Meeting Summary
The symposium featured presentations from Tommaso Simoncini, Professor of Obstetrics and Gynaecology, Department of Clinical and Experimental Medicine, University of Pisa, Italy; Zoe Schaedel, a UK General Practitioner and British Menopause Society (BMS) Menopause Specialist, Brighton and Hove NHS Menopause Clinic, UK; Petra Stute, Director of the Menopause Center, Department of Obstetrics & Gynaecology, Inselspital Bern, Switzerland; and Rosalia Silvestri, Associate Professor of Neurology, University of Messina School of Medicine, Italy.
The faculty explored insomnia disorder as a prevalent, often overlooked disease during the menopausal transition. Its impact on quality of life and long-term health outcomes is profound, making it essential for clinicians to understand the burden and explore effective solutions.
Simoncini and Schaedel opened the session by describing the differences between insomnia disorder and ‘poor sleep’, highlighting how insomnia disorder can lead to long-term adverse consequences, including increased occurrence of health conditions such as cardiovascular disease and diabetes, and psychiatric disorders and neurological diseases, including dementia and Alzheimer’s disease. Stute addressed the occurrence of insomnia disorder specifically in menopausal women, highlighting this as a standalone disease occurring in 26–57% of women transitioning through menopause, with severe cognitive and cardiometabolic impacts on patients. Stute also underlined the challenges around managing the disease, including the fact that most menopausal guidelines do not distinguish between insomnia disorder and other sleep disturbances, highlighting a consequent management gap. Silvestri focused on treatment challenges to bridge this gap, including the need to initiate pharmacological treatment of insomnia disorder if first-line cognitive behavioural therapy is not effective, and addressing common comorbidities to be considered when making treatment decisions, such as obstructive sleep apnoea (OSA), restless leg syndrome, and nocturia. Finally, Schaedel presented data on targeted pharmacotherapeutic solutions for insomnia disorder during the menopausal transition, focusing on orexin receptor antagonists, such as daridorexant, to directly address the state of hyperarousal underlying insomnia disorder.
Introduction
Approximately 26–57% of women undergoing menopausal transition experience insomnia disorder,1-3 a chronic disease that negatively impacts women’s wellbeing and is associated with cognitive deficits and an increased risk of psychiatric, neurological, and cardiometabolic disorders.4,5 Although menopause hormone therapy (MHT) can address common vasomotor symptoms (e.g., hot flushes and night sweats) associated with the oestrogen decline in perimenopause/menopause that may disrupt sleep,6,7 not all women with menopausal insomnia report vasomotor symptoms,8 treating vasomotor symptoms does not always improve sleep, and, importantly, MHT is not indicated for the treatment of insomnia disorder. This article discusses the importance of viewing insomnia disorder during the menopause as a standalone disease rather than simply a menopausal symptom, and the need for effective treatments for insomnia to reduce both the short- and long-term health impacts in women.
Setting the Stage: Understanding Insomnia Disorder During Menopausal Transition
The Burden of Insomnia Disorder
The symposium started with a short movie telling the story of a mid-40s, Berlin-based researcher and mother of three who lives with insomnia disorder and shares the impact it has on her physical and mental health, daytime functioning, work, and relationships with family. Following this moving story, Simoncini and Schaedel highlighted that insomnia disorder, a chronic, frequent inability to maintain sufficient sleep and impairment of daytime functioning, clearly delineated from ‘poor sleep’, is associated with several serious health conditions, including depression,9 cardiovascular diseases such as hypertension,10 and metabolic disorders, including Type 2 diabetes.11 Around 6–10% of adults in the overall population meet the criteria for insomnia disorder. The disorder is characterised by a difficulty falling asleep and/or staying asleep occurring three or more times per week that has been ongoing for at least 3 months and has negatively impacted an individual’s daytime functioning.
