Abstract
Mature cystic teratoma is a germ cell tumour with a bimodal peak of occurrence. Common sites of involvement include the pelvis, retroperitoneum, and mediastinum. It manifests with non-specific symptoms of abdominal distension with mass effect. Owing to the large size at presentation, ultrasonography is the first-line investigation; however, it does not always yield all three components. Hence, contrast-enhanced CT is diagnostic, as it enables the identification of all the components of the teratoma. It also delineates the relations with other abdominal organs, facilitating surgical excision and histopathology. The authors present a case of primary retroperitoneal mature cystic teratoma in a 3-year-old child.
In this case, the imaging findings are sufficient to arrive at a probable histopathological diagnosis and obviate the need for pre-operative imaging-guided biopsy. The take home message is the importance of localisation of lesions to the retroperitoneal cavity and to a specific compartment with further classification based on morphological characteristics, like presence of fat, cystic tissue, and calcifications.
Key Points
1. Benign mature cystic teratomas, known as dermoid cysts, are the most common germ cell tumours and have bimodal peak of occurrence. They primarily occur in the gonads, but may arise in the anterior mediastinum, retroperitoneum, or sacrococcygeal region.2. Mature cystic teratomas contain derivatives of at least two germ cell layers and can show the characteristic presence of cystic components, fat, teeth, or cartilage not seen in unison in other tumours.
3. They can be diagnosed and prognosticated based solely on imaging findings, obviating the need for pre-surgical biopsy, which is crucial, especially in paediatric patients.
INTRODUCTION
Mature cystic teratoma is a germ cell tumour with a bimodal peak of occurrence. It is a benign lesion with very low propensity for malignant transformation and, hence, good prognosis.1 It manifests with non-specific symptoms of abdominal distension with mass effect.2 Therefore, information derived from history and clinical examination is limited. The authors present a case of primary retroperitoneal mature cystic teratoma in a 3-year-old child. In the authors’ case, the imaging findings are sufficient to arrive at a probable histopathological diagnosis. This helps avoid the need for pre-operative biopsies and subjects patients to invasive procedures just once during surgical excision. It highlights the importance of pattern recognition and the use of epidemiological information to arrive at a differential diagnosis, aiding the surgeon in decision-making for excision.
CASE PRESENTATION
A 3-year-old girl presented with vague abdominal pain and distension for 3 weeks. She was brought to the emergency department by her parents, concerned about the enlarging abdomen. She had no gastrointestinal/urinary complaints. There was no pedal oedema or other features of fluid overload. She had no antecedent medical history or past hospital admissions. She had undergone no prior investigations regarding the complaint before visiting the authors’ hospital. Abdominal examination showed a large, palpable lump in the right lumbar region, crossing the midline (Supplementary Figure 1A). The rest of the systemic examination was normal. Blood investigations were suggestive of anaemia. The child was referred for a radiograph of the abdomen, followed by a contrast-enhanced CT scan.
IMAGING FINDINGS
Abdominal radiograph showed a large, homogenous soft tissue opacity occupying the right half of the abdomen with displacement of bowel loops to the left. There was elevation of the right hemi-diaphragm with blunting of the right costophrenic angle (Supplementary Figure 1B).
The next investigation performed was an abdominal-pelvic ultrasound, which showed a large, well-defined, mixed echogenicity, retroperitoneal mass containing anechoic cystic spaces and hyperechoic fat. The calcific component seen on the CT scan of the abdomen was not well-appreciated on the abdominopelvic ultrasound, perhaps due to its medial location. There was mild peripheral vascularity within the walls of the mass on Doppler studies.
Owing to the presence of cystic and fatty components, a benign cystic lesion of the retroperitoneum was suspected. Hence, Wilm’s tumour and neuroblastoma were not considered as possible differentials.
Axial contrast-enhanced CT of the abdomen in the venous phase showed a large, well-defined, right retroperitoneal lesion containing macroscopic fat, chunky calcific foci, and cystic components. It was seen displacing the right lobe of the liver anteriorly (Figure 1A). The lesion was seen crossing the midline and displacing the right kidney medially and anteriorly (Figure 1B). It contained solid enhancing component amidst the loculated cystic component. There was right pleural effusion with atelectasis of the underlying lung. The left kidney was seen separately in its native position (Figure 1C).
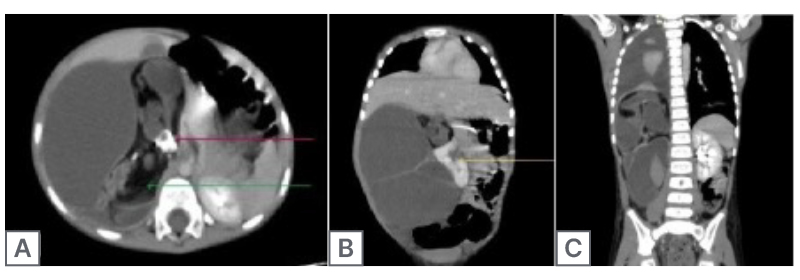
Figure 1: Contrast-enhanced CT of the abdomen.
