigastric pain, reflux symptoms, and an increase in episodes of debilitating reactive hypoglycaemia.
Over the previous 2 months, she had experienced progressive weight loss of 2.5 kg, from 55 kg (BMI 18.6 kg/m²) to 52.5 kg (BMI 17.7 kg/m²). On examination, she was found to have normal systemic objectivity except for diffuse abdominal tenderness and muscle hypotrophy.
Laboratory tests showed hypovitaminosis D, compensated metabolic acidosis, mild normocytic anaemia (iron deficiency and inflammation), and a reduction in urinary electrolytes over 24 hours. Blood and urine tests also enabled a diagnosis of Type I renal tubular acidosis to be made (for which treatment with sodium bicarbonate was initiated orally, restoring blood values to normal levels).
The clinical history and medical records indicated chronic functional intestinal insufficiency due to dysmotility, with symptoms so severe as to affect normal eating. Therefore, it was deemed necessary to resume parenteral support once the allergy testing had been completed.
After initiating chronic antihistamine therapy, under protected conditions parenteral mixtures were tested, including some that had already been administered and some that were new. The allergy tests were negative, so, PN was started after placement of a subclavian tunnelled central venous catheter (CVC) on the right.
Three months later, a fever of up to40 °C appeared, accompanied by pain at the CVC site, increasing during PN infusion, and persisting after the feverish phase. A vascular surgical consultation was therefore performed, raising the possibility of pain at the cuff site due to a subcutaneous fibrinoid reaction, since the CVC showed no signs of malfunction. However, three sets of blood cultures were taken from the CVC and peripheral venous catheter, as well as blood samples for biological indices of inflammation, all of which were subsequently found to be negative. Due to painful symptoms persisting after the fever subsided, 1 month later, the tunnelled CVC was replaced with a non-cuffed tunnelled CVC with subclavian access on the left.
During her check-up 3 months later, she reported experiencing pain in the left clavicle-neck area for approximately 2 weeks while using the CVC in a supine position. The pain was exacerbated by a malfunctioning infusion pump, but subsided when she moved to a semi-sitting position. The chest X-ray showed correct positioning and the course was within normal limits. Therefore, its use continued.
The patient returned for a check-up the next month, reporting that, a week after the CVC dressing was replaced and the stat lock system was changed, she had experienced pain at the CVC site, radiated to her shoulder and neck. This made it impossible to administer hydration and PN. A few days later, vascular surgery was performed to remove the left central venous access and insert a centrally inserted central catheter in the right subclavian vein. Macroscopic evaluation of the device showed a probable abnormal subcutaneous fibrinous reaction related to the underlying disease (Figure 2). The authors hypothesised that the fibrinous reaction might be linked with connective tissue fragility, inflammatory changes, and abnormal wound-healing patterns in hEDS.

Figure 2: Non-cuffed tunnelled central venous catheter.
A) and B) Macroscopic evaluation of the device showed an abnormal subcutaneous fibrinous reaction related to the underlying disease.
The following month, the patient weighed 61 kg and had a BMI of 20.6 kg/m². Due to a sudden worsening of painful symptoms while using the CVC, she independently reduced her home PN. After discussing the case with the authors’ team and the patient, the authors decided to try enteral nutrition (EN).
A nasogastric tube was therefore inserted, and nasogastric feeding was initiated with five 200 mL boluses. After 2 weeks, given the patient’s good tolerance of EN, the centrally inserted central catheter was removed and EN continued.
However, 3 months later, an increase in episodes of hypoglycaemia and POTS, as well as a reduction in tolerance to EN boluses and water, accompanied by epigastric pain, necessitated the replacement of the nasogastric tube with a nasojejunal tube. The authors then continued EN by nasojejunal feeding for weight maintenance, initially via an infusion pump, mainly at night, and then in boluses, with good tolerance. This suggests an important role for visceral hypersensitivity and pyloric hypertonicity, which are often present in patients with hEDS and GI symptoms.
DISCUSSION
hEDS is associated with a range of GI disorders, including acid reflux, abdominal pain (particularly following food intake or during enteral feeding), and constipation. Autonomic dysregulation is also frequently observed, most commonly manifesting as POTS, chronic urinary retention due to impaired urethral sphincter relaxation (Fowler’s syndrome), and episodes of hypoglycaemia. MCAS is being increasingly reported, especially among individuals with POTS.
A growing number of patients with joint hypermobility and GI dysmotility are being referred to nutrition support teams for management of weight loss and malnutrition.4
Several interrelated mechanisms may contribute to altered GI function in hEDS:5
- Altered compliance of the GI wall and mechanoreceptor dysfunction: Increased elasticity (compliance) of the GI tract wall may cause excessive distension in response to a given intraluminal pressure, leading to overstimulation of mechanoreceptors.
- Dysmotility: Changes in wall compliance, elasticity, and mechanoreceptor function may also disrupt normal GI motility.
- Visceral hypersensitivity: Direct alterations in neuronal signalling have been proposed as a potential mechanism underlying visceral hypersensitivity and associated symptoms in hEDS.
- Altered vascular compliance: Venous pooling in the lower limbs, attributed to connective tissue abnormalities within blood vessel walls, has been documented in hEDS. This phenomenon may contribute to the cardiovascular and autonomic manifestations observed in these patients. Similar alterations in splanchnic circulation may occur, potentially playing a role in GI symptom development.
Non-GI mechanisms have also been proposed as contributors to symptoms in hEDS, in particular, autonomic dysfunction. Cardiovascular autonomic manifestations may include orthostatic hypotension, orthostatic intolerance, neurally mediated hypotension, and POTS. Multisystem involvement is common, and non-cardiovascular symptoms, particularly GI issues such as nausea, irregular bowel movements, abdominal pain, bloating, and constipation, are often reported. These symptoms are likely multifactorial and may arise from altered splanchnic circulation, small fibre neuropathy, or changes in vascular compliance associated with generalised tissue laxity in hEDS.
