Abstract
This case report highlights the successful use of UVA1 phototherapy in the treatment of linear morphea temporally associated with vaccination. A 13-year-old female presented at the dermatology department with localized skin hardening and tightening of the right gluteal region, which developed a few days following Bacille Calmette–Guérin vaccination. Histopathologic examination revealed sparse perivascular and peri-adnexal inflammatory infiltrates with thickened collagen bundles, findings consistent with morphea. Treatment with UVA1 phototherapy resulted in marked clinical improvement, with a reduction in skin firmness, tightness, and induration, supporting the effectiveness of UVA1 phototherapy in managing inflammatory and sclerosing cutaneous disorders.
Key Points
1. Morphea is a rare inflammatory disorder characterized by skin fibrosis, with linear morphea representing a distinct subtype that can lead to significant functional and cosmetic complications. Reports of morphea following vaccination are extremely uncommon, with only a few cases documented globally.2. This case report describes the effective use of UVA1 phototherapy in treating linear morphea potentially triggered by vaccination. A 13-year-old girl presented to the dermatology clinic with localized skin induration and tightening over the right gluteal region, which developed a few days after receiving the Bacille Calmette–Guérin vaccine.
3. Because there is no gold standard in the treatment of Morphea, management must be highly patient centered and individualized. Treatment selection is guided by several factors such as severity of the disease, its activity and extent, anatomic distribution, depth of distribution, and rate of progression, although non-life-threatening, functional, and cosmetic issues must be addressed. Vaccine-related morphea is rare yet treatable and may be self-limited.
INTRODUCTION
Morphea, also called localized scleroderma, is a rare fibrosing disorder affecting the skin and underlying tissues. Its incidence is estimated at 0.4–2.7 per 100,000 people. Morphea is more frequently seen in White individuals and women, with a female-to-male ratio of about 2:1 to 4:1. It is distinguished from systemic sclerosis by the absence of sclerodactyly, Raynaud phenomenon, and nailfold capillary changes.
PATIENT INFORMATION
The authors report the case of a 13-year-old female who presented with localized skin hardening and tightening over the right gluteal region, which developed a few days following Bacille Calmette–Guérin (BCG) vaccination. Subsequently, similar lesions appeared on the left anterior and lateral thigh, characterized clinically as firm and indurated plaques. Past medical history was unremarkable. Family history revealed no similar lesions within the family.
Approximately 4 years after the initial onset, the patient’s mother noted the development of an abnormal gait, prompting dermatologic consultation. Baseline laboratory investigations, including complete blood count, liver function tests, and renal function tests, were within normal limits. A skin biopsy performed at that time demonstrated histopathologic findings, which included compact hyperkeratosis and flattened epidermis, and, within the dermis, there was a superficial and deep perivascular and periadnexal infiltrate of lymphocytes and histiocytes. The deep reticular dermis was characterized by thick compact collagen bundles; all were consistent with early morphea. Treatment with topical calcipotriol was initiated but resulted in no clinical improvement, after which the patient was lost to follow-up.
During the intervening period, the lesions persisted and progressively increased in size. Upon re-evaluation, the patient underwent narrowband UVB phototherapy three times weekly for nine sessions, without a significant clinical response. Consequently, UVA1 phototherapy was recommended as the subsequent therapeutic intervention, hence referral to the dermatology department.
CLINICAL FINDINGS
Cutaneous examination revealed multiple ill-defined, irregularly shaped plaques ranging from skin-colored to hyperpigmented, with areas of induration and focal atrophy. The lesions were located on the left anterior thigh (measuring 6×10 cm), left lateral thigh (27×11 cm), and right gluteal region (5×11 cm). There was noted limb length discrepancy. Notably, there was effacement of the intergluteal cleft, attributed to cutaneous fibrosis resulting in skin tightening (Figure 1).
