Author: Evangelia Papadavid,1 Maarten Vermeer2
1. Optimum Dermatology and the National and Kapodistrian University of Athens, Greece
2. Leiden University Medical Center, the Netherlands
Disclosure: Papadavid and Vermeer have been engaged by Helsinn Healthcare SA (“Helsinn”) to present as speakers at the Helsinn-sponsored symposium titled “Targeting Skin, Shaping the Microenvironment: The Impact of Cl-gel in MF-CTCL” at EORTC Cutaneous Lymphoma Tumour Group Annual Meeting on October 16, 2025, Athens, Greece. This article is intended for healthcare professionals only.
Disclaimer: Prescribing Information for LEDAGA® (chlormethine hydrochloride) can be found here. Local prescribing conditions may vary. Please refer to prescribing information in your country of practice. Always consult local prescribing information in country of practice as information may vary. Adverse Events Reporting information can be found at the bottom of this article.
Support: The content of this article is based on a non-promotional symposium. The symposium and publication of this article were made possible by the contribution of Helsinn Healthcare SA.
Acknowledgements: Writing assistance was provided by Helen Boreham, HB Medical (UK) Ltd, Wetherby, UK
Meeting Summary
Cutaneous T cell lymphomas (CTCL) are a heterogenous group of rare malignancies primarily involving the skin, of which mycosis fungoides (MF) is the most frequent type. During this symposium at the 2025 annual European Organisation for Research and Treatment of Cancer (EORTC) Congress, leading experts in dermatological oncology discussed the impact of chlormethine gel (CL-gel) in treating patients with MF-CTCL and explored how targeting the skin can reshape the tumour microenvironment (TME). Evangelia Papadavid, Chair of the EORTC Cutaneous Lymphoma Tumour Group; and Professor of Dermatology-Venereology at the National and Kapodistrian University of Athens in Greece, opened the session with a deep dive into underlying clinical and TME insights in MF-CTCL. She went on to present interim findings from a translational study in patients with MF treated with CL-gel, emphasising the link between tumour immune cell infiltration, dermatitis, and clinical outcomes. Results from a further study exploring CL-gel’s effect on cluster of differentiation (CD)8+ T cell populations and changes in the TME during treatment were discussed by Maarten Vermeer, Head of Dermatology at Leiden University Medical Center in the Netherlands. Collectively, this evidence presented at EORTC 2025 demonstrates that topical chlormethine is able to activate the TME in patients with MF-CTCL and highlights the key link between dermatitis and clinical response.
The Evolving Lens: Rethinking Mycosis Fungoides Cutaneous T Cell Lymphoma Through Clinical and Tumour Microenvironment Insights
MF is the most frequent subtype of primary cutaneous lymphomas but remains a rare disease, with an estimated incidence ranging from 0.3–0.9 per 100,000 cases per person-years.1 The early stages of MF are characterised by inflammatory patches and plaques.2 Although most cases are indolent, approximately 30% of patients will progress to an advanced disease stage, at which point their prognosis diverges, with a median overall survival of less than 5 years.2-4
The TME in MF is a highly structured ecosystem containing malignant cells surrounded by diverse non-cancerous cells, which plays a critical role in disease progression, as well as treatment and resistance. As Papadavid explained, there is a need to connect this underlying TME biology to clinical staging in order to guide disease management and aid in the discovery of biomarkers that may improve clinical decision-making.
The TME is not a static bystander in MF, Papadavid emphasised; instead, the immune landscape undergoes an evolution from immunosurveillance to immunoescape as the disease advances (Figure 1). In the microenvironment of normal skin, tumour growth is restrained, while MF progression reflects a gradual loss of this immune surveillance. Early-stage MF is characterised by low numbers of malignant cells with immune surveillance mediated through Th1 skewed immunity, high CD8+ T cells, and M1 macrophages, which control tumour activity.5 In contrast, advanced stages of the disease are marked by a high number of tumour cells and immune evasion, with a dominant Th2 response. This is associated with a loss of CD8+ T cells, an increase of immunosuppressive cells such as Th2, regulatory T cells (Tregs), and M2 macrophages, and B cell aggregates and angiogenesis.5 Cancer-associated fibroblasts additionally promote fibrosis, and there is upregulation of checkpoint molecules.5 Understanding this evolution of the TME in MF is important because it enables stage-adapted and biology-guided therapy selection, Papadavid stressed.