Insomnia Disorder During Menopausal Transition
The speakers highlighted that insomnia disorder is particularly prevalent during menopausal transition, where 40–90% of women experience insomnia symptoms,1,5 and up to 57% meet criteria for clinical insomnia disorder.1,2 Yet, as they noted, “the distinction between sleep disturbances […] and insomnia disorder is not always clear in women’s health literature.”
This distinction matters: midlife women often require targeted treatments to address the drivers of disrupted sleep during menopause and to mitigate the substantial physical, occupational, and quality-of-life burdens associated with insomnia.
The Hidden Burden: Unmasking the Impact of Insomnia Disorder During Menopausal Transition
Insomnia Impacts Quality of Life During Menopause
Stute presented data from an observational cross-sectional study12 of 480 women aged 45–65 years presenting to a specialised menopause unit. Nearly 40% of the cohort reported that insomnia had a negative impact on their quality of life. Using the 16-item Cervantes-Short Form (C-SF) scale, which assesses quality of life across menopause-related health, mental health, sexuality, and relationship domains, insomnia emerged as a stronger detractor of quality of life in the menopause domain than night sweats, hot flashes, or fatigue.
On a 0–5 scale (0–1: low impact; 2–3: moderate; 4–5: high), insomnia scored 2.76 (SD: 2.1), compared with 2.53 (SD: 1.6) for night sweats, 2.48 (SD: 1.6) for hot flashes, 2.27 (SD: 1.7) for fatigue, and 2.83 (SD: 1.7) for dry skin. These findings underscore insomnia as a significant and often underappreciated contributor to reduced quality of life among midlife women.
Sleep Maintenance is the Most Common Sleep Difficulty During Menopause
In the 7-year longitudinal SWAN study3 of 3,045 White, African American, Chinese, Japanese, and Hispanic women aged 42–52 years not receiving MHT, 26% of premenopausal or perimenopausal women reported sleep maintenance difficulties, with symptoms experienced at least three times per week. This prevalence increased to over 40% among women in the late menopausal transition.
Across all stages assessed, premenopause, early menopausal transition, late menopausal transition, and postmenopause, difficulty maintaining sleep was the most commonly reported sleep problem, surpassing both sleep onset difficulties and early morning awakenings. Transition stages were determined using standard bleeding pattern criteria.
Stute further emphasised the broad health consequences of insomnia disorder. Daytime fatigue was identified as the most frequent symptom,13 alongside notable impairments in daytime functioning. These included cognitive difficulties, such as memory issues and reduced motor control; emotional impacts, including irritability and distress; and behavioural consequences, such as increased food intake contributing to weight gain.
Many Factors are Associated with Sleep Disturbances During Menopause
Hormonal changes, vasomotor symptoms, and stressful life challenges, including psychosocial and socioeconomic factors,5 are three key factors associated with sleep disturbances during menopausal transition, explained Stute. Declining oestrogen levels disrupt vasomotor control and normal thermoregulation, causing hot flashes and night sweats, which can significantly impair both the quantity and quality of sleep.6,7
However, not all menopausal sleep problems can be explained by vasomotor symptoms alone.8 In a cross-sectional study involving 34 women (mean age: 50.4 years; SD 2.7) and a total of 63 nights of polysomnographic monitoring, researchers found that only 27% of awakenings were associated with hot flashes.14 Decreased levels of oestrogen also have other physiological effects that impact sleep, including decreased melatonin production,15 increased nocturnal levels of cortisol,16 reduced serotonin activity,6 and increased orexin signalling.17,18
Stute emphasised that clinicians should therefore consider factors beyond vasomotor symptoms when evaluating sleep disturbances during the menopausal transition.
Important to Distinguish Between General Sleep Disturbances and Insomnia Disorder
Many menopausal guidelines do not clearly distinguish between general sleep disturbances and insomnia disorder, creating a gap in targeted management for women experiencing persistent insomnia during menopausal transition.19,20 According to the British Menopause Society (BMS) Guidelines,21 menopause symptoms causing sleep disruptions should first be addressed by managing contributing symptoms. Recommended options include MHT, with evidence that micronised progesterone may offer superior sleep benefits compared with other progestogens, as well as non-hormonal therapies, such as selective serotonin reuptake inhibitors or other agents that reduce vasomotor symptoms, and menopause-specific cognitive behavioural therapy (CBT).
