Meeting Summary
The ‘Multi-immune modulation: a paradigm shift in understanding and treating immune thrombocytopenia (ITP) and warm autoimmune haemolytic anaemia (wAIHA)’ symposium took place on 12th June 2025 as part of the European Hematology Association (EHA) Congress held in Milan, Italy.
The symposium covered the immune pathophysiology, unmet clinical needs, and emerging therapies for primary ITP and wAIHA, two rare autoimmune conditions that share a common mechanism of immune dysfunction and chronic inflammation.
During the symposium, Chair Waleed Ghanima, Østfold Hospital, Sarpsborg; University of Oslo, Norway, and the speakers Alessandro Lucchesi, Romagna Institute for the Study of Tumors, Meldola, Italy; and Maria Eva Mingot-Castellano, Virgen del Rocio University Hospital, Sevilla, Spain, all experts in the treatment of autoimmune cytopenias, outlined how many current treatments do not target the complex immune-driven disease mechanisms underlying ITP and wAIHA and, as a consequence, are limited in delivering many of the clinical outcomes and impacts that matter most to patients living with these diseases.
The symposium finished with an overview of emerging therapies targeting multiple pathways to attain multi-immune modulation in ITP and wAIHA towards comprehensive disease control.
Introduction
ITP and wAIHA are rare autoimmune cytopenias characterised by complex immune dysregulation and chronic inflammation.1-5 ITP has an incidence of three per 100,000 person-years3 and is characterised by low platelet counts caused by immune-mediated platelet destruction and impaired platelet production.1-3 It can be primary (80% of cases) or secondary to haematological malignancies or existing autoimmune diseases.1
wAIHA is a serological subtype of AIHA characterised by red blood cell haemolysis due to autoantibodies and/or complement activation.4,5 AIHA has an incidence of 1.77 cases per 100,000 per year, and wAIHA is the most common form, accounting for two-thirds of cases.4
ITP is characterised by increased bleeding but patients also present with fatigue, anxiety6,7 and cognitive dysfunction.8 wAIHA is characterised by anaemia, but patients also present with fatigue, dizziness, syncope and exertion induced dyspnoea.5 Both diseases are associated with an increased risk of thromboembolism.5,9,10
Consensus- and/or evidence-based guidelines11-14 recommend the use of immuno-modulating agents and thrombopoietic agents like romiplostim, eltrombopag, and avatrombopag in ITP,11-13 and erythropoietin in wAIHA.14 Immuno-modulating agents commonly used are corticosteroids, intravenous Ig, and rituximab.11-13 Rituximab is used as a second-line treatment in both conditions, and a growing range of immune-targeted treatments may be used in second- or third-line therapies, either alone or in combination with other drugs.11-14 Splenectomy is an option in patients who have relapsed.11-14
All current treatments have limitations in their combined safety and efficacy profile, and there remain unmet treatment needs for both conditions, namely the failure to address underlying disease mechanisms15 and the impact on quality of life.6-8
The symposium reviewed the latest understanding of the immunological drivers of ITP and wAIHA, identified unmet clinical needs and treatment burdens, and discussed the potential of emerging multi-immune modulating treatments to improve clinical outcomes.
Immunological Drivers of Autoimmune Cytopenias
The symposium began with Lucchesi explaining how chronic inflammation is a key driver of autoimmune cytopenias: “Even if chronic inflammation initially represents a way for cells to survive in the face of sustained pathogenic stimuli, if prolonged, it becomes a persistent and maladaptive immune dysregulation that can lead to phenomena, such as cytopenias, disruption of immune tolerance, and end-stage fibrosis. All these phenomena are driven by activation of the innate immune system, and, specifically, the NLRP3 (NOD-, LRR- and pyrin domain-containing protein 3) inflammasome.”
Lucchesi outlined how autoimmunity is also driven by other mechanisms, such as molecular mimicry, which occurs when viral antigens resembling human-derived proteins cause B and T cells to recognise both viral and self-antigens.16 A further mechanism is bystander activation, where self-antigens are released into an inflammatory environment and presented by antigen-presenting cells to autoreactive T cells.16
Once inflammation is established, there is continuous disruption and damage of tissues, causing epitope spreading, in which cryptic self-antigens derived from tissue damage are presented by antigen-presenting cells17, leading to “a loop of inflammation that keeps autoimmunity constantly active,” said Lucchesi.