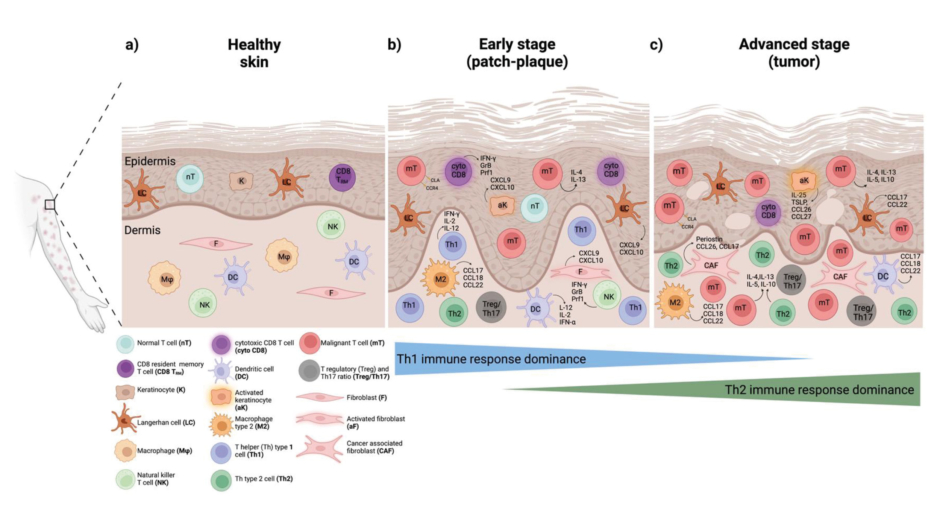
Figure 1: Evolution of the immune landscape in mycosis fungoides.5
aF: activated fibroblast; aK: activated keratinocyte; CAF: cancer associated fibroblast; CCL: chemokine ligand; CCR4: C-C chemokine receptor 4; CD8: cluster of differentiation 8; CD8 TRM: CD8 resident memory T cell; Cyto CD8: cytotoxic T cell; CLA: cutaneous lymphocyte antigen; CXCL: chemokine; DC: dendritic cell; F: fibroblast; K: keratinocyte; GrB: granzyme B; IFN: interferon; LC: Langerhan cell; M: macrophage; M2, macrophage Type 2; mT: malignant T cell; NK: natural killer T cell; nT: normal T cell; Prf: perforin; Treg/Th17: T regulatory and Th17 ratio.
Skin-directed therapies (corticosteroids, CL-gel, and light-based approaches such as psoralen plus UVA or narrowband-UVB and localised radiotherapy) are first-line treatment options for the early stages of MF. Systemic treatments (resiquimod and the newer intravenously administered monoclonal antibodies such as mogamulizumab, pembrolizumab, and lacutamab) represent first-line options for patients in the advanced stage. Both conventional and more recently developed therapies for MF act to remodel the TME via Th1 activation and immune reprogramming.6-16 These effects therefore underscore a shared biological goal, which is to re-establish anti-tumour immunity. As Papadavid explained, although drug-induced dermatitis or rash is usually considered an adverse effect of these treatments, it is generally caused by the overreaction of the immune system, and might therefore equate to beneficial effects on the TME. A study of the rash associated with mogamulizumab treatment found lesions infiltrated by CD8+ T cells, macrophages, and monocytes, together with tumour cell reduction and the recruitment of new benign closes. Notably, this skin rash also tracked with better clinical outcomes, Papadavid pointed out. Patients developing skin rash during mogamulizumab treatment had significantly higher overall survival compared to those who did not.14
However, as Papadavid acknowledged, in clinical practice, it can prove difficult to distinguish between treatment-related inflammation, such as dermatitis, and true disease progression. Clinical tools with the capacity to differentiate between the two are lacking, and existing scales such as the Modified Severity-Weighted Assessment Tool (mSWAT) and Composite Assessment of Index Lesion Severity (CAILS) may misinterpret inflammation as progression. Skin biopsies have limited utility due to disease heterogeneity and small sampling area, and there is also significant clinical and histological overlap between MF and other inflammatory skin diseases.
