Meeting Summary
The ‘Mastering the Relapsed Refractory Multiple Myeloma (RRMM) Treatment Landscape: Removing Barriers and Unleashing the Potential of CAR T’ satellite symposium took place on 14th June 2025 as part of the European Hematology Association (EHA) 2025 Congress held in Milan, Italy.
The symposium covered insights from recent clinical and real-world evidence on factors influencing CAR-T outcomes, strategies for treatment optimisation, and how age may influence the decision to offer patients CAR-T therapy. It also explored the unmet need in the heavily pre-treated triple- and quadruple-class exposed RRMM population, and the need to identify and investigate mechanisms of resistance and explore alternative targets.
During the symposium, Chair Doris Hansen and speakers Aurore Perrot and Susan Bal, all experts in the treatment of multiple myeloma (MM), outlined the challenges involved in optimising care for heavily pre-treated patients with relapsed and refractory disease, and highlighted several opportunities to optimise and improve outcomes. These involved selecting candidate patients through an optimal evaluation of baseline characteristics instead of restricting by age, establishing optimal sequencing and bridging strategies, and implementing supportive care measures that could expand the eligibility for CAR-T cell therapy to more patients.
The symposium ended with an exploration of the potential of using CAR T therapy directed towards emerging myeloma targets such as G protein-coupled receptor, class C group 5 member D (GPRC5D).
Introduction
Multiple myeloma (MM) is the second most common haematological malignancy globally (10–15% of all cases) with an increasing global incidence.1 The 5-year survival rate increased from 25% in 1975–1977 to ~60% in 2012–2018 in the USA and Europe, but despite a range of novel treatments now included in treatment guidelines, the disease remains incurable because most patients relapse or become resistant to therapy.1
Bone disease is the most common complication in MM, and 80% of patients present with bone-related events such as bone pain, fractures, and spinal cord compression at the time of diagnosis.1 The International Myeloma Working Group (IMWG) define MM diagnosis as a bone infiltration of >10% clonal plasma cells, along with at least one myeloma-defining event (renal failure, anaemia, bone lesions, hypercalcaemia).2
First-line treatment is usually autologous haematopoietic cell transplantation (HCT) in eligible patients, with a combination of daratumumab, bortezomib, and lenalidomide-dexamethasone, followed by daratumumab-lenalidomide maintenance therapy.1 After one or two relapses, relapsed refractory MM (RRMM) is more likely to be triple-class exposed (to proteasome inhibitors, immunomodulatory drugs, and and anti-CD38 antibodies).1 The next choice of therapy is determined based on prior lines of treatment and drug sensitivities, and now includes a range of immunotherapy options, including bispecific antibodies and CAR-T cell therapy.1,3
During this symposium, the speakers emphasised the progress that has been made in treating RRMM in the past two decades, with approvals of proteasome inhibitors, protease inhibitors, immunomodulatory agents, antibodies, and CAR-T cell therapy. They highlighted the complexities of optimising care for heavily pre-treated patients who present with RRMM.
This article describes approaches to address this challenge discussed in the symposium, including strategies that can help clinicians optimise the use of CAR-T cell therapy in the RRMM setting.
CAR-T Cell Therapies for RRMM: Updates from Clinical Trials and Real-World Data
There are currently two CAR-T therapies available for use in RRMM in the USA and most of Europe, both directed toward B cell maturation antigen (BCMA). Idecabtagene vicleucel (ide-cel) was approved in the USA in March 2021,4 and granted conditional marketing authorisation in the EU in August 2021,5 and ciltacabtagene autoleucel (cilta-cel) was approved in the USA in February 2022,6 and granted conditional marketing authorisation in the EU in March 2022.7 Aurore Perrot began the presentations with long-term updates from the KarMMa and CARTITUDE-1 trials of ide-cel and cilta-cel, respectively, highlighting comparable outcomes with real-world data from the USA and France.