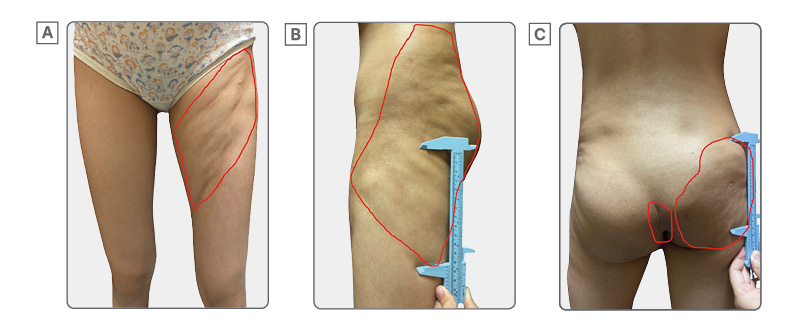
Figure 1: Multiple atrophic, indurated plaques on thighs and buttocks.
DIAGNOSTIC ASSESSMENT
Dermoscopy demonstrated fibrotic beams along with loss of hair follicles within the affected areas (Figure 2). A repeat skin biopsy demonstrated superficial and deep perivascular and periadnexal inflammatory infiltrates, with thick, compact collagen bundles in the deep reticular dermis. These findings were consisten with morphea (Figure 3).
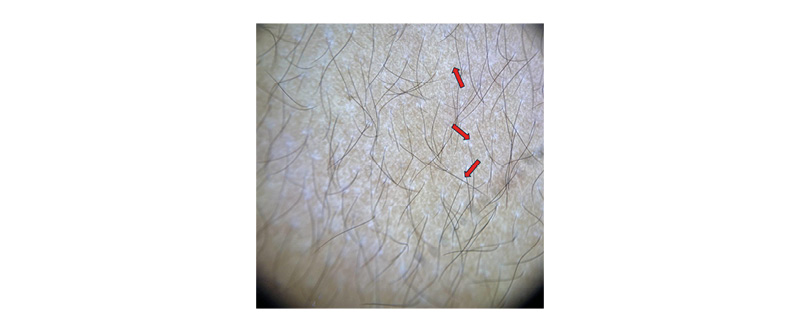
Figure 2: Dermoscopy: fibrotic beams (red arrows) and loss of hair.
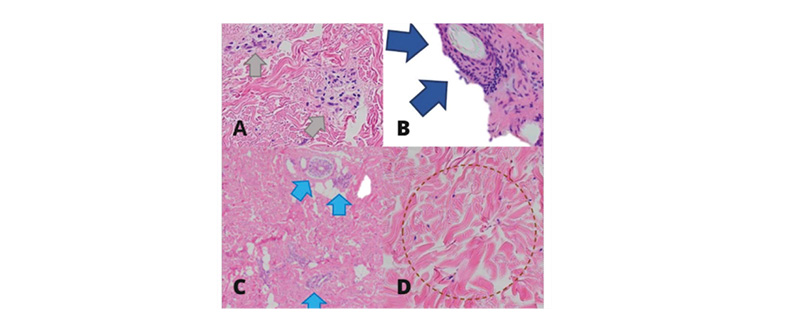
Figure 3: A and B) Sparse perivascular periadnexal infiltrates; C) entrapped eccrine glands; D) thick collagen bundles.
THERAPEUTIC INTERVENTION
UVA1 phototherapy was started at 2,000 mJ/cm2, with increments of 1,000 mJ/cm2 twice a week for a total of 82 sessions. Future plans would include continuing UVA1 phototherapy at 40,000 mJ/cm2, one to two times a week. As the patient’s treatment duration has progressively increased on a weekly basis, she has developed difficulty in tolerating standing for periods exceeding 1.5 hours. Notably, the patient is 13 years old. In light of this, the treatment plan is to transition from UVA1 phototherapy to narrowband UVB phototherapy, which is also an effective modality for the management of morphea. The patient was also referred to rehabilitation medicine to evaluate limb length discrepancy and abnormal gait. The complete timeline is depicted in Supplementary Table 1.
LIMITATIONS OF THIS CASE
This report has several limitations. First, as a single case report, the findings are inherently limited in generalizability, and a causal relationship between vaccination and the development of morphea cannot be established. The observed association is temporal and may be coincidental.
