Abstract
Neurogenic dysfunction of the lower urinary tract is a complex condition with variable, often profound, and multisystem impacts that require longitudinal, multidisciplinary care that becomes more complex as patients age. This review covers key pathophysiology and outlines clinical priorities in the management of patients with neurogenic lower tract dysfunction. The current landscape of transitional urologic care is also described with attention to health system components, challenges, and patient-level tools/facilitators.
Key Points
1. Neurogenic bladder dysfunction occurs secondary to a neurologic insult and can include a wide range of urinary symptoms and their sequelae. Patients with neurogenic bladder dysfunction require lifelong multidisciplinary care.2. Preservation of renal function is the foremost clinical priority for these patients, and management of bladder emptying, continence, sexual function/fertility, prevention of infection, and screening for sexual abuse are all essential components of their long-term comprehensive care.
3. Transition from paediatric to adult care systems remains challenging with highly variable structures and success between systems and nations. Effective transition can minimise recurrent hospitalisations and escalation of care in adulthood, and necessarily involves the expertise and active participation of the patient and their caregivers.
INTRODUCTION
The term neurogenic bladder is defined as bladder dysfunction resulting from damage to the nerves that control the bladder in the central or peripheral nervous system. The term is often, mistakenly, used as a ‘diagnosis’, even though it occurs secondary to a neurological diagnosis, which could include spinal dysraphism, exstrophy, spinal cord injury, cerebrovascular accident, multiple sclerosis, iatrogenic pelvic nerve injury, etc. The impact of a neurological lesion may be impaired storage or voiding or altered bladder pressures, which may result from dis-coordinated voiding. Management of lower urinary tract neurogenic dysfunction requires attention to multiple domains of patient pathophysiology and navigation of the intersections of physiology, psychosocial environment, and health system capabilities. Longitudinal multidisciplinary care is paramount for these patients, as the character and degree of a neurologic dysfunction may evolve over time and can affect multiple systems (e.g., urological, nephrological, colorectal, orthopaedic, psychological). Involvement of psychology, physiotherapy, occupational therapy, and social work is particularly important for many of these patients. Given the complexity of pathophysiology and care models, individuals with congenital pathology require active transition between paediatric and adult care systems. This review will cover priorities in the clinical care of patients with neurogenic bladder and the current landscape of transitional care for these patients internationally.
CLINICAL PRIORITIES
Upper Urinary Tract Preservation
Historically, renal failure was a source of significant morbidity and mortality among patients with neurogenic bladder; it was the leading cause of death among patients with spina bifida >16 years old and after spinal cord injury.1,2 Though patients with spina bifida do not typically suffer congenital renal dysplasia, 50% will develop renal insufficiency within 5 years without routine urologic care.3 Patients with renal dysplasia secondary to abnormal in utero lower urinary tract mechanics are at even higher risk of renal disease. Modern series demonstrate that active management of lower tract neurologic dysfunction can substantially reduce morbidity, with severe renal impairment in as few as 1.6% of patients at one centre.4 Thus, renal preservation is the foremost clinical priority for these patients.
If neurogenic bladder is suspected, investigation often begins with uroflowmetry (if able to void volitionally) and post-void residual assessment. Adults with low-risk clinical features (suprapontine or peripheral nerve lesion, normal renal function, minimal/stable symptoms) and normal assessment can safely forego invasive testing.5,6 Importantly, the level of the neurological lesion may not correlate with clinical phenotype, so careful initial assessment is crucial. If there are abnormalities on initial testing or high clinical suspicion of neurologic pathology, quantitative evaluation of bladder mechanics via video urodynamics is recommended. Storage and detrusor leak point pressure greater than 40 cm H2O have long been used as the threshold at which bladder pressures are considered unsafe.7 However, newer analyses demonstrate increased risk of secondary vesico-ureteral reflux (VUR) and hydronephrosis with storage pressures ≥15 cm H2O,8 so many clinicians now use this finding to trigger stratification as higher risk of upper tract deterioration. Impaired compliance (<20 cm H2O), detrusor overactivity, and detrusor sphincter dyssynergia have also been associated with upper tract deterioration.6,8 In children, bladder capacity is lower and deterioration in compliance, even if absolute pressure is still within a ‘safe’ range, often prompts changes in management. In patients with established neurologic dysfunction, new or changed symptoms should prompt re-evaluation.