Reduced oral intake due to eating-related symptoms should prompt nutritional evaluation. Nutrition support should be provided in a multidisciplinary context. The primary approach should optimise the oral diet and eventually add oral nutritional supplements. If oral feeding is insufficient or impractical, EN is recommended. In case of intolerance or refusal of EN, a switch to PN is recommended. However, PN with hydration is primarily indicated in case of objectively demonstrated severe malnutrition with electrolyte disturbance.
A literature review was conducted using a systematic approach. The PubMed database was searched, and a manual search was added (last update August 2025). The research showed no hEDS cases treated with artificial nutrition, only several reviews about assessment, diagnosis, and management of GI symptoms in hEDS with discordant results. A standardised therapy has yet to be established, and an integrated management approach is therefore recommended.
If PN is initiated, any iatrogenic complications should prompt a reassessment of the intervention’s risk–benefit profile. Once nutritional status has stabilised, strategies to optimise oral or enteral intake and employ less invasive forms of nutritional support should be explored, with the aim of transitioning to the least invasive approach feasible over the long term.
PN may become a necessary intervention when patients face severe, life-limiting malnutrition and cannot tolerate enteral feeding. Its implementation should be accompanied by appropriate psychological support and close collaboration with the multidisciplinary clinical team.
In this clinical case, PN was crucial in restoring satisfactory nutritional status and adequate lean body mass, enabling the woman to resume normal walking and daily activities. However, the success of the parenteral support was offset by vascular complications, which were probably caused by abnormal fibrinoid reactions in the subcutaneous tissue at the device insertion site. Once this was established, the enteral option was reconsidered, despite not having been initially pursued due to the clinical situation and the patient’s preferences. Enteral feeding allows for slow, small, and concentrated delivery of nutrients. This minimises the sudden shift of blood to the gut and the gastric wall distention, reducing pain and vascular-triggered symptoms (POTS). Moreover, EN remains the most physiological option, preventing gut atrophy, is less labour-intensive and easier to manage than PN, and is significantly cheaper with shorter hospital stays compared to parenteral alternatives.
Switching to enteral support enabled the patient to regain in quality of life, as she no longer had to deal with complicated vascular access and could receive nutrition not only at home. Once the patient had accepted the feeding tube and EN had been started, the next step was to find the most suitable type of administration in accordance with the clinical presentation. Post-pyloric feeding (delivery of nutrients directly into the duodenum or jejunum) was advantageous because of her pyloric hypertonicity and visceral hypersensitivity, bypassing the dysfunctional and painful gastric phase of digestion. Moving the feeding site to the small intestine, which typically remains more functional despite gastric issues, allows for better tolerance of nutritional goals. For patients with hEDS, this route is often a critical step in preventing the need for more invasive artificial nutrition when gastric intolerance is severe. Switching from a nasogastric tube to a nasojejunal tube bypassed gastroparesis and pyloric hypertonicity, resulting in a significant improvement in pain and intestinal discomfort related to the mixture’s administration. Actually, her weight has stabilised and painful symptoms during feeding has been significantly reduced. In addition, episodes of POTS and hypoglycaemia have decreased, and there has been a marked improvement in mobility and muscle tone, with better activity tolerance.
CONCLUSIONS
GI symptoms, associated comorbidities, both physical and psychological, and nutritional challenges significantly affect morbidity and quality of life in patients with hEDS. The pathophysiology of GI symptoms appears to involve both GI and non-GI mechanisms.6
The co-occurrence of POTS, MCAS, and hEDS within the same individual is well documented, with POTS identified as a strong predictor of GI dysmotility in hEDS.
Nutritional management requires a multidisciplinary approach aimed at stabilising patients and delivering care via the least invasive methods possible, while minimising the risk of iatrogenic complications associated with clinically assisted nutrition.
Optimal care for GI symptoms, psychological comorbidities, POTS, MCAS, and nutritional issues necessitates an integrated approach involving primary care providers and multiple specialties, including nutritionists, dietitians, gastroenterologists, psychologists, cardiologists, orthopaedists, pharmacists, and physiotherapists.
Recognising these patients and understanding the implications of the disorder provides the opportunity to offer timely reassurance, arrange multidisciplinary care when needed, and minimise unnecessary investigations and therapies, thereby improving these patients’ long-term outcomes.7
In conclusion, this is the case of a young woman with hEDS, POTS, and MCAS, whose quality of life improved significantly after she started receiving artificial nutrition. This case is of particular interest, as, during the initial phase of PN, several central vascular accesses failed prematurely due to an abnormal subcutaneous fibrinoid reaction. Switching to EN was crucial in resolving the complications associated with central venous access, restoring intestinal function, and improving quality of life.
Patient Perspective
Living with hEDS felt like a constant battle against my own body, especially as my GI symptoms worsened to the point where eating became a source of pain rather than nourishment. Before starting EN, my quality of life was severely limited by profound fatigue, sleepless nights, and the anxiety of unpredictable complications and flare-ups. Transitioning to post-pyloric feeding was a significant adjustment, but the impact was transformative. For the first time in years, I am receiving consistent nutrition without the debilitating GI distress that followed every meal. This stability has restored my energy levels, allowing me to engage in daily activities that were previously impossible. While managing a feeding tube has its challenges, the ability to reclaim my life and focus on my future rather than my symptoms has been invaluable. I finally feel like I am managing my condition rather than it managing me.