Second, although the onset of lesions occurred shortly after BCG vaccination, there was a prolonged interval before definitive diagnosis and treatment, which may introduce recall bias and limits the precision in establishing the exact timeline of disease progression.
Third, comprehensive immunologic workup was not performed, which limits further characterization of underlying autoimmune predisposition or systemic involvement.
Fourth, the lack of standardized objective outcome measures (e.g., validated skin scoring systems or imaging-based assessment) restricts the ability to quantitatively evaluate treatment response to UVA1 phototherapy.
Finally, while the patient demonstrated significant clinical improvement, long-term follow-up data are limited, and the durability of response, as well as risk of relapse, remain uncertain.
DISCUSSION
Morphea is a chronic inflammatory disease that affects the dermis and subcutaneous tissue. It is also known as localized scleroderma. Progressive skin sclerosis, thickness, induration, and variable degrees of atrophy are its defining characteristics. Morphea is rarely fatal, though it can lead to functional and aesthetic problems. In rare instances, it is self-limited, but more frequently it follows a chronic, relapsing–remitting pattern, which over time leads to a considerable cumulative disease burden.
Given the clinical presentation, several conditions were considered in the differential diagnosis, including systemic sclerosis, atrophoderma of Pasini and Pierini, stiff skin syndrome, and eosinophilic fasciitis. Systemic sclerosis was considered due to the presence of cutaneous sclerosis; however, Raynaud phenomenon and other signs and symptoms were not present, hence this diagnosis is less likely. Atrophoderma of Pasini and Pierini was also entertained given the presence of dermal atrophy, but the lack of the typical cliff-drop borders argued against this entity. Stiff skin syndrome was considered in view of skin induration; however, the clinical course and distribution were not consistent with its usual presentation. Eosinophilic fasciitis was likewise included in the differential due to skin thickening, but the absence of peripheral eosinophilia, fascial involvement, and groove sign made this diagnosis unlikely. Taken together, these findings support the final diagnosis of morphea.
The pathogenesis of morphea remains incompletely understood; however, accumulating evidence points to multiple immunoinflammatory and profibrotic pathways. In linear morphea there is immune activation, vascular injury, and progressive fibrosis. In the early phase, Th1/Th17-mediated inflammation leads to the release of cytokines such as IL-6 and TNF-α, causing endothelial damage and immune cell infiltration into the skin and deeper tissues.1 The disease then shifts toward a Th2-dominant response, where IL-4 and IL-13 promote fibroblast activation and collagen deposition, with TGF-β acting as a key profibrotic mediator.1 In linear morphea, these processes extend beyond the dermis to involve subcutaneous tissue, muscle, and bone, leading to structural deformity and functional impairment.1
The development of morphea is thought to result from an interplay between predisposing host factors and external triggers.2 Predisposing factors include a personal or family history of autoimmune disease, concurrent autoimmune conditions, cutaneous mosaicism, and specific human leukocyte antigen (HLA) subtypes, particularly HLA-DRB104:04 and HLA-B37, which have been associated with increased susceptibility to generalized and linear forms of morphea.2,3 These genetic associations were also present in the authors’ patient.
Epigenetic mechanisms are thought to serve as a link between genetic susceptibility and environmental factors.4 External triggers such as injuries, injections, or vaccinations can initiate disease in individuals who are genetically predisposed, as seen in this case. After such triggers, a complex process involving epidermal keratinocytes and dermal fibroblasts occurs, engaging innate and adaptive immune responses along with profibrotic signaling pathways. This process ultimately results in excessive collagen deposition and fibrosis.4 In the present case, the onset of cutaneous induration occurred within a few days following BCG vaccination, suggesting a close temporal association. Similar temporal relationships have been described in previously reported cases of vaccine-associated morphea, where lesions developed days to weeks after immunization, often at or near the injection site. This pattern supports the hypothesis that vaccination may act as a triggering event in predisposed individuals rather than a direct causative factor.
From a pathophysiological standpoint, vaccination may induce localized immune activation through multiple mechanisms. Mechanical trauma from injection may initiate an exaggerated wound-healing response, while vaccine components or adjuvants may stimulate innate immune pathways, including Toll-like receptor signaling, leading to the release of pro-inflammatory and profibrotic cytokines such as TGF-β. This cascade promotes fibroblast activation, excessive collagen deposition, and, ultimately, cutaneous sclerosis.