Renal function may be monitored with laboratory measures, ultrasonography, and functional imaging, such as nuclear renal scans. Cystatin C is the most accurate serum measure of renal function available in most clinical contexts for patients after the first year of life.9 In patients who are non-weight-bearing, serum creatinine is not an accurate measure of renal function due to reduced muscle mass but can be useful to trend if more accurate labs, such as cystatin C and chromium-51 edetic acid, are not readily available.9 Changes in renal evaluation despite stable urinary symptoms should prompt re-evaluation of bladder dynamics and consideration of more intensive bladder management. Multisystem effects of these patients’ primary pathologies (e.g., kyphosis resulting in respiratory compromise, feeding difficulties, bone resorption), non-urologic comorbidities (e.g., obesity, cardiovascular disease), and urologic disease beyond high bladder pressures (e.g., urinary tract infections [UTI] and urolithiasis) also pose threats to renal function.9
There is historic controversy regarding reactive versus proactive management of patients with congenital neurogenic bladder. Reactive strategies involve observation without intervention until new symptoms, changes in bladder dynamics, or renal dysfunction develop.10 This strategy theoretically confers lower risk of iatrogenic injury or infection, less risk of medical trauma and its sequelae, and less burden on caregivers and cost to the family and health system during observation. Proactive management strategies include urodynamics in the first months of life regardless of voiding symptoms, early initiation of anticholinergics and clean intermittent catheterisation (CIC), and surveillance renal function testing and ultrasound every 6–10 months (Figure 1).10,11 Reviews in recent years have demonstrated significantly lower rates of secondary VUR, UTIs, and renal deterioration with proactive strategies, and current European Association of Urology (EAU) guidelines advocate for proactive management.10-12
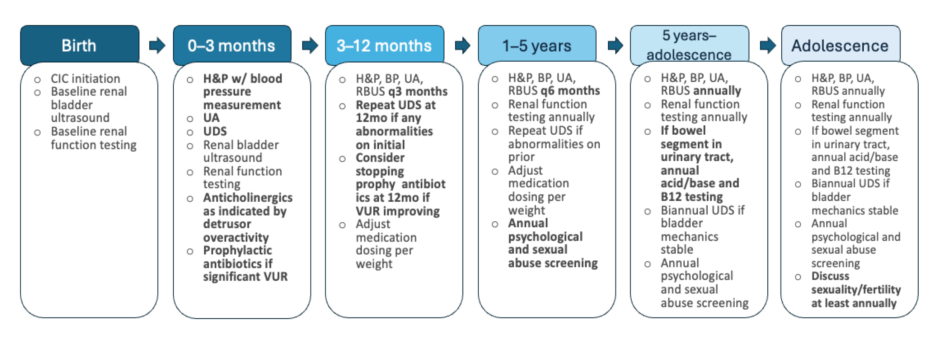
Figure 1: Outline of recommended care for patients with lower tract neurogenic dysfunction by age.
Represents a proactive management strategy with a primary goal of renal preservation. Note that UDS evaluation is performed in the first 3 months of life, ideally 2–3 months after closure of any spinal defects. For patients not undergoing spinal surgery, UDS may be performed prior to hospital discharge after birth.
Adapted from European Association of Urology (EAU)/European Society for Paediatric Urology (ESPU) guidelines on the management of neurogenic bladder in children and adolescents.11
BP: blood pressure; CIC: clean intermittent catheterisation; H&P: history and physical; mo: month; RBUS: renal and bladder ultrasound; UA: urinalysis; UDS: urodynamic studies; VUR: vesico-ureteral reflux; w/: with.