Cells of the innate immune system respond to these tissue insults by recognising damage-associated molecular patterns, such as mitochondrial DNA, reactive oxygen species, and haem, through specialised toll-like receptors18 and Nod-like receptors (NLR).19 Of the NLRs, NLRP3 is most studied because of its involvement in autoinflammatory disorders.19-21 NLRP3 responds to damage-associated molecular patterns, forming an inflammasome that triggers release of proinflammatory ILs, which in turn cause caspase-mediated pyroptosis, a form of cell death.19-21 “The activation of the inflammasome can normally self-limit, but it becomes persistently primed in chronic stress,” said Lucchesi.
The NOD-, LRR- and Pyrin Domain-Containing Protein 3 Inflammasome
“NLRP3’s role in pyroptosis is particularly relevant because it is the form of cell death that we usually find in vascular disorders such as atherosclerosis,” said Lucchesi. “This is important because ITP and wAIHA are not only cytopenias but also thrombotic disorders.”
Lucchesi emphasised how NLRP3’s role in ITP is further evidenced by its elevated expression aligning with T cell dysfunction in ITP pathogenesis22 and its activation (alongside other pro-inflammatory markers) in peripheral blood mononuclear cells in ITP.23 Normalisation of these inflammatory markers in patients with ITP in remission further suggests a dependency on inflammation.24
“Inflammasome activation can then spread to myeloid cells via two particular mechanisms, the release of mitochondrial DNA and haemoglobin, and this loop sustains inflammation,” said Lucchesi. “Once the myeloid cells are involved, this can activate platelets again, which act as innate immune cells in a vicious cycle that we need to break with therapy.”
In wAIHA, NLRP3 is activated by free circulating haem, causing both an impaired immune response and inducing complement.25 In the first mechanism, activated NLRP3 triggers caspase-1 and the release of ILs (IL-1β and IL-18), leading to inflammation and tissue injury.25 As an example, Lucchesi shared a case report of an 82-year-old male with wAIHA who experienced a spike in IL-18 during a haemolytic crisis, and an absence of elevated IL-12, highlighting a skewed immune response in wAIHA.26
In this second mechanism, the complement-induced membrane attack complex can transfer from the surface of complement-opsonised particles to the plasma membrane of macrophages, activating the NLRP3 inflammasome and driving crosstalk between adaptive and innate immunity.27
“We know that molecular mimicry can lead to mature plasma cells releasing increasing amounts of autoantibodies,” said Lucchesi. “These antibodies can trigger complement and formation of membrane attack complex, leading to intravascular haemolysis, but this second mechanism of epitope spreading can also trigger inflammation.”
Thrombo-inflammation
Another key therapeutic target in ITP and wAIHA is thrombo-inflammation, the inflammatory response in blood vessels following injury, infection, or non-infectious inflammatory triggers.28 Thrombo-inflammation involves key immune cells mediated by the inflammasome, including platelets, macrophages, neutrophils, and the endothelium, and the formation of neutrophil extracellular traps (NET).29 While important in protecting from infection, NETs can cause thrombo-inflammation if caused by sterile inflammation.30 Moreover, the NLRP3 inflammasome can induce neutrophils to produce NETs, but the NETs themselves can also enable NLRP3 inflammasome activation and subsequent IL release, resulting in a thrombo-inflammatory loop.30
“Complex and profound immune dysregulation is a key driver of both ITP and wAIHA. Both diseases share mechanisms, including autoantibody production, T cell dysregulation, and central pathways, including NLRP3-driven inflammasome activation,” concluded Lucchesi. “It is important to target the engine, not just the exhaust.”
Clinical Impact of Immune Dysregulation in Immune Thrombocytopenia and Warm Autoimmune Haemolytic Anaemia
The second part of the three-part presentation highlighted the key clinical considerations of ITP and wAIHA, beyond the usual measures of thrombocytopenia and anaemia. “We have to start to measure what matters to patients to try to improve their quality of life,” said Mingot-Castellano. “We have to talk about fatigue, which is common to both diseases, as well as risk of thromboembolism, treatment-associated adverse effects, increased incidence of other immune and haematological diseases, and the psychological and social impact.”