If sleep disturbances persist despite menopause-focused treatment, clinicians are advised to check specifically for insomnia disorder, characterised by difficulty initiating or maintaining sleep, early awakening, and daytime impairment. For confirmed insomnia, the BMS recommends evidence-based therapies such as CBT for insomnia (CBTI), and, when medication is appropriate, options include melatonin for short-term use, antidepressants in selected cases, or daridorexant, a dual orexin receptor antagonist (DORA) licensed for the treatment of insomnia disorder.
However, in view of the bidirectional relationship between menopause symptoms and insomnia disorder, and the increased risk of comorbid conditions such as depression and OSA, Stute emphasised that clinicians should routinely screen for insomnia disorder during menopausal transition. Recognising and treating insomnia disorder as a serious, standalone condition may help reduce the risk of future health consequences in midlife women.
Bridging the Gap: Navigating Treatment Challenges and Patient Needs During Menopausal Transition
Sleep Disturbances During Menopause and the Development of Insomnia Disorder
Silvestri highlighted that sleep disruption frequently emerges or worsens during menopausal transition, even among women who previously slept well. One longitudinal study5 of 60 premenopausal women, assessed at baseline and 6 years later, demonstrated a clear decline in sleep quantity and maintenance: total sleep time decreased and wakefulness after sleep onset increased, even after adjustment for vasomotor symptoms, BMI, and mood. These findings underline that hormonal changes accompanying menopausal transition can exert long-lasting effects on sleep architecture, regardless of whether women initially identify sleep as a problem.
The evolution from occasional disturbed sleep to clinically significant insomnia disorder can be explained by the established 3P model, which Silvestri described in the menopausal context.22 Predisposing factors, including neuroticism, depression, and obsessive personality traits, plus circadian irregularities related to age and hormonal fluctuations, lead to a premorbid likelihood of insomnia. Precipitating factors such as age-related poor health, stress, pain, and vasomotor symptoms can provoke acute insomnia. If these disturbances persist, perpetuating hyperarousal, negative mood, maladaptive sleep habits, and continued vasomotor symptoms can perpetuate sleep disruption and shift women into insomnia disorder. These trajectories reinforce that menopause-related sleep disturbances arise from multiple interacting factors, with vasomotor symptoms explaining only a proportion of the overall burden.
Insomnia Disorder is an Independent Disorder During Menopausal Transition
Building on these observations, Silvestri emphasised the importance of recognising insomnia disorder as an independent clinical condition during the menopause and not merely a symptom of menopause. Although the treatment of vasomotor symptoms can improve sleep to some extent, these effects are only partial, and MHT provides, at best, only a modest benefit for insomnia. Importantly, MHT is not indicated for insomnia disorder.23,24 In contrast, CBTI is effective even if vasomotor symptoms persist,23 and is the first-line treatment for adults with insomnia of all ages, regardless of comorbidities.
When is Pharmacological Treatment Recommended for Insomnia Disorder?
Silvestri also outlined how treatment decisions may differ depending on the phenotype of insomnia.
The first phenotype,25 characterised by normal sleep duration and the absence of physiological hyperarousal, is generally associated with an anxious ruminative profile, normal stress system activity, and no increased cardiometabolic risk. In these cases, CBTI remains the cornerstone of management, and pharmacological therapy is frequently unnecessary. The second phenotype involves physiological hyperarousal accompanied by short sleep duration. This form is associated with a biological vulnerability to insomnia, impaired neurocognitive functioning, and a heightened risk of cardiovascular morbidity and mortality. In such patients, CBTI alone is often insufficient,25 and pharmacological therapy is typically required as part of an individualised treatment plan (Figure 1).26

Figure 1: The European insomnia guidelines.26
Clinical treatment algorithm for insomnia disorder.