A) Axial contrast-enhanced CT of the upper sections of the abdomen in the venous phase shows a large, well-defined right retroperitoneal lesion containing macroscopic fat (green arrow), chunky calcific foci (pink arrow), and cystic components. It is seen displacing the right lobe of the liver anteriorly. B) Coronal contrast-enhanced CT of the abdomen in the venous phase shows the lesion crossing the midline and displacing the right kidney medially and anteriorly (yellow arrow). C) Coronal contrast-enhanced CT of the abdomen in the venous phase shows the mass to have solid enhancing components. Additional findings of right pleural effusion with atelectasis of the underlying lung are seen. The left kidney is seen separately in its native position.
The displacement of the right lobe of the liver anteriorly, right kidney antero-medially, and bowel loops to the left of the abdomen suggested a right retroperitoneal tumour. The lesion was seen separately from the solid organs and colon, suggesting a primary retroperitoneal tumour. The presence of fat, cystic spaces, chunky calcific foci, and solid enhancing components in a child suggested mature cystic teratoma. Other less likely differentials included cystic neuroblastoma or pleomorphic liposarcoma.
Neuroblastoma was deemed less likely due to the presence of fat and predominant cystic components without encasement of vascular structures. Nephroblastoma was deemed less likely due to the extra-renal origin, presence of fat, and absence of vascular invasion.
In view of a well-delineated, benign-appearing lesion of the retroperitoneum without invasion of the surrounding structures, the parents were counselled regarding a good prognosis.
The patient underwent an exploratory laparotomy with complete excision of the retroperitoneal solid-cystic mass. Owing to the non-invasive nature of the lesion, the paediatric surgeons had easy retroperitoneal access and found maintained fat planes with the displaced kidney and liver, allowing easy surgical excision. Gross pathological examination showed variegated solid-cystic areas on cut section (Figure 2A). On microscopic examination, elements of ectoderm, endoderm, and mesoderm were seen. Areas of mature cartilage, adipose tissue, and glandular epithelium were identified (Figure 2B and 2C). Histopathology confirmed the radiological diagnosis. The child was discharged after a post-operative observation period of 7 days, which was uneventful (Table 1).
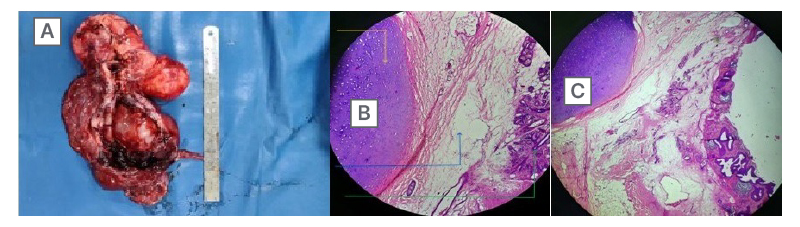
Figure 2: Gross specimen and histopathology photo.
A) Gross specimen of the resected retroperitoneal lesion. B and C) Histopathology slide showing cartilaginous component (yellow arrow), glandular tissue (green arrow), and adipose tissue (blue arrow) in the resected specimen.
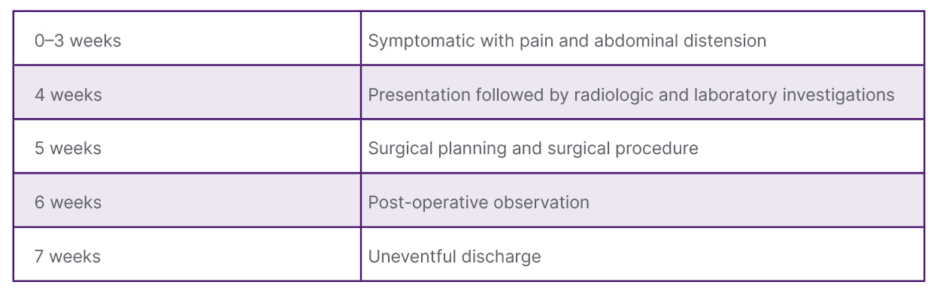
Table 1: Timeline of events from symptom onset to discharge.
Table shows the timeline of the patient from symptom onset to post-treatment discharge.
Unfortunately, the limitations of the case report were that the authors did not perform an abdominal MRI due to the need for sedation and paediatric anaesthesia owing to long scan times. The authors were also unable to maintain a long-term follow-up of the child to ensure a disease-free outcome.