“We now have 5-year median follow-up data for both ide-cel and cilta-cel showing deep and durable responses with CAR-T cell therapy in triple-class exposed RRMM,” she said, “and now we can see similar efficacy in real-world cohorts.” Table 1 summarises data presented showing comparisons of clinical trial and real-world efficacy data.
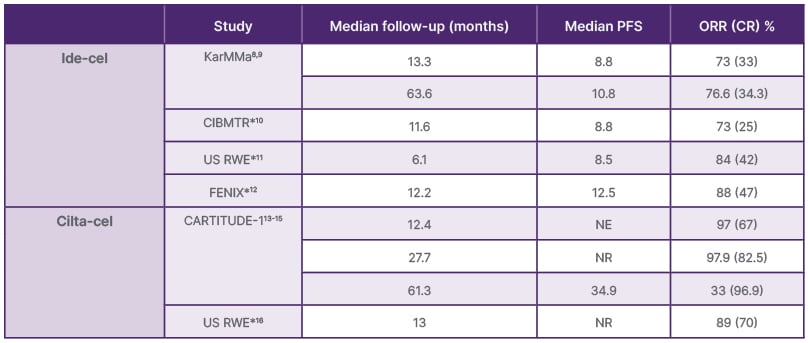
Table 1: Efficacy data for ide-cel and cilta-cel from clinical trials and real-world data.
*Real-world studies
CIBMTR: Center for International Blood and Marrow Transplant Research; cilta-cel: ciltacabtagene autoleucel;
CR: complete response; ide-cel: idecabtagene vicleucel; NE: not estimable; NR: not reached; ORR: objective
response rate; PFS: progression-free survival; RWE: real-world evidence.
Should CAR-T Cell Therapy Be Used Earlier to Optimise Treatment?
Perrot went on to discuss the use of CAR-T cell therapy in a less heavily pre-treated patient population, sharing a subgroup analysis from the KarMMa-3 trial of progression-free survival (PFS) according to the number of prior lines of treatment (Table 2).17
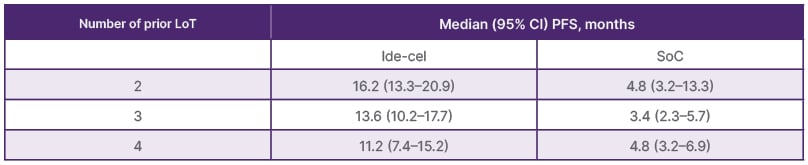
Table 2: Progression-free survival after ide-cel according to lines of prior therapy in the KarMMa-3 trial.17
*Based on Kaplan-Meier approach
ide-cel: idecabtagene vicleucel; LoT: lines of therapy; PFS: progression-free survival; SoC: standard of care.
“This analysis shows that ide-cel use in less pre-treated patients can lead to improved outcomes,” she highlighted. “PFS was longer when patients were treated earlier with ide-cel.”
Cohort 2c of KarMMa-2 (n=31) and BMT CTN 1902 (n=40) trials are also investigating CAR-T cell therapy in earlier lines of treatment.18,19
What Strategies Can Be Used to Optimise CAR-T Cell Therapy Outcomes?
Patient Evaluation
“CAR-T therapy should be considered for use as early as possible, but with special considerations for optimised patient outcomes,” said Perrot. Figure 1 shows an overview of the patient, disease, treatment history, and other factors Perrot proposed should be considered for ide-cel specifically.