Adapted from Riemann et al.26
BZ: benzodiazepine; BZRA: benzodiazepine receptor agonist; CBTI: cognitive behavioural therapy for insomnia; DORA: dual orexin receptor antagonist; FR: fast-release.
It is therefore important to recognise insomnia disorder as an independent disease and to actively treat insomnia disorder whenever it presents.26 Recognising insomnia disorder as a discrete condition means treating it whenever it occurs. While CBTI is the recommended first-line therapy, it may not always be available or adequate. In these situations, pharmacotherapy becomes necessary.
Treatments recommended in the 2023 European Insomnia Guideline26 include DORAs such as daridorexant, which act by blocking orexin-1 and orexin-2 receptors to reduce hyperarousal. DORAs are currently the only pharmacological class granted the highest-grade (A) recommendation for short, medium, and, in certain cases, long-term treatment. Benzodiazepines and benzodiazepine receptor agonists are recommended only for short-term use of 4 weeks or less because of their potential for tolerance and dependence.27 Melatonin receptor agonists can be used in patients older than 55 years, with evidence for benefit including a meta-analysis of 24 studies in 1,173 postmenopausal women showing improvements in subjective sleep quality.28 The 2023 guideline also acknowledges that sedating antidepressants may be used off-label for brief periods of no more than 4 weeks, though clinicians must carefully consider contraindications and should remember that this class is not approved for the treatment of insomnia disorder.
Comorbidities Require Careful Treatment Considerations
Silvestri also noted that insomnia disorder during menopause is frequently accompanied by other medical conditions and may worsen or contribute to their development. The relationship between insomnia and these comorbidities is often bidirectional, affecting psychiatric disorders such as depression and alcohol misuse, as well as cardiometabolic conditions.29-31 Restless legs syndrome is approximately twice as common in women over 40 years as in men,32 and is frequently observed alongside insomnia during menopausal transition. Sleep-disordered breathing also increases in prevalence: in a cohort of 589 premenopausal and menopausal women, the odds of five or more apnoea or hypopnoea events per hour rose to 2.6 in postmenopause, and the odds of 15 or more events per hour rose to 3.5.33 Nocturia is another common condition in this demographic, affecting 34% of women over 40 years in a broader cross-sectional analysis.34
OSA is a particularly important comorbidity given its frequent co-occurrence with insomnia disorder. Approximately 30–50% of people with OSA experience comorbid insomnia symptoms,35 and 30–35% of individuals with insomnia disorder are estimated to have underlying OSA. This co-presentation, referred to as Co-morbid Insomnia and Sleep Apnoea (COMISA), is characterised by fragmented sleep, non-restorative rest, daytime impairment, and worsening quality of life. Managing COMISA typically involves a dual approach using CBTI alongside continuous positive airway pressure,36 although adherence to continuous positive airway pressure is often poor. Although hormonal changes may influence sleep-disordered breathing, there is no clear evidence supporting MHT as a treatment for OSA. The use of sedative sleep medications such as benzodiazepines requires caution, as they may depress respiratory function37 or contribute to daytime drowsiness.38 In contrast, DORAs, including daridorexant and lemborexant, as well as slow-release melatonin, have demonstrated good tolerability and benefit in patients with COMISA,39-41 offering alternative strategies when CBTI is inadequate.