Adequate Emptying
Incomplete bladder emptying may contribute to increased bladder pressures, secondary VUR, bladder calculi, and recurrent UTI. In children ≤6 years old, post-void residual assessments >20 mL or >10% expected bladder capacity are considered abnormal.13 Residuals typically decrease in adolescence, then slowly increase in adulthood with volumes up to 100 mL being accepted as normal.14,15 Most patients with neurogenic bladder dysfunction will require some form of assistance with emptying, including vesicostomy, indwelling catheter (urethral or suprapubic), CIC via the urethra or a catheterisable channel, or creation of an incontinent urinary diversion. CIC is favoured over indwelling catheter given the lower risk of catheter-associated UTI.5,6 Impaired mobility and spasticity may challenge self-catheterisation, particularly via the urethra. Catheterisable channel creation may facilitate independent or caregiver access to the urinary tract, improve patient comfort, allow CIC despite urethral stricture or bladder neck closure, minimise caregiver interaction with the patient’s genitals, and minimise wheelchair transfers for patients with female anatomy.6
Establish Continence
Urinary incontinence, whether from the native urethra or a catheterisable channel, affects >50% of patients with neurogenic bladder secondary to spina bifida and is a significant challenge to social life, independence, and sexuality.16-18 Not surprisingly, health-related quality of life in these patients tracks with degree of primary urinary continence.19 In patients with neurogenic bowel dysfunction or chronic constipation, optimisation of bowel management may be all that is required to improve or resolve urinary incontinence. Other interventions may include changes in catheterisation schedules, anticholinergics or β-agonists, intravesical onabotulinum toxin, bulking agents, slings or bladder neck reconstructions/closures, or continent urinary diversions with bowel pouches. If intravesical onabotulinum toxin is used, the surgeon must verify dosing by other specialties (for skeletal muscle spasticity, anal sphincter relaxation, etc.) to avoid overdose.
Care must be taken to ensure efforts toward continence do not create a bladder environment hostile to the upper tracts. For this reason, surgical interventions aimed at continence are often deferred until a patient is old enough to desire social continence, has demonstrated the ability or willingness to catheterise as necessary post-procedure, and has undergone necessary interventions to create a safe lower urinary tract. Bladder neck closures are reserved as a terminal incontinence procedure for patients with stable symptoms and renal function, safe bladder dynamics, and an established catheterisable channel.
Minimise Infections
Diagnosis of UTI in the neurogenic bladder population is complex, as altered sensation may preclude traditional symptoms and bacteriuria secondary to urinary tract instrumentation is common. These barriers to accurate diagnosis can lead to over-exposure to antibiotics and, alternatively, to diagnosis of infection only once it has spread to the upper tracts.20 Pyelonephritis and its sequelae are the most common causes of hospitalisation among patients with neurogenic bladder who experience more than 10-times the number of hospitalisations of their peers.21-23 Incomplete bladder emptying, incontinence and genital skin breakdown, VUR (in the absence of high bladder pressures), and urinary calculi are all modifiable risk factors for UTI that should be addressed as part of longitudinal care for neurogenic bladder (Figure 2).