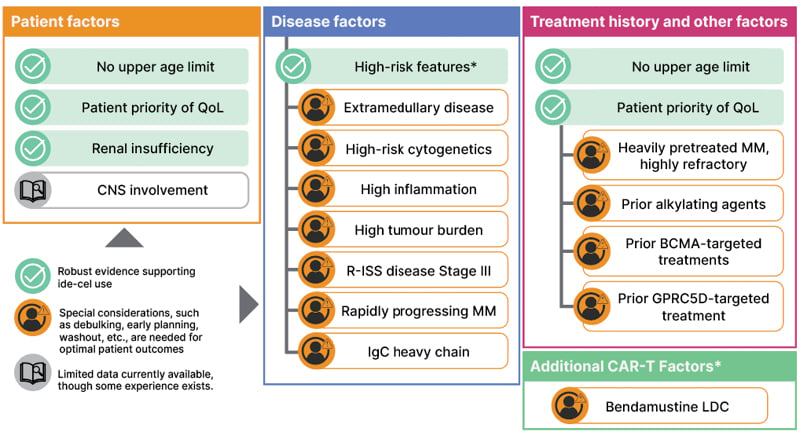
Figure 1: Eligibility for ide-cel with special considerations for optimised and long-term patient outcomes.
*High-risk features include high-risk cytogenetics, extramedullary disease, high tumour burden, and R-ISS disease Stage III.
BCMA: B-cell maturation antigen; CNS: central nervous system. GPRC5D: G-protein coupled receptor family C group 5 member D; ide-cel: idecabtagene vicleucel: LDC: lymphodepleting chemotherapy; MM: multiple myeloma; QoL: quality of life; R-ISS: Revised International Staging System; RRMM: relapsed refractory multiple myeloma.
Focusing on one of the high-risk disease features, Perrot shared data from a real-world study involving 34 patients with extramedullary disease (EMD) and 100 with paraskeletal disease (PSD) or bone-marrow-only disease who received BCMA-directed CAR-T cell therapy.20 The median PFS in patients with EMD was 9 months, compared with 21.4 months in patients with PSD and 24.2 months in patients with bone-marrow-only disease.20 “This shows that EMD with no bone involvement is associated with poorer outcomes from BCMA-directed CAR-T cell therapy compared with PSD,” said Perrot, “and we don’t yet know if a different bridging strategy is needed for these patients.”
Perrot also shared data from the KarMMa8,21 and CARTITUDE-1 trials,13 suggesting that patients who obtain a complete response from CAR-T cell therapy have better outcomes. On the KarMMa trial, patients with a partial response had a median duration of response (DoR) of 4.5 months (2.9–6.7) versus 21.5 months (12.5-not estimable) in patients with a complete response.8,21 In CARTITUDE-1, complete responders had a 12-month PFS rate of 85% compared with 62% to those who had a very good or partial response.13
Bridging Therapies
Bridging therapy is defined as anti-myeloma therapy given after leukapheresis and before lymphodepletion chemotherapy.22 All panellists agreed that an effective bridging strategy can optimise post-CAR-T outcomes. “Debulking of disease burden can lead to prolonged PFS, and there are some examples of debulking disease burden using specific antibodies,” said Perrot. They shared data from KarMMa-3 trial which showed that the median PFS was 20.7 months (11.2–not reached [NR]) in patients whose disease had decreased after conventional bridging treatment compared with 15.1 months (12.4–17.3) for patients who had no change in disease burden and 6.9 months (95% CI: 2.4–11.8) in patients whose disease increased.23 The same trend was shown with real-world data where bispecific antibodies were used as bridging therapy to debulk disease before CAR-T cell infusion.24,25
“In terms of bridging therapy, we need to take several factors into account: what the patient has been exposed to, and what they are likely to respond to,” said Bal. “We recommend using something that has the highest response rate that can be given in the first few weeks after leukapheresis, and is expected to have a high response rate.” Bal also suggested avoiding agents that are lymphotoxic, such as bendamustine. “We have experience with bispecific antibody bridging therapy, but the optimal duration and washout period remains to be seen,” they added. “As CAR-T therapy moves to earlier line therapy, we think the bridging options will generally expand.”
Immunomodulation
“There is a biological rationale for the role of immunomodulation after CAR-T cell therapy,” said Perrot, “but there are no clinical studies to prove it.”