Linking Tumour Immune Infiltration and Clonality to Clinical Outcomes in Mycosis Fungoides Treated with Chlormethine Gel
In the second part of Papadavid’s presentation, she showcased results from a translational study recently conducted by her research group, which linked TME changes to clinical response and dermatitis in patients with MF treated with CL-gel. CL-gel is currently recommended in all major international guidelines for the first-line treatment of adult patients with MF Stages IA–IIA, and can be used to treat patches and plaques in patients with advanced disease (Stages IIB–IVB) in combination with other treatments.19-23 As Papadavid explained, CL-gel exerts its antitumour effects primarily through DNA alkylation, which leads to impaired DNA repair and apoptotic death of malignant cells. However, it also has other potential immune stimulatory mechanisms, which include: counteracting the tumour protective immunosuppressive TME; induction of an inflammatory cytotoxic effect via dermatitis; and dendritic cell-driven epidermal immunotherapeutic response following apoptosis.24
The study in question enrolled 14 adult patients with early-stage IA-IB MF, 12 of whom were treated with CL-gel and two of whom were controls. Patients were followed for up to 18 months, with clinical efficacy evaluated at specific time points. Skin biopsies were taken at baseline, on development of dermatitis, and at clinical response in order to characterise treatment-induced changes in the skin TME at a single cell level. The study also sought to identify predictive biomarkers of dermatitis and clinical response. Single-cell mass cytometry employing a panel of 48 metal-conjugated antibodies on single cells isolated from skin biopsies
was used to characterise 52 different immune phenotypes.
Outlining clinical efficacy results from the study, Papadavid highlighted how most patients achieved a complete response (CR) after 18 months of treatment with CL-gel. Patients initially attained stable disease and partial responses during the first 3 months of treatment, which was followed by clinical improvement to CR over time. In contrast, no control patient achieved CR over the 18-month study period. Two patients discontinued treatment due to severe dermatitis, and one patient has not yet reached the 12-month time point, reducing the overall evaluable cohort to nine patients.
In total, eight out of 12 patients (67%) developed dermatitis during the study, with a mean time to onset of 2.4 months. In the majority of patients (6/8; 75%), this dermatitis was mild or moderate in severity, and CL-gel treatment was continued. However, as previously mentioned, two patients developed severe dermatitis and discontinued treatment after the third month.