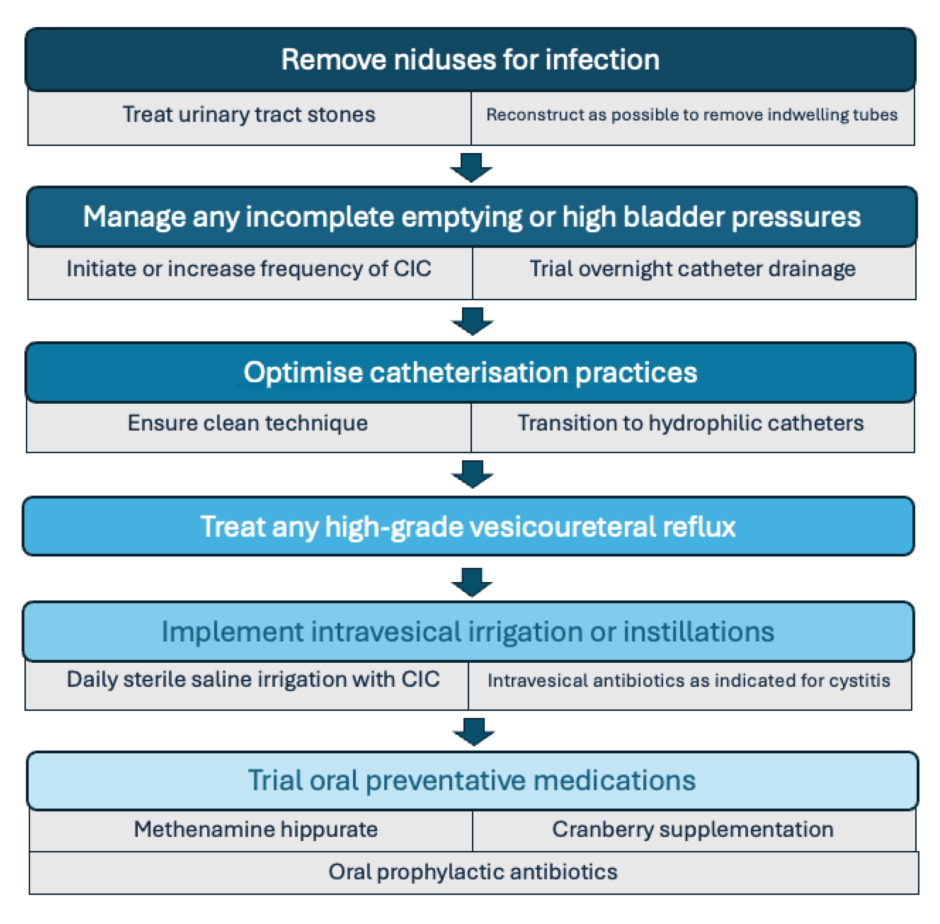
Figure 2: Hierarchy approach to addressing recurrent urinary tract infections in patients with neurogenic bladder.
Note the preference for intravesical antibiotic administration for infections limited to the lower urinary tract and the exhaustion of other options prior to considering oral prophylactic antibiotics.
CIC: clean intermittent catheterisation.
Preservation of Options for Sexuality/Fertility
Though the spectrum of sexual experience and reproductive potential in the setting of neurogenic bladder are outside the scope of this review, it is important to note that interventions targeting the bladder can impact these domains. Indwelling urethral catheters, urethral trauma or erosion from catheterisation, and procedures altering bladder neck anatomy can preclude antegrade ejaculation. Reconstructive surgeries may impair genital sensation and sexual function, and failure to achieve continence negatively impacts sexual experience.18,24 Women who have bowel in the urinary tract (e.g., bladder augment) have a 75% false-positive rate with urine pregnancy tests and must rely on serum human chorionic gonadotropin (hCG) testing or ultrasound to diagnose pregnancy.25 Asymptomatic bacteriuria and UTI are more common among these women and require close attention during pregnancy to prevent obstetric complications.26 Access to the bladder via catheterisable channel may be challenged by a gravid uterus, so some women need an indwelling catheter in the third trimester. Lastly, pregnancy amongst these patients requires coordination between obstetricians and urologists to determine safe delivery plans if pelvic anatomy is altered enough to preclude vaginal delivery or if reconstructed structures may be encountered during Caesarean section. Emergency Caesarean sections can significantly increase risks to mothers with complex reconstructions and their babies.27
Despite robust evidence that patients with neurogenic bladder are as interested in sexual activity as their peers, 9/10 paediatric urologists report their training did not include instructions on sexual and reproductive health in the context of congenitalism, and a majority of patients with spina bifida report inadequate education regarding their sexual and reproductive health from providers.28,29 Questionnaires used to screen for sexual dysfunction in the general population, such as the International Index of Erectile Function (IIEF), are not validated in and do not necessarily reflect the unique concerns of the neurogenic bladder population.30 Comprehensive and appropriately-tailored assessment and education regarding anatomy, sexuality, and fertility are indicated for these patients.29,31-33
Screening for Sexual Abuse
Children with disabilities are approximately three-times more likely to be victims of sexual violence than non-disabled peers; estimates from case series of patients with spina bifida suggest rates ≥30% by early adulthood.34-36 Risk of abuse does not normalise in adulthood and is increased in the setting of intellectual and sensory disabilities.37 Reliance on caregivers for bladder management, particularly CIC via the urethra, creates vulnerability multiple times per day and increases tolerance of physical intrusion. Nearly half (44%) of assailants against this population acted as caregivers for their victims.35 Given the prevalence of sexual violence in this population and the vulnerability bladder management creates, screening for sexual abuse and education regarding (in)appropriate touch from childhood is a critical component of comprehensive care for these patients.
