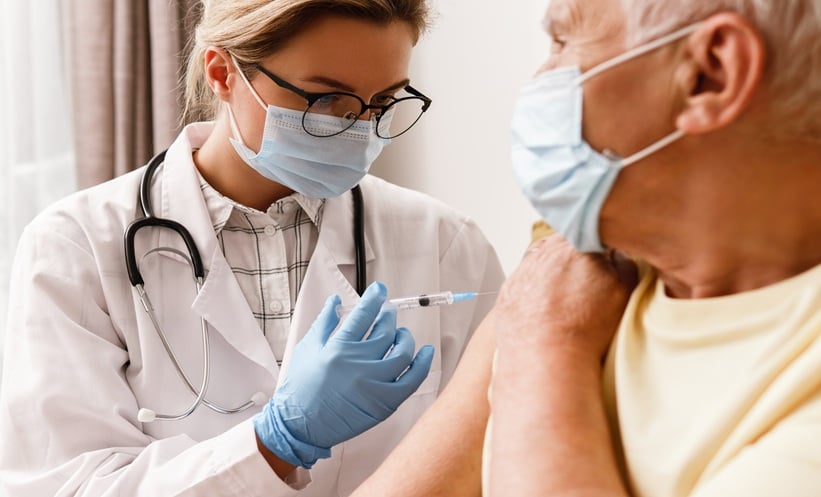
COVID-19 vaccination is strongly recommended for immunocompromised patients for the 2025–2026 season, according to new guidance.
Why These COVID-19 Vaccination Guidelines Matter
Immunocompromised patients remain at increased risk of severe COVID-19 outcomes, and vaccine protection can be reduced or less durable in some groups. To support clinical decision-making for the 2025–2026 respiratory virus season, updated guidance focuses on U.S.-licensed COVID-19 vaccines and how best to use them across common immunocompromising conditions and therapies. The scope spans patients with hematologic malignancy, primary immunodeficiency, autoimmune disease treated with immunosuppressive medications, severe HIV immunosuppression, solid organ transplant, hematopoietic cell transplantation, CAR-T therapy, and those receiving chemotherapy for solid tumors.
Evidence Supporting Seasonal COVID-19 Vaccination
The recommendations were informed by a systematic evidence review covering mid-2024 through mid-2025 and assessed using a structured certainty framework. Across available observational data, COVID-19 vaccination in immunocompromised patients was associated with fewer hospitalizations, alongside signals for reduced critical illness and mortality. Reported effectiveness estimates for preventing hospitalization varied across studies, reflecting differences in populations, vaccine products, circulating variants, and time since vaccination. The guidance also notes that many studies had limited follow-up, which can make protection appear higher if outcomes are assessed soon after vaccination, before waning becomes more apparent.
COVID-19 Vaccination Timing in Immunocompromised Patients
The guidance issues a strong recommendation for age-appropriate 2025–2026 COVID-19 vaccination for all immunocompromised individuals aged 6 months and older. Timing should be individualized based on the patient’s immunosuppressive regimen, clinical status, and local transmission patterns. In higher intensity immunosuppression, the guidance highlights practical timing considerations, including vaccinating before planned transplant when feasible, and delaying vaccination after transplant or cellular therapies to improve the likelihood of a meaningful immune response. It also addresses situations where longer intervals after B-cell depleting therapy may be considered when clinically appropriate.
Layered Prevention Beyond Vaccination
Recognizing that vaccine responses can be attenuated, the guidance emphasizes layered protection strategies. These include ensuring household and close contacts are up to date with vaccination and maintaining timely access to antiviral treatment when infection is suspected or confirmed. Together, these measures aim to reduce the likelihood of severe outcomes during seasonal COVID-19 surges in a population with wide variability in immune function and treatment exposure.
Reference: Nellore A et al. IDSA 2025 Guidelines on the use of vaccines for the prevention of seasonal COVID-19 infections in immunocompromised patients. Clin Infect Dis. 2026;doi:10.1093/cid/ciag115.