Despite these proposed mechanisms, current evidence remains largely limited to case reports and small case series, and a definitive causal relationship between vaccination and morphea has not been established. Therefore, the observed association should be interpreted with caution.
Eight cases of morphea have been associated with different vaccines, including measles–mumps–rubella, diphtheria–tetanus–pertussis, BCG, hepatitis B, and tetanus.5 Given the variety of vaccines involved, the authors propose that injection-related trauma or vaccine preservatives may serve as the actual triggers, rather than specific vaccine antigens.6 It is also suggested that mechanical trauma from vaccination could provoke an exaggerated wound-healing response, resulting in abnormal fibrosis.
Recently, several cases of morphea were reported following COVID-19 vaccination. One case involved an 80-year-old man who developed morphea profunda after vaccination and was successfully treated with oral methotrexate.6 Although a temporal association does not establish causality, these reports highlight the potential for vaccine-related immune activation to serve as a trigger in individuals who are genetically or immunologically predisposed. Current evidence supporting vaccine-associated autoimmunity is largely derived from case reports and observational data, and a definitive causal relationship has not been established. The management of morphea remains challenging, since there is no single therapy that is regarded as the gold standard.7 Treatment selection is directed by disease activity, extent, anatomical distribution, depth of involvement, and rate of progression.8 Consequently, a patient-centered therapeutic approach is essential.9
In pediatric patients with linear morphea, management is guided by disease severity and depth of involvement, with the primary goal of controlling inflammation early to prevent permanent fibrosis and deformity. For mild, superficial lesions, especially in cosmetically sensitive areas like the face, topical tacrolimus may be used as a first-line or adjunctive therapy.9,10 It works by inhibiting T cell activation and reducing inflammatory cytokines, helping to decrease erythema and soften lesions; however, its limited tissue penetration makes it insufficient for deeper or progressive disease.1,11 In cases of moderate-to-severe, rapidly progressive, or function-threatening linear morphea, systemic therapy is required, with methotrexate serving as the cornerstone of treatment.9 Methotrexate suppresses immune activity and fibrosis and has the strongest evidence for reducing disease activity and preventing progression in children; it is typically administered weekly and continued for at least 12 months or longer.9 Because methotrexate has a delayed onset of action, it is commonly combined initially with systemic corticosteroids such as prednisone or intravenous methylprednisolone to achieve rapid control of active inflammation; these steroids are used as short-term induction therapy and then tapered, as they are not suitable for long-term disease control.9 For patients who do not respond adequately to or cannot tolerate methotrexate, mycophenolate mofetil is an effective second-line option, offering immunosuppressive effects through inhibition of lymphocyte proliferation and serving as a useful steroid-sparing agent.9,11 Overall, treatment often involves a combination approach (early systemic therapy for active disease, with adjunctive topical agents for superficial lesions) to optimize outcomes and minimize long-term complications.
