BACKGROUND AND AIMS
Percutaneous access is essential in procedures such as percutaneous nephrolithotomy, biopsy, and ablation, requiring precise needle insertion while avoiding critical organs and parts.1,2 Success depends heavily on operator skill and image guidance, typically with ultrasound (US) or fluoroscopy.3,4 However, these methods can be difficult to use and inefficient, leading to variability in accuracy and outcomes. The authors propose a robotic system that integrates US navigation to help the surgeon perform percutaneous renal access.
METHODS
The main procedural steps of the proposed US-guided robotic system are shown in Figure 1.
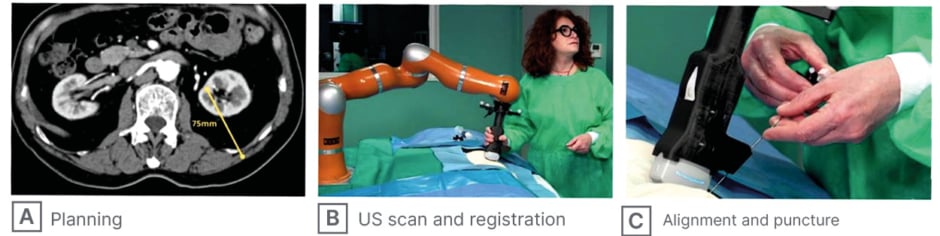
Figure 1:Key procedural steps of the proposed US-guided robotic system.
The figure illustrates the three main steps of the robotic system workflow: (A) pre-operative planning on CT scan with trajectory definition; (B) intraoperative ultrasound scan acquisition and CT-to-US registration with the robot; and (C) probe alignment and needle puncture guided by the robotic system.
US: ultrasound.
The system combines a collaborative robot with a US probe mounted in a custom holder that includes a needle guide. The surgeon first plans a safe trajectory on a pre-operative CT scan. An intraoperative US scan is then acquired, and the kidney is automatically segmented using a neural network. Because the US images are spatially tracked, a 3D point cloud of the kidney surface is reconstructed and aligned with a corresponding point cloud from CT segmentation. A registration algorithm refines the alignment, enabling precise mapping of the planned trajectory to the patient in the operating field. Once registration is complete, the system offers two workflow options: the robot automatically positions the needle guide for manual insertion along the planned path, or the surgeon continues to manually navigate the probe with real-time guidance, using 2D/3D visual feedback provided by a user interface to correctly orient the probe for accurate needle placement.
RESULTS
The authors conducted preliminary tests with both experienced and novice urologists, asking them to perform percutaneous renal access on a phantom, first using conventional freehand technique and then using the US-guided robotic system. Freehand: 75% of experts and 21% of novices succeeded on the first attempt; 25% of experts and 36% of novices required two attempts; 29% of novices required three attempts; and 7% of novices required four. With the robotic system, all participants succeeded on the first attempt. The authors’ results indicated that robotic assistance improved performance during the renal access phase of percutaneous nephrolithotomy compared with the manual approach. In the manual procedure, residents required substantially longer execution times (mean: 290 sec; median: 214 sec) than experienced surgeons (mean: 73 sec; median: 70 sec) and showed high variability. With robotic assistance, execution times between the two groups became similar and variability decreased.
Accuracy also improved: during manual procedures, residents experienced several failed insertions and experts recorded one failure, whereas no failures occurred with robotic assistance. Analysis of US probe motion revealed that residents performing the manual procedure travelled longer distances and exhibited greater orientation deviations (average distance: 2.46 m; orientation deviation: 1906°) compared with experienced surgeons (0.35 m; 199°). Robotic guidance reduced both metrics for all participants. Usability assessment through the Surgery Task Load Index (SURG-TLX) questionnaire indicated an overall reduction in perceived workload, particularly for residents, narrowing the gap between inexperienced and expert surgeons.
CONCLUSION
The proposed approach reduces the performance gap between novice and expert surgeons in percutaneous access. Unlike other solutions, it does not require intraoperative CT, eliminating radiation exposure for both patients and clinical staff while providing real-time accurate guidance.