The drug pomalidomide is thought to prevent CAR-T cell apoptosis, in addition to its cytotoxic effects, and in a small, single-centre real-world study, addition of pomalidomide after BCMA-targeted CAR-T cell therapy improved both time to progression and overall survival (OS).26
Returning to an earlier clinical case study, Perrot presented data showing a patient who only had a partial response 1-2 months after ide-cel infusion, converted into a complete response after the addition of pomalidomide for 2 months.
“This approach is not a guideline recommendation,” said Perrot, “but we know there have been several similar observations where using an immunomodulatory agent post-CAR-T has impacted on response outcomes.”
Is Age a Barrier to CAR-T Cell Therapy?
MM is a disease of older people, with a median age of onset of 69 years, and the majority of patients are diagnosed between 65–74 years, a population that is not reflected by clinical trial populations, which often exclude older patients.27
“Certainly, we have clinical trials that develop these great products, but the patients in clinical trials tend to be younger, fitter, and have fewer comorbidities, whereas the patients we see in clinical practice might not be the same,” they said. “In real-world practice, about 75% of patients on real-world studies would be ineligible for the KarMMa trials and around 54% would be ineligible for the CARTITUDE-1 study.”
Hansen said the most common reasons for exclusion were poor Eastern Cooperative Oncology Group (ECOG) performance, cytopenias, renal impairment, and prior exposure to BCMA-targeted agents. Yet, in a real-world study comparing efficacy outcomes for CAR-T therapy compared with the bispecific antibody teclistamab, a subgroup analysis found that older age (≥70 years) was associated with better OS benefit from CAR-T cell therapy than from bispecific antibodies.28
Another real-world study involving 821 patients found that older age and frailty were not associated with an inferior outcome from CAR-T cell therapy.29 In this study of ide-cel, the 6-month PFS rate was 60.4% in people aged <70 and 68.3% in those aged ≥70 years.29 OS was also similar (6-month OS rate of 82.6% in people aged <70, and 85.5% in those aged ≥70 years), and baseline characteristics and safety outcomes were comparable between groups.29
In a similar real-world study, analysis of patients aged <65 years versus ≥65 years found that PFS and OS outcomes were similar, as was the proportion of patients who obtained a complete response.30
“Is there an age or frailty threshold for CAR-T cell therapy?” an audience participant asked. “Age is truly just a number,” responded Bal. “As we see outcomes in patients >70 years, we start to understand that perhaps it’s a composite factor of their comorbidities, frailty that really matters. But even among the frail, the outcomes look comparable to clinical trial outcomes.”
The panel agreed that if patients are fit and can carry out their daily activities, they are candidates for CAR-T cell therapy. “We’ve treated some very fit >80-year-olds with CAR-T, but of course, patient selection and optimal caregiver support is very important,” said Hansen. “Renal impairment is a manifestation of RRMM, and patients with renal impairment are usually excluded from clinical studies.” Yet, in a real-world multicentre analysis of 233 patients with and without renal impairment (median age 69 versus 63 years) ide-cel had comparable efficacy and safety, suggesting that renal impairment might not be a barrier to receiving CAR-T cell therapy in this context.31
In response to a question about whether renal function exclusion criteria should be relaxed in MM trials, Perrot said, “Perhaps we can’t relax them totally, but we could potentially decrease the cut-off for renal clearance, as in many other trials.”
A follow-up question was asked about dose optimisation or reduction strategies that might be used in these patients. Bal and Hansen shared their experiences of using strategies such as dose reduction during lymphodepletion prior to CAR-T, reducing doses of fludarabine, for example, by 20–50% depending on creatinine clearance levels. “With this approach, we’ve been able to successfully get patients even on dialysis through CAR-T cell therapy,” said Bal. “These patients do quite well, but tend to have a higher incidence of early cytopenia that tends to improve later. We need aggressive supportive care measures for these patients.”