UVA1 phototherapy uses long-wavelength UV radiation (340–400 nm), which penetrates more deeply into the dermis than UVB or broadband UVA.8 Its therapeutic effect in morphea is attributed to immunomodulatory actions, including attenuation of inflammatory responses, suppression of fibroblast activity and collagen synthesis, and upregulation of matrix metalloproteinases (e.g., matrix metalloproteinase-1) involved in collagen degradation.8 These effects are thought to be mediated by activation of the aryl hydrocarbon receptor pathway, resulting in antifibrotic activity at the cellular level.8
Several studies have demonstrated that UVA1 phototherapy significantly improves cutaneous sclerosis in morphea, leading to plaque softening and enhanced skin elasticity.1,3,10 Both medium and high-dose regimens have shown efficacy, with documented improvements in clinical severity scores and objective assessments of skin elasticity.12 In a prospective study, approximately 82% of patients reported symptomatic improvement, accompanied by ultrasonographic evidence of reduced skin thickness following UVA1 treatment.6
In the short term, adverse effects of UVA are generally mild and include erythema (a sunburn-like reaction), skin dryness and pruritus due to epidermal barrier disruption, and hyperpigmentation from increased melanogenesis; patients may also experience heat-related discomfort during treatment because UVA1 devices generate significant warmth, and, rarely, phototoxic reactions can occur if combined with photosensitizing agents.1 Long-term risks are more concerning, especially in pediatric patients, and include photoaging from cumulative UV exposure leading to collagen breakdown, resulting in skin laxity, wrinkling, and dyspigmentation.1 There is also a theoretical risk of carcinogenesis due to deep dermal penetration of UVA1 and oxidative DNA damage, with potential associations with melanoma, basal cell carcinoma, and squamous cell carcinoma, although pediatric data remain limited and much of the risk is extrapolated from adult and psoralen plus UVA studies.1,13
Cutaneous involvement in morphea can result in restricted range of motion, limb-length discrepancy, joint deformities, and contractures, particularly in cases of linear morphea, where musculoskeletal complications have been reported in approximately 45–56% of patients.8 Lesions that cross joint lines confer the highest risk for functional impairment.8 In pediatric patients, morphea may be associated with substantial morbidity, including adverse effects on growth, physical function, and quality of life, whereas fewer studies have evaluated these outcomes in adults.8
The relationship between morphea subtype and quality of life using the Dermatology Life Quality Index (DLQI) and Children’s Dermatology Life Quality Index (CDLQI) were evaluated. Among 202 adult patients, 75 individuals experienced a moderate or greater impact on quality of life (DLQI >5), with the greatest impairment observed in those with generalized morphea; however, no statistically significant correlation was identified between morphea subtype and DLQI scores (F=1.97; p=0.12). In contrast, among 75 pediatric patients, 21 (28%) reported a moderate or greater impact on quality of life, nearly all of whom had linear morphea.11 Similarly, no significant association was found between morphea subtype and CDLQI scores (F=0.45; p=0.72).11 Overall, children demonstrated a lower frequency of moderate-to-severe quality-of-life impairment compared with adults.13
Although morphea is rarely life-threatening, it can lead to significant functional and cosmetic morbidity.7 While the disease may be self-limited in some cases, it more commonly follows a chronic or relapsing–remitting course, resulting in a substantial cumulative disease burden over time.8
Morphea temporally associated with vaccination is a rare occurrence and, despite its chronic and potentially relapsing course, remains a treatable condition.14 Notably, spontaneous remission has been reported in a subset of patients.6 These observations underscore the importance of avoiding undue alarm and emphasize the need for individualized clinical assessment and management, as patient responses to disease processes, pharmacologic therapies, and vaccinations may vary.14 Importantly, the well-established benefits of vaccination substantially outweigh the potential risks.14 Consequently, rare adverse cutaneous events such as morphea should not deter vaccination, particularly in the context of ongoing public health challenges.
CONCLUSION
Although morphea can result in functional and cosmetic impairment, it is rarely life-threatening. The disease may be self-limited in some individuals; however, it more commonly follows a chronic or relapsing–remitting course, leading to a substantial cumulative disease burden over time. Given the wide range of vaccines reported in association with morphea, it has been suggested that injection-related trauma or vaccine preservatives, rather than specific vaccine antigens, may act as potential triggers. Mechanical trauma from vaccination may initiate an exaggerated wound-healing response, resulting in dysregulated fibrosis. UVA1 phototherapy is an effective therapeutic modality for the management of inflammatory and sclerosing cutaneous disorders, including morphea.
PATIENT PERSPECTIVE
Before starting the treatment, my skin felt very tight and hard, which made it difficult for me to move comfortably and do activities of daily living. I cannot play sports with my friends due to the limitation of movement. When I started phototherapy, I did not know what to expect, but the treatments were generally tolerated. One downside was I had to stand up for a long period of time since the duration of treatment became progressive over time. During my phototherapy sessions, I watched short films to keep myself entertained, which helped pass the time without me noticing it. After completing the treatment, I noticed that my skin became softer and less tight. I was able to move more easily, and my daily activities were less difficult. I felt relieved and more confident with the improvement.