Immune profiling carried out in the 12 patients with MF before and after CL-gel treatment revealed distinct immuneprofiles. Notably, there was a shift towards a less immunosuppressive TME profile following treatment, as evidenced by significant reductions in Tregs and M2 macrophages (Figure 2).
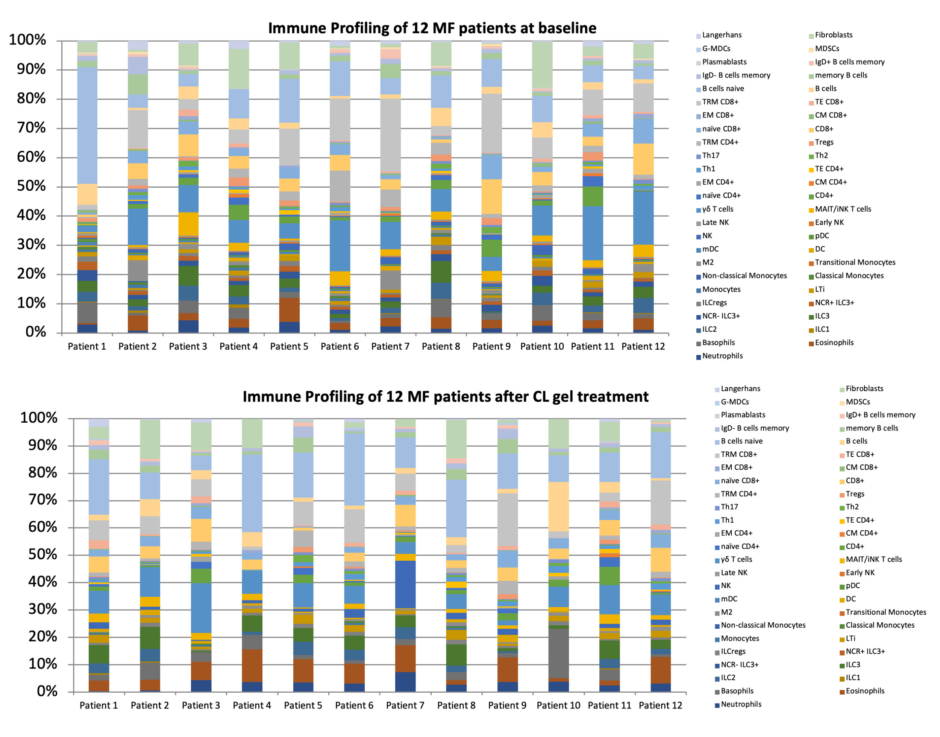
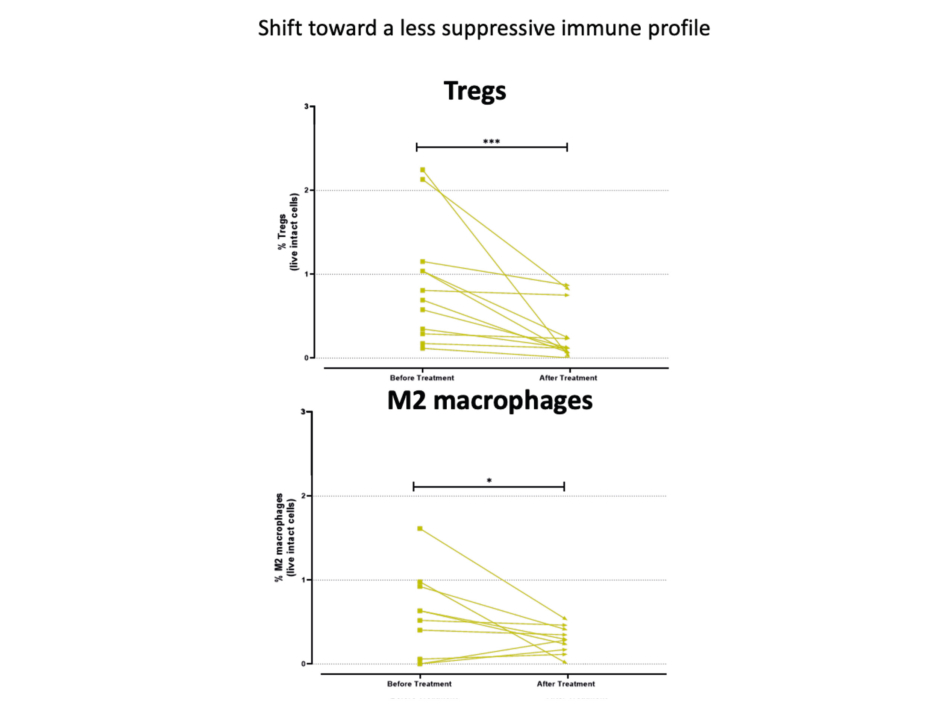
Figure 2: Shift towards a less suppressive immune profile in patients treated with chlormethine gel.
CD: cluster of differentiation (T cell); CL: chlormethine; CM: central memory T cell; DC: dendritic cell; EM: effector memory T cell; G-MDCs: granulocytic myeloid-derived suppressor cells; ILC: innate lymphoid cells; ILCregs: regulatory innate lymphoid cell; LTi: lymphoid tissue inducer cell; M2: macrophage type 2; MAIT/iNK: mucosal-associated invariant T cell/invariant natural killer T cell; mDC: myeloid dendritic cells; MDSCs: myeloid-derived suppressor cells; MF: mycosis fungoides; NCR: natural cytotoxicity receptor; NK: natural killer cell; pDC: plasmacytoid dendritic cells; TE: terminal effector T cell; Th: T helper; Tregs: regulatory T cell; TRM: tissue-resident memory T cell.
As part of the clinical assessment, it is therefore important to engage other molecular approaches and TME biomarkers to attain diagnostic accuracy, stressed Papadavid. High-throughput T cell receptor sequencing is able to quantify malignant clone frequency and provides a strong prognostic marker.17,18 Blood biomarkers such as circulating tumour DNA are also under investigation for disease monitoring. Finally, AI and other molecular tools are important elements in an integrated approach that improves the distinction between disease progression and treatment-related inflammation.
In summary, Papadavid reiterated the importance of differentiating between biologic and clinical response in patients with MF-CTLC. Biological response typically precedes visible changes. TME remodelling can therefore be seen as the first measurable therapeutic signal, while clinical resolution is the ultimate outcome. As a result, it is important to engage in biology-guided precision treatment in MF by integrating all clinical, histological, and molecular data, including clonality tracking and immune profiling, she concluded.
From an immune profile point of view, pre-treatment analysis showed that patients shared a relatively similar immune profiling across patches and plaques, suggesting a common baseline immunological architecture. However, the biological effect of CL-gel was more pronounced in patches compared to plaques, with patches showing superior immune activation and significantly reduced M2 macrophages compared to plaques.