Targeted Solutions: Daridorexant Insights in Insomnia Disorder During Menopausal Transition
Orexin and Hyperarousal: A Mechanistic Target in Menopausal Insomnia
Schaedel began by underscoring that understanding insomnia requires an appreciation of the central role of orexin in regulating wakefulness. Orexin, a relatively recently discovered neuropeptide produced in the lateral hypothalamus, functions as a master switch controlling the transition between wake and sleep.42 When orexin levels are high, they stimulate multiple wake-active neurons and maintain alertness, an effect entirely appropriate during the day. At night, the wake-promoting system ordinarily quietens, allowing sleep-active neurons to predominate in a flipflop mechanism that enables stable sleep. In insomnia disorder, however, this balance is disrupted, not because the sleep system is weak, but because the wake system remains overactive, driven by elevated orexin signalling and resulting in the persistent hyperarousal that typifies the condition. During the menopausal transition, declining oestrogen may further heighten orexin activity, contributing both to insomnia and to symptoms such as vasomotor instability.43 Daridorexant, a DORA, directly targets this overactive wake system by blocking the two orexin receptors, thereby reducing hyperarousal and facilitating sleep.44 This mechanism has been validated over 2 decades of research in animal and human studies, demonstrating that inhibiting orexin signalling induces sleep in a physiologically coherent way.
Efficacy and Safety of the DORA Daridorexant in Insomnia Disorder
Schaedel presented the results of the large Phase III programme evaluating daridorexant, published in 2022.45 More than 1,800 adults with insomnia disorder across 18 countries were enrolled and randomised to receive either daridorexant at different doses or placebo for 3 months. Objective sleep outcomes were assessed using overnight polysomnography in sleep laboratories, capturing EEG-derived sleep architecture, eye movements, muscle tone, ECG, and oxygen saturation to characterise sleep stages and continuity. Alongside these objective measures, participants recorded twice daily electronic sleep diaries, providing subjective assessments of their sleep experience and the daytime consequences of insomnia.
A clear dose–response relationship was observed for efficacy, with 50 mg demonstrating greater benefit than 25 mg while maintaining a similar tolerability profile, making it the recommended dose for most patients. After 3 months of treatment, daridorexant 50 mg reduced the time needed to fall asleep, with a decrease in latency to persistent sleep of 34.8 minutes (95% CI: –38.1–-31.5; p<0.0001), and improved sleep maintenance by reducing wake after sleep onset by 29.4 minutes (95% CI: –33.4–-25.4; p<0.0001; Figure 2). The safety profile of daridorexant was favourable, with adverse events comparable across treatment and placebo groups and no evidence of a dose-related increase in adverse events.
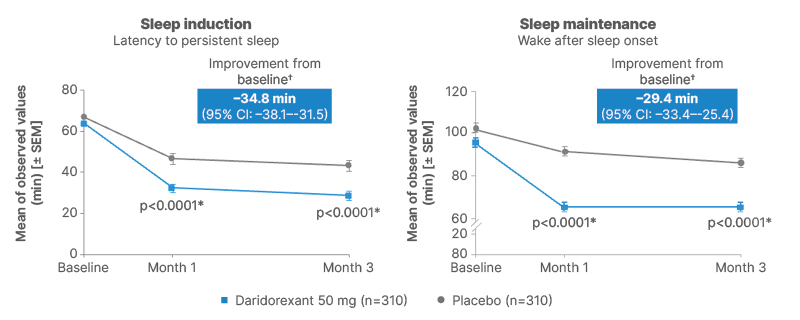
Figure 2: Sleep induction and maintenance outcomes in daridorexant Phase III trials.45
*Statistically significant versus placebo after multiplicity adjustment.
†LSM change from baseline (95% CI).
Mean changes from baseline in sleep induction and WASO times.
Adapted from Mignot et al.45
LSM: least squares mean; SEM: standard error of the mean; WASO: wake after sleep onset.
Patients completing the initial 3-month treatment period were offered the opportunity to enrol in a 40-week extension study46 to assess long-term outcomes. Improvements in sleep measures and daytime functioning were observed for up to 1 year.46 Improvements in self-reported total sleep time (sTST) continued during the extension period: at Week 12 of the extension, participants receiving daridorexant 50 mg reported a placebo-adjusted mean increase of 20.4 minutes in sTST (95% CI: 4.2–36.5). Improvements in sTST were numerically greater compared with placebo and remained statistically significant at Week 36 (p=0.038). These findings indicate that improvements in sleep measures with daridorexant were sustained with long-term treatment.