Mingot-Castellano emphasised this point with data from the ITP World Impact Survey (I-WISh), which surveyed patients with ITP (n=1,507; mean age: 47 years; 65% female; median ITP duration: 5 years) and haematologists or haemato-oncologists (n=472) who treat patients with ITP across 13 countries.6 These data revealed that patients and doctors have different concerns and perspectives regarding the burden of ITP, with the most common symptoms experienced by patients emphasised as fatigue, petechiae, and bruising, while physicians focused on hearing about fatigue less often.6 Patients with ITP also reported that fatigue has a significant impact on daily living, with aspects affected ‘most of’ or ‘all the time’ cited as energy levels (48% of adults; n=41/86), ability to concentrate on everyday activities (40%; n=35/87), and capacity to exercise (43%; n=37/87).31 Adults living with wAIHA also report fatigue as a core symptom, with several participants noting it as their most bothersome symptom.32
“When patients come to the door and say they feel fatigued, we might say it’s not related to platelet counts, but nowadays we can absolutely say it’s related to inflammation,” said Mingot-Castellano. One challenge is that, although fatigue may be driven by the underlying immune dysregulation in ITP and wAIHA, it is also influenced by interaction with medical comorbidities, lack of activity (perhaps restricted due to bruising and bleeding risk), psychological symptoms, and cognitive factors such as illness beliefs, perceived stress, and mood.33
“It is important to try to find the biological explanations and consider how we can develop treatments to intervene here,” said Mingot-Castellano, “but we must not forget to listen and to give tools to patients to help with impacts such as anxiety and depression.”
Thromboembolism
Moving on to risks associated with thromboembolism, Mingot-Castellano shared data from a USA study of patients with different autoimmune disorders (n=98,308) evaluating the incidence of venous thromboembolism (VTE).34 This showed that the risk of VTE is elevated within the first 90 days following diagnosis with ITP and AIHA, with an adjusted hazard ratio (HR) of 11.99 and 6.04 for 18–40 and 41–64 year age groups for AIHA, respectively, and an adjusted HR of 7.57 and 4.89 for 18–40 and 41–64 year age groups with ITP, respectively.34
The factors behind this increased VTE risk in people with ITP include a delay in clot formation in patients with ITP35 and resistance to fibrinolysis due to increased plasminogen activator inhibitor-1 levels.36 In wAIHA, platelet activation and aggregation are thought to be driven by NET formation.37
“You might think this increased risk is down to cardiovascular risk factors,” said Mingot-Castellano, “but my opinion is this reflects the inflammation at the very beginning of the disease, and we need to treat it early to prevent chronicity.”
Other Clinical Impacts
Mingot-Castellano next moved on to the risk of infections in patients with ITP and wAIHA. In a Danish health registry study that analysed long-term survival, comorbidities, and causes of death in 1,762 patients with chronic ITP and 74,781 age- and sex-matched comparators, infection was one of the leading causes of death in ITP, with a 5-year cumulative mortality of 1.7% (95% CI: 1.2–2.5) compared with 0.71% in comparators (95% CI: 0.64–0.78).38 A French study assessing infection risk in patients with AIHA receiving rituximab also reported a high incidence of hospitalisations with infection: 6-month cumulative incidence of hospitalisations with infection was 17.6% (95% CI: 15.2–20.0) with a 30-day overall mortality rate after hospitalisation of 12.5% (95% CI: 8.0–18.0).39 Around 40% of these infections were pulmonary, and patients aged >70 years treated with corticosteroids were at the highest risk.39
“These patients receive corticosteroids for a long time, and strategies that minimise toxicity are crucial for reducing mortality,” said Mingot-Castellano. “We must look at treatments to prevent infections and osteoporosis and to treat our patients very soon to prevent them from staying on steroids.”
Patients with ITP also experience a higher incidence of fractures within the first year following diagnosis (adjusted cause-specific HR: 1.82 [95% CI: 1.39–2.40] for primary ITP and 2.78 [95% CI: 1.58–4.91] for secondary ITP). Risks remained elevated for 2–5 years but diminished in the following years.40
In the third and final part of the presentation, Mingot-Castellano presented data on the prevalence of mental health disorders in autoimmune cytopenias, highlighting that patients with ITP are nearly four times more likely to be at risk of mental health disorders (2.3% versus 0.7%; cause-specific HR: 3.57),41 with an increased risk of depression (1.2% versus 0.3%; cause-specific HR: 3.53), and similar findings for anxiety and fatigue, although these risks diminished after 1–5 years.41 There are fewer data for wAIHA, but most patients report a significant impact on their daily living, work, leisure, and sleep.42
To conclude, Mingot-Castellano outlined several strategies for improving outcomes in patients with ITP and wAIHA. “Traditional metrics such as platelet counts and haemoglobin levels are crucial for disease monitoring but do not capture the broad impact of ITP and wAIHA on patient lives,” said Mingot-Castellano. “The emotional, social, and daily life challenges highlight the need for a holistic disease management approach that prioritises mental health and quality of life. The complex immune pathophysiology of ITP and wAIHA extends beyond clinical outcome; the right treatment should effectively address the full spectrum of the disease.”