Supporting Mental Health and Overall Quality of Life
Patients with neurologic dysfunction of the bladder report lower quality of life in mental health domains and may have twice the risk of depression of matched peers.38,39 Caregivers of children with these diagnoses report significant burden, particularly with CIC, and high rates of burnout and distress.40-42 The availability of mental health providers for patients and their caregivers is limited, particularly after transition to adult care settings. Medical management strategies should be adapted to minimise psychological distress, patient re-traumatisation, and burden on patients and caregivers to the extent possible without compromising upper tract preservation. Psychological care and caregiver respite should be ideally integrated into transitional programmes.
CHALLENGES AND OPPORTUNITIES IN TRANSITIONAL CARE
Historically, urologic care of patients with congenitalism was limited to paediatric urology given early patient mortality and limited familiarity with relevant pathophysiology and reconstruction techniques among adult providers. This paradigm is necessarily changing with substantially improved life expectancy and the subsequently increased prevalence of congenitalism. Though many management principles of congenital and acquired neurogenic bladder are shared, patients who have required lifelong management face different challenges than adults with newly developed dysfunction, and the transition between care in childhood and adulthood is a particular challenge for patients and providers alike. Though many investigations regarding the transition process, thus far, have focused on emotional/functional readiness to transition as an end point, ‘successful’ transition discussed here is defined as engagement in an adult urological care system without a significant (i.e., 2 years or more) gap from paediatric care.43
In settings where transition has not been structured or prioritised, transfer rates to adult healthcare can be below 10%.43 Across high resource nations with focus on transitional care, up to 40% of patients with congenitalism fail to move into adult specialty care without a significant interruption, and, as recently as 2005, 76% could not identify a primary care provider.9,23 Once engaged in adult urologic care, 52% of transitional patients were experiencing incontinence and 97% required some form of intervention, with 71% changing bladder management and 38% undergoing major surgery.16,44 Analyses of hospitalisations for patients with spina bifida have estimated that 34% (equating to 182 million USD of services per year) could have been avoided with more active management.45
Transitional Care Models
How transition happens depends heavily on the design and limitations of the local healthcare system. Figure 3 outlines a variety of models and some benefits and challenges of each. Differing locations, health record systems, and reimbursement models between paediatric and adult practices create significant challenges in the transition process. National or regional centres with lifelong disease-specific care likely offer the smoothest transition for patients and providers but are rare globally and may require significant travel for patients and families. Systems with divisions between both clinical providers and payors for paediatric and adult patients, such as the Children’s Health Insurance Program (CHIP) versus Medicaid/Medicare in the USA, are possibly the least well adapted to transitional care, because they involve multiple levels of barriers to collaboration. Health coverage/reimbursement and clinic structures that adequately reflect the complex needs of these patients compared to the general urology patient population (e.g., longer appointment times, rooms that accommodate wheelchairs, etc.) are rare and can limit engagement of adult practices.47
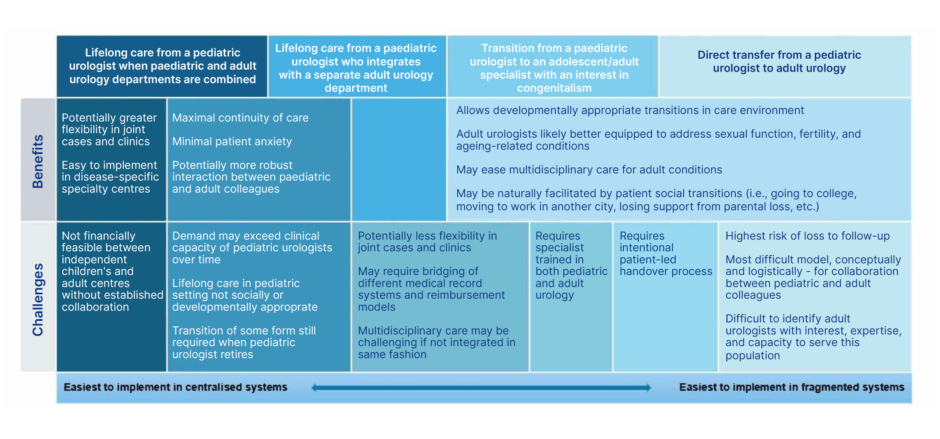
Figure 3: General models for care transition.