Daridorexant Improved Daytime Functioning in Patients with Insomnia Disorder
A strength of the daridorexant Phase III programme, as highlighted by Schaedel, was the systematic assessment of daytime functioning, an outcome often omitted from insomnia trials despite being central to patients’ lived experience. The Insomnia Daytime Symptoms and Impacts Questionnaire (IDSIQ) was used to evaluate the effect of daridorexant on daytime performance across domains, including sleepiness, cognitive functioning, mood, and physical activity. Lower scores on the IDSIQ correspond to reduced daytime impairment.
In the extension study,46 patients receiving daridorexant 50 mg showed clinically meaningful and sustained improvements in daytime functioning.47 At Weeks 12, 24, and 36, IDSIQ total scores decreased by –9.3 (95% CI: –15.1–-3.6), –9.5 (95% CI: –15.4–-3.5), and –9.1 (95% CI: –15.6–-2.7), respectively, compared with placebo (Figure 3). These changes reflect clinically meaningful improvements in key areas of daytime functioning measured by the IDSIQ, including alertness/cognition, mood, and sleepiness, with effects maintained for up to 1 year.
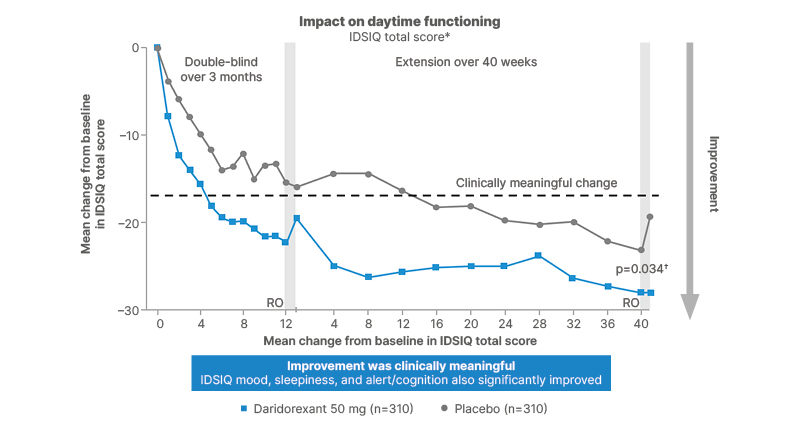
Figure 3: Sustained daytime functioning (IDSIQ) improvements over 1 year.46,47
*Mean of IDSIQ total scores, at study timepoints in patients who entered the extension study from Trial 1 and received daridorexant 50 mg or placebo.
†Two-sided p values shown are versus placebo, calculated using the linear-effects model for repeated measures. p values are for descriptive purposes only. Grey bar indicates the 7-day placebo run-out period. Baseline refers to the baseline of the confirmatory study. p-value versus placebo.
Adapted from Kunz et al.46 and Phillips-Beyer et al.47
IDSIQ: Insomnia Daytime Symptoms and Impacts Questionnaire; RO: run-out.
Daridorexant Improved Sleep in Patients with Insomnia and Comorbid OSA, Nocturia, or Mental Health Disorders
Schaedel went on to discuss several patient groups particularly relevant to clinical practice in menopausal women, beginning with those experiencing comorbid OSA. OSA frequently coexists with insomnia disorder during the menopausal transition, forming the phenotype known as COMISA. In a post-hoc analysis of the Phase III programme, daridorexant 50 mg improved key sleep outcomes in a subgroup with mild COMISA, reducing the time taken to fall asleep, decreasing nocturnal wakefulness, and increasing subjective total sleep time. Additionally, despite longstanding concerns about the use of hypnotics in patients with OSA, daridorexant did not increase daytime sleepiness or produce any signals of respiratory compromise.39,48 These findings reinforce that targeting the orexin system can improve insomnia symptoms in COMISA without adversely affecting safety.