The Potential Role of Multi-Immune Modulation
In the final presentation, Ghanima provided an overview of the current and emerging treatment landscape for ITP and wAIHA. There are currently no approved therapies for wAIHA, but the main treatment strategy is similar to ITP, with immune modulation playing a key role alongside stimulation of platelets or erythrocytes. The current treatment recommendations for both conditions are summarised in Table 1.11-14
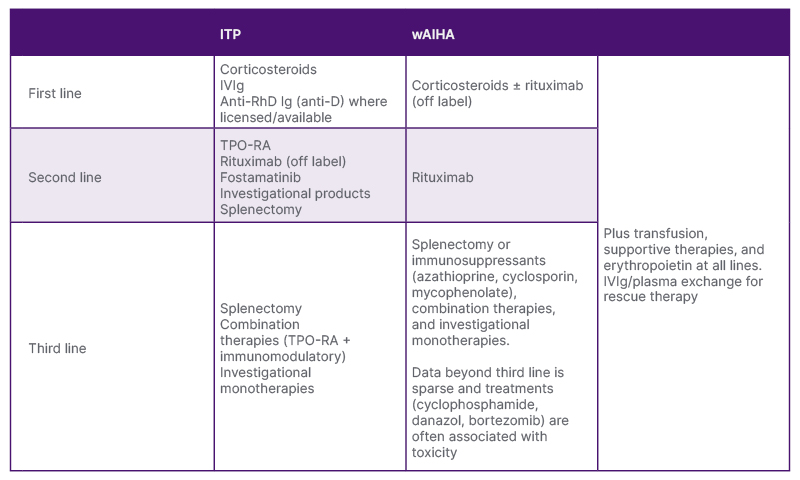
Table 1: Current treatment recommendations for immune thrombocytopenia and warm autoimmune haemolytic anaemia.11-14
ITP: immune thrombocytopenia; IV: intravenous; RhD: Rhesus D; TPO-RA: thrombopoietin receptor agonist; wAIHA: warm autoimmune haemolytic anaemia.
Despite having so many treatment options, not all patients respond, and there remain significant unmet treatment needs. To illustrate this, Ghanima shared more data from the I-WISh study on patient satisfaction with current treatments. The highest-rated treatment, rituximab, had a satisfaction rate of 79%, followed by 76% for thrombopoietin receptor agonists, 69% for intravenous Ig, 53% for corticosteroids, and 38% for patients who had undergone splenectomy.43Moreover, Ghanima reiterated fatigue as a major unmet need, with 73% and 65% of patients considering fatigue to be severe at diagnosis and at survey completion, respectively.6 “These are supposedly treated patients,” said Ghanima, “yet half of them had fatigue, and most said it was severe.”
Ghanima went on to outline emerging therapies in clinical trials that are targeting mechanisms within the complex immune dysregulation pathophysiology of ITP and wAIHA and have the potential to resolve some of these unmet needs (Table 2). These include agents targeting B cell activation and phagocytosis via spleen tyrosine kinase (Syk),46,62 B cell activating factor, and/or a proliferation-inducing ligand, preventing proliferation and activation of B cells and their differentiation into plasma cells.63,64
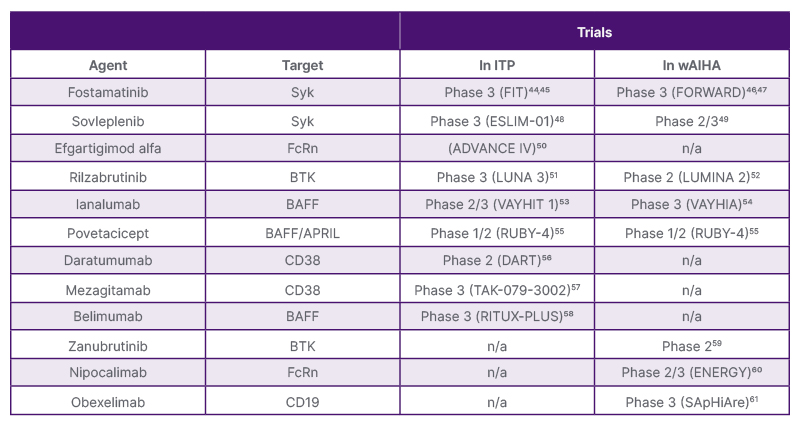
Table 2: Immune-targeted therapies in development for immune thrombocytopenia and warm autoimmune haemolytic anaemia.