Adapted from Wood et al.46
Beyond urologic care, effective multidisciplinary cooperation needs to be maintained in transition. Multidisciplinary clinics are much less common in adult centres, and adult patients face intersectional challenges between their longstanding diagnoses and diseases of ageing. If necessary, repeat surgical interventions come with a substantial risk of multi-organ system complication; the risk of any complication exceeds 90% and the readmission rate exceeds 40%.48 Ultimately, regardless of the system, successful transition requires active and thoughtful interaction between paediatric and adult urologists, colleagues in different specialties, and patients and their families. Structured, dedicated transition programmes and clinics have demonstrated success rates exceeding 85% in varied health delivery systems.43
Patient Support Pathways
The transition process involves not only hand-off of care between paediatric and adult providers but is, more importantly, the process by which patients learn how to assume responsibility for their healthcare. As such, there is no universally applicable transition schedule. Effective transition is a gradual process that is often initiated by patient interest, though it may require prompting by providers. Surveys of patients with spina bifida indicate up to 40% of patients have no desire to transition and require motivational interviewing and coaching through the process, often requiring additional non-clinical visits to accomplish this goal.49,50
The Spina Bifida Association (SBA) recommends starting conversations about transition as early as birth.51 Other programmes and centres may recommend initiating conversations around 12 years of age, with a goal of transition completion around 18–20 years.9,48,52-54 On average, adolescents with spina bifida develop autonomy in activities of daily living 2–5 years after their peers and tend to overestimate their ability to complete tasks independently.55-57 Validated (Transition Readiness Assessment Questionnaire [TRAQ]) and non-validated (GotTransition, Ready Steady Go, Urology Care Foundation guide) tools have been created to guide assessment of patient knowledge and ownership in care. Improvements in these domains has been associated with increased transitional readiness.43
For some patients, the goal of transition to independence may be unachievable, despite the best efforts of all concerned. By the age of 30 years, only one-third of patients with spina bifida are independent and one-third require supervision and occasional assistance, despite being able to perform some care tasks.58 Careful evaluation and acknowledgement of each patient’s cognitive and physical abilities and functional capacity/readiness are critical components of transition planning and goal-setting for long-term success.
Patients as Expert Advisors
Though urologists study and care for individuals with neurogenic bladder, they cannot truly understand life with and management of the condition as an observer. These patients and their families quickly develop expertise not available to their care teams and beyond just lived experience.59 They can detect unease, lack of familiarity, and disinterest in providers and systems, all of which can result in a loss of trust in the therapeutic relationship and preclude transition and effective care.46 Ideally, transitional programmes should be developed and designed in collaboration with patient advisors.
CONCLUSION
The effects of neurogenic lower urinary tract dysfunction and its management extend beyond the urinary system and across the lifespan. As these patients age, the primary clinical challenges remain the same, protecting renal function, ensuring adequate bladder emptying, and achieving continence, but impetuses to prevent infection, avoid hospitalisation, and manage sexuality/fertility emerge. Screening for safety concerns and sexual abuse is necessary from early in life given the vulnerabilities created by developmental disabilities and the intimacy of bladder management. Many patients experience interruptions and avoidable escalations in care in the transition from paediatric to adult care, which is variably managed depending on health system structure and capabilities. There are tools available to clinicians, patients, and families to guide and prepare for transition, and the expertise of patients and caregivers living with neurogenic bladder should be utilised in the development of transitional programmes.