Specific immune cell alterations were also detected in patients with CL-gel-induced dermatitis. In particular, there was a correlation between dermatitis and heightened effector immune activity and a less immunosuppressive TME. Papadavid explained that CL-gel-induced dermatitis had an effect on both innate and adaptive immunity, triggering a shift towards a less immunosuppressive TME. Natural killer (NK) cells were significantly increased, whereas immunosuppressive cells such as M2 macrophages and Tregs, as well as Th2, were significantly decreased in patients with dermatitis. The onset of CL-gel-induced dermatitis was also marked by a significant increase in total and terminal effector CD8+ T cell subpopulations and decreased regulatory innate lymphoid cell (ILCreg) frequencies, indicating an early effector response and cytotoxic immunity associated with clinical dermatitis.
Patients attaining CR with CL-gel treatment showed reduced frequencies of aberrant immune cells, including CD3+CD4+CD26-CD7- T cells and CD3+CD8+CD26-CD7- T cells. As Papadavid noted, this impact was more pronounced in the aberrant cells that were double negative and expressed a low C-C chemokine receptor Type 4 (CCR4) phenotype. The correlation of baseline immune cell levels with clinical outcome was also evaluated in this study as a potential predictive model for response. Low baseline levels of naïve CD4+ T cells, Th2, and Tregs, as well as an elevated terminal effector CD8+ T cell/Treg ratio, were found to be predictive of CR after 9 months of CL-gel treatment.
Papadavid went on to share examples of two ‘patch’ patients from this study who developed CL-gel-induced dermatitis, and where clinical pathological results could be clearly correlated to changes in the immune profile. The first patient had CD4+ MF Stage T2bN0M0B0 (IB) moderate dermatitis at Month 2. Distinguishing malignant MF cells from skin inflammation proved difficult in this case due to the lichenoid drug reaction. However, a strong immune response was evident, as represented by an increase in terminal effector CD8+ T cells and reduction in Tregs. The patient continued with CL-gel treatment and achieved CR after several months. The second patient with CD4+ MF (Stage T2aN0M0B0, IB) and dermatitis showed complete clinical and histological remission after 6 months of CL-gel-treatment. This clinical response correlated with absence of aberrant T cells on histology and restoration of a balanced TME.
In summary, Papadavid reiterated that, based on the interim analysis, CL-gel is effective in early-stage MF not only through cytotoxicity, but via meaningful immunomodulation of the TME towards a less immunosuppressive profile. Despite MF heterogeneity, M2 macrophages and Tregs decrease consistently in all patients. CL-gel-induced dermatitis is common and immunologically meaningful as it indicates a shift towards a more effective and less immunosuppressive TME. This is evidenced by increased NK and terminal effector CD8+ T cells and decreased Th2 and immunosuppressive populations such as ILCregs, Tregs, and M2 macrophages. Aberrant T cell subsets also decline after treatment, particularly CD4+CD7-CD26-CCR4- cells. Finally, Papadavid emphasised that low baseline levels of naïve CD4+ T cells, Th2, and Tregs predict a faster CR after CL-gel treatment, potentially paving the way for biology-guided precision treatment.
Changes In Tumour Microenvironment During Treatment in Cutaneous T Cell Lymphoma
The final presentation by Vermeer focused on changes occurring in the TME of MF plaques and patches during treatment with CL-gel. He began by recapping the key role of the TME in lymphoma pathogenesis, highlighting it as both a therapeutic target and component of the therapeutic response.25,26 In MF specifically, the TME provides a target for immunotherapy, as supported by both clinical observations and findings from molecular and histology studies. Notably, disease progression is accompanied by a shift from an effective tumour immune response to a Th2-dominated TME that favours immune evasion and tumour cell proliferation.26,27
In this study conducted by Vermeer and his team, 21 patients with MF Stage IA/IB received 16 weeks of open-label CL-gel treatment following an initial 6-week observation period. Suction blisters and biopsies were performed at baseline and after 16 weeks of treatment. Interstitial fluid from suction blisters was used in proteomics analysis and flow cytometry, while tissue from skin punch biopsies was analysed by haematoxylin and eosin staining, mass cytometry imaging, and high-throughput T cell receptor sequencing.