“Older and frailer patients deserve time off from therapy too!”, concluded Hansen. “One of the benefits of CAR-T cell therapy is that it can give older and frail patients a treatment break and improved health-related quality of life.” Indeed, patient reported outcomes from the KarMMa-3 trial highlight improved global health status and physical functioning of patients on ide-cel compared with standard of care (SOC)17, and clinically meaningful health-related quality of life improvements were also observed with cilta-cel compared with SOC on the CARTITUDE-4 trial.32
What Is the Optimal Sequencing of RRMM Therapies?
In the third and final session of the symposium, Bal explored unmet needs in heavily pretreated patients with RRMM, and the impact of treatment sequencing in this population. They shared data showing that response rates after CAR-T cell therapy can be compromised if patients have received prior BCMA-directed antibody-drug conjugates or bispecific antibodies,33 whereas data from the MajesTEC-1 trial (Cohort C; n= 40; mean follow-up: 28 months) suggested favourable outcomes if bispecific treatment follows CAR-T cell therapy as salvage therapy.34 In this study, a small number of patients with RRMM (N=40) received teclistamab treatment after BCMA-directed antibody-drug conjugate or CAR-T cell therapy. “Although PFS was poor at 4.5 months,” said Bal, “those patients who did respond had a manageable and good duration of response of about 15 months.”34 Real-world data also suggest that the use of a bispecific antibody after relapse on CAR-T cell therapy results in better outcomes compared with immunomodulatory or other salvage therapy.35
The IMWG now recommends sequencing an anti-BCMA CAR-T cell therapy before use of bispecific antibodies or ADCs, assuming equal access and eligibility, said Bal, “because patients who receive ‘one-and-done’ CAR-T cell therapy are less likely to experience T cell dysfunction and loss of tumour antigen, allowing for salvage therapy later”.
Can We Further Optimise CAR-T Cell Therapy with Additional Therapeutic Classes?
Relapse after CAR-T cell therapy can be caused by loss of tumour antigen, T cell exhaustion or dysfunction, or an immunosuppressive tumour environment. Target antigen loss is common after BCMA-directed therapies, although less frequent after CAR-T cell treatment (<10%) than other modalities.37-40
An alternative treatment target is G-protein coupled receptor family C group 5 member D (GPRC5D), an orphan receptor expressed on MM plasma cells.41 Bal shared data for an anti-GPRC5D bispecific antibody, talquetamab, which has shown durable efficacy and safety responses in T cell redirecting (TCR)-treated and TCR-naïve patients with RRMM (median PFS of 7.5–11.2 months in Phase I and II trials).41 “Several real-world studies with very short follow-up suggest similar response rates,” said Bal, “but long-term durability remains to be seen.”
GPRC5D could have distinct advantages over BCMA-targeted therapies because its protein expression is limited to plasma cells,41 and GPRC5D-directed CAR-T cells are also in development, with several candidates such as arlocabtagene autoleucel in active clinical trials. Bal described a real-life case from her clinic who received a GPRC5D-directed CAR-T cell therapy as part of a clinical trial, and continues to be in remission at 24 months of follow-up.
“Infections are key determinants of quality of life for patients with RRMM,” stated Bal. “Because GPRC5D is expressed on myeloma plasma cells and has a relative B cell-sparing effect, we see the total number of high-risk infections is much lower,” said Bal.
To emphasise this point, GPRC5D-directed bispecific antibody talquetamab (20.5%) has a lower grade 3/4 infection rate than BCMA-targeted bispecific antibodies (~40–55%).43-45 BCMA CAR-T therapy8,13 is comparable to GPRC5D BsAbs at around 20%.46
“The most exciting strategy is combining targets,” said Bal. “About 70% of patients will express both targets, but there are clones that are BCMA or GPRC5D null, so by using these products together, we can improve response rates.”
Bal concluded that if patients have equal access to both bispecifics and CAR-T therapy, we recommend treating with CAR-T cells first based on the data we’ve reviewed. Switching targets and using agents with novel mechanisms of action may be more optimal following BCMA-directed CAR-T therapy.”