APRIL: a proliferation-inducing ligand; BAFF: B cell activating factor; BTK: Bruton’s tyrosine kinase; CD: cluster of differentiation; FcRn: neonatal Fc receptor; ITP: immune thrombocytopenia; n/a: non-applicable; Syk: spleen tyrosine kinase; wAIHA: warm autoimmune haemolytic anaemia.
Other agents target cluster of differentiation (CD)38 on plasma cells65 and neonatal Fc receptor (FcRn) to prevent Ig recycling.66 In wAIHA, anti-CD19 is being explored in addition to immune targets shared with ITP.63,65
Bruton’s tyrosine kinase (BTK) is a key signalling node in innate immunity and inhibits B-cell activation without depletion. It could be a promising multi-immune modulator for both ITP and wAIHA.67,68 “BTK is a very interesting target present in the macrophage, B cell, neutrophil, and platelet,” said Ghanima. “In the macrophage, blocking BTK will interrupt platelet phagocytosis; in the B cell, it will reduce activation and antibody production; mast cell and in the platelet itself, it will inhibit the inflammasome, which we believe is so related to the fatigue we see even in treated patients.”
Many of these trials are ongoing, and results to date have shown durable responses for several emerging agents. In ITP trials measuring platelet counts (≥50×109 /L at the fourth of six visits), the Syk inhibitors fostamatinib and sovleplenib had response rates of 18% versus 2% (n=101 versus n=49; p<0.0030)69 and 48% versus 0% (n=126 versus n=62; p<0.0001)70 versus placebo, respectively. A response rate of 22% versus 5% was seen for the FcRn-blocking drug efgartigimod alfa (n=78 versus n=40; p=0.032),71 and the response for the BTK inhibitor rilzabrutinib in the LUNA-3 trial was 23% versus 0% (n=133 versus n=69; p<0.0001).72
In wAIHA, the primary response endpoint was not reached for fostamatinib,46 but 48% of patients responded to sovleplenib.62 In the ongoing LUMINA trial, an interim analysis reported a durable response in 41% of patients receiving rilzabrutinib as monotherapy or with corticosteroids.68
Of note, patients with ITP receiving rilzabrutinib in the LUNA-3 trial reported a clinically meaningful improvement in fatigue. The least squares mean differences in physical fatigue for rilzabrutinib versus placebo were 8.08 (95% CI: 1.82–14.34; p=0.01) at Week 13 and 12.0 (p=0.0003) at Week 25.72 “There was a substantial improvement in fatigue already at 13 weeks and maintained at 25 weeks versus placebo,” said Ghanima. “But importantly, if we look at those who did not have a durable platelet response, they still had a good improvement in fatigue, probably because we are targeting the inflammation.” Ghanima went on to share data showing that, among the available second-line therapies, only sovleplenib and rilzabrutinib have been reported as significantly improving fatigue compared with placebo or standard of care.46,62,70,72
“The goal of therapy for autoimmune cytopenias should not only be to improve platelet and red blood cell counts but also to restore and maintain patient health-related quality of life,” concluded Ghanima. “Emerging therapies targeting multi-immune mechanisms of these diseases could help to improve patient outcomes and disease control.”
| Adverse events should be reported. Reporting forms and information can be found at www.mhra.gov.uk/yellowcard or search for MHRA Yellow Card in the Google Play or Apple App Store. Adverse events should also be reported to Genzyme Corporation on https://ae.reporting.sanofi/ or the FDA at 1-800-FDA-1088 or www.fda.gov/medwatch. |
MAT-GLB-2600452 – 1.0 – 03/2026