Another important subgroup comprised patients with comorbid nocturia (defined as three or more voids per night for at least 1 month), a highly prevalent issue among menopausal women, often driven by a bidirectional relationship between sleep disruption and nocturnal awakenings. In a double-blind, placebo-controlled, crossover study of daridorexant 50 mg over 4 weeks,49 improvements in both sleep and nocturia were observed. Daridorexant increased sTST by almost an hour and reduced the number of nocturnal voids, with changes perceived as clinically meaningful. It also extended the time to the first nocturnal void by approximately 30 minutes.
Schaedel also highlighted women with comorbid mental health disorders, another group in whom insomnia symptoms are common and often intertwined with mood disturbance. In a real-world, longitudinal study from Italy that included patients with unipolar or bipolar depression, many of whom were taking antidepressants, daridorexant improved insomnia severity at both 1 month and 3 months. Symptoms of depression measured in parallel also improved over time. These findings echo larger studies demonstrating that effectively treating insomnia can support improvement in mood symptoms, which is particularly relevant given the increased prevalence of depressive episodes during the menopausal transition.
Safety and Tolerability
Across all analyses, daridorexant was well tolerated. Rates of adverse events were similar to placebo, with no excess somnolence, increase in falls, or narcolepsy-like events. Long-term data from the extension study revealed no new safety signals. Daridorexant has not been associated with dependence, withdrawal symptoms, rebound insomnia or any evidence of abuse potential, and no increase in suicidality was observed compared with placebo. These features distinguish daridorexant from earlier classes of sleep medications, and are of particular importance when considering long-term management of insomnia disorder.
In Women Aged 47–55 years, Daridorexant Improved Sleep and Daytime Functioning
Schaedel then summarised findings from a post-hoc analysis of a Phase III study focusing on women aged 47–55 years with insomnia disorder,50 a demographic that likely includes most peri-and early postmenopausal participants. Over 3 months of treatment, daridorexant 50 mg numerically decreased the number of minutes spent awake after sleep onset and decreased the time to fall asleep compared with placebo, with no evidence of deviation from the overall study population. A visual analogue scale measuring morning sleepiness showed an improvement in morning sleepiness, denoting the lack of next-morning residual effects with daridorexant. Daridorexant was well tolerated in this demographic, with the most frequently reported adverse events of nasopharyngitis and headache occurring at similar rates to placebo (four women [11%] versus five [13%] and three women [9%] versus one [3%], respectively).
Finally, Schaedel mentioned that antagonising orexin may not only improve insomnia, but could also have potential benefits for vasomotor symptoms. Given evidence in rats that oestrogen decline increases orexin signalling and may heighten sympathetic activity,51 reducing orexin-driven hyperarousal may theoretically attenuate hot flashes as well as improve sleep. This hypothesis will be explored in a Phase IV study52 in North America comparing daridorexant with digital CBTI and trazodone in peri- and postmenopausal women.
Conclusion
This symposium emphasised the importance of recognising insomnia disorder in menopausal transition as a distinct, chronic disease, rather than a secondary menopausal symptom. Speakers emphasised the need for proactive identification and targeted management to avoid long-term health consequences and reduce the substantial burden on quality of life, daytime functioning, and overall wellbeing.
While CBTI remains the recommended first-line therapy, the discussions underscored that it is often unavailable or insufficient for many women. Within this context, DORAs, such as daridorexant, were discussed as one of the pharmacological approaches supported by evidence from clinical studies and reflected in current guideline recommendations for patients who require pharmacological treatment.
The data presented, covering efficacy, safety, and outcomes in relevant menopausal subgroups, illustrated how addressing hyperarousal through orexin pathway modulation can improve both nighttime symptoms and daytime functioning. These insights contribute to a broader understanding of how targeted therapies may fit into the overall management strategy for insomnia disorder in women undergoing menopausal transition.
| Adverse events should be reported. For UK based HCPs, reporting information and forms can be found at https://yellowcard.mhra.gov.uk/. For HCPs based outside of the UK, adverse events should be reported at: https://poweredbyscience.idorsia.com/contact/ae-reporting. |