DISCUSSION
The retroperitoneum is an abdominal cavity lined anteriorly by the posterior parietal peritoneum and posteriorly by the posterior body wall. It is divided into three compartments by the anterior and posterior perinephric fasciae of Gerota and Zuckerkandl, respectively. These compartments include the anterior and posterior pararenal spaces and the perinephric space. An additional vascular space is seen anterior to the vertebral column in the midline containing the aorta and infrahepatic inferior vena cava. The retroperitoneum contains bowel loops, including the ascending and descending colon and the distal duodenum, solid organs like the kidneys, suprarenals, and the pancreas, and mesenchymal tissue, including fat, muscle, vessels, and nerves.3
Retroperitoneal lesions can arise from one of these organs or from the mesenchyme. Lesions that arise from the mesenchyme are known as primary retroperitoneal and can occur in any of the compartments. Lesions can be localised to the retroperitoneum by observing anterior displacement of peritoneal organs and other retroperitoneal structures. Further, a lesion can be designated as primary retroperitoneal on the basis of the claw Sign, embedded organ sign, and the phantom organ sign.4 Occasionally, very large tumours may be difficult to localise when they occupy the majority of the abdominal cavity owing to size.
Further characterisation of the retroperitoneal lesions is done based on morphology, components like cystic spaces, calcification, fat, enhancement, blood supply, and patterns of spread. Cystic primary retroperitoneal lesions can be non-neoplastic, like urinomas, pancreatic pseudocysts, and epidermoid cysts, or neoplastic, like lymphangiomas, teratomas, cystadenomas, and cystic mesotheliomas.5 Similarly, fat-containing primary retroperitoneal lesions include common diagnoses, like lipomas and liposarcomas, or rare lesions, like myelolipomas, teratomas, neurogenic tumours, fat necrosis, and extra-medullary haematopoiesis.6 Growth pattern can be used as an identifying feature in cases of neurogenic tumours, which are elongated and extend along the sympathetic chain or lumbar nerves, and lymphangiomas and lymphomas, which tend to extend and insinuate between adjacent structures.7 Solid primary retroperitoneal lesions can be classified based on tissue of origin into lymphoid, neurogenic, mesodermal, and germ cell neoplasms.8
Retroperitoneal tumours usually present as abdominal distension with vague abdominal pain and, due to non-specific symptoms, are often diagnosed late at large sizes. Occasionally, they may present with secondary symptoms caused by mass effect on adjacent structures, like vessels or ureters.
Teratomas are the most common germ cell tumour. They can be classified into monodermal, mature, or immature types. Immature teratomas are the second most common malignant germ cell tumours after dysgerminomas and consist of immature tissue arising from one or multiple germ cell layers.1 Mature teratomas, known as dermoid cysts, are the most common types of teratomas and are benign in nature. They usually arise from the gonads; however, they may primarily originate in the mediastinum (anterior compartment), retroperitoneum (suprarenal location), sacrococcygeal region, or within the cranial cavity.9 They contain well-differentiated tissues from a minimum of two germinal layers. When arising in the retroperitoneum, they may be difficult to differentiate from neuroblastomas or nephroblastomas.
Teratomas present as abdominal lumps with or without symptoms caused by secondary mass effect. They may also cause vague abdominal pain.2 In children with such symptoms, the first-line investigation is an abdominal ultrasound, which would reveal the solid-cystic nature of the lesion with or without fat/calcifications; however, exact delineation is not always possible due to the large size of the lesion. The most characteristic finding on abdominal ultrasound is the Rokitansky nodule, which appears as a hyperechoic nodule arising from the inner aspect of the cyst. It may contain fat, hair, or calcium.10
Characterisation of the organ of origin and delineation of relations is best performed on contrast-enhanced CT. The typical appearance on contrast-enhanced CT is of a large, well-defined, heterogenous lesion of solid-cystic nature, with fat or calcification in varying proportions.11 MRI is a handy tool in the identification of constituent tissue components in less obvious cases, small foci, or rests of tissue. T1 imaging, especially, can help identify fat and keratin, while T2 imaging can help identify glandular tissue and cartilaginous components.12
Mature cystic teratomas are slow-growing, benign lesions. Rarely, however, they may undergo malignant transformation, most commonly into squamous cell carcinomas. Features suggesting malignant transformation include invasion of adjacent structures and focal irregular wall thickening.13 About 50% of malignant teratomas show elevated α-fetoprotein (AFP) levels in serum. Pathognomonic features include a chemical shift artefact and a fat–fluid level due to contained sebum.13
Preoperative diagnosis based on typical imaging findings can be made. However, surgical excision of the tumour followed by histopathology is usually necessary in atypical cases. Large retroperitoneal teratomas may cause perioperative complications, like rupture of the cyst components, vascular injury, or renal dysfunction despite being benign; however, disease-free survival is seen on follow-up.11
CONCLUSION
The first step in radiological diagnosis of a retroperitoneal lesion is precise localisation of lesions to the retroperitoneal cavity and a specific compartment of the retroperitoneum. Further classification of the lesion is based on morphological characteristics seen on imaging. The concomitant presence of fat, cystic tissue, and calcifications indicates possibility of teratoma in a young child with limited differentials. Contrast-enhanced CT is the investigation of choice and helps identify individual germ cell derivatives; however, definitive diagnosis is based on histopathology post-surgical resection if resectable, or imaging-guided biopsy.