Results from suction blister analysis revealed a higher number of immune cells in lesioned versus non-lesioned skin, which decreased with 16 weeks of CL gel treatment. The specific types of cells reduced by CL-gel treatment were total CD4+ T cells, activated CD4+T cells, and CD4+CD26- T cells (tumour cells). Patients who responded to CL-gel treatment after 16 weeks were found to have a higher baseline number of activated cytotoxic T cells compared to non-responders. It is important to note that response to CL-gel can require 12 months or more of treatment, and in this study, both responders and non-responders refer to a very short treatment period.19
Analysis of blister fluid on a proteome panel showed a clustering of samples with more abundant inflammatory proteins in MF lesions at baseline, MF lesions with dermatitis, and MF lesions after 16 weeks of treatment in non-responders. Further supervised analysis of these findings revealed 33 significant results. Responders to CL-gel displayed an increase in cytokines, specifically IL12 and interferons (IFN), chemokines (CXCL9, CXCL10, CXCL11), and cytotoxic proteins (granzyme B), typical of an effective cytotoxic immune response. Reflecting on these data, Vermeer suggested that CL-gel treatment may act by increasing IL12, in turn inducing IFN, which then activates the three key cytokines. These then attract CXCR3-positive T cells and NK cells, which augment the activated cytotoxic T cell response in MF plaques and patches.
Presenting cytometry by time of flight data from the study, Vermeer explained that responders exhibited a greater reduction in immune cell abundance across all major cell types during CL-gel treatment compared to non-responders at 16 weeks. “This makes sense,” he noted, because “with effective treatment, the skin normalises” and reverts to its normal composition.
The next pertinent question addressed in the study was whether changes in the abundance of key immune cell phenotypes during CL-gel treatment were accompanied by an impact on tumours. Vermeer outlined how analysis of clonal diversity using T cell receptor sequencing in patient responders revealed a “dramatic” decrease in the size of the clonal T cell compartment after 16 weeks of treatment. Not only was the frequency of the dominant clone reduced, but absolute cell numbers also decreased considerably (Figure 3). In contrast, similar analyses in CL-gel non-responders showed that the dominant clone remained and did not decrease in frequency during treatment.
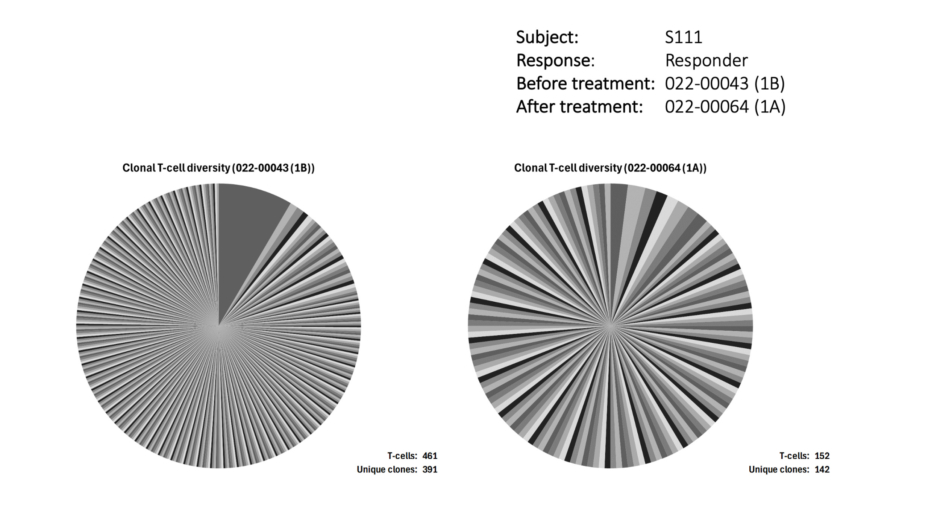
Figure 3: Clonal diversity in responders to chlormethine gel treatment.
Finally, the study considered the potential influence of the inflammatory reaction on treatment response. Vermeer presented results showing that dermatitis was significantly correlated with response to CL-gel treatment after 16 weeks (p<0.001). Time to response was also closely linked to time to first dermatitis (p=0.03).
Adverse events should be reported. Reporting forms and information for the United Kingdom can be found at www.mhra.gov.uk/yellowcard or search for MHRA Yellow Card in the Google Play or Apple App Store. Adverse events should also be reported to Helsinn Healthcare SA via email at https://[email protected]./.






