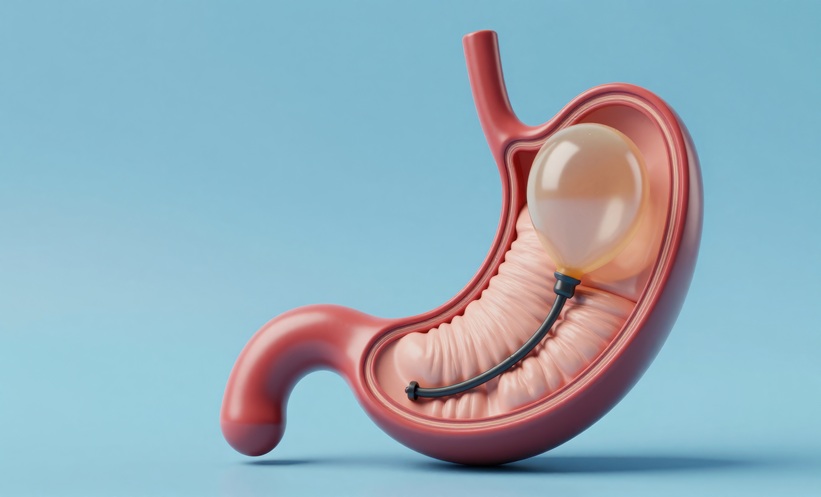
A SWALLOWABLE gastric balloon for weight loss has been granted premarket approval by the U.S. Food and Drug Administration (FDA), offering a drug-free, procedure-free option for adults with obesity.
The Allurion Gastric Balloon System was indicated for adults aged 22–65 years with obesity who had previously attempted weight loss through a structured programme without success. Designed to occupy space in the stomach and promote early satiety, the swallowable gastric balloon can be swallowed during a 15-minute office visit without the need for surgery, endoscopy, or anaesthesia.
Once ingested as a capsule, the balloon is filled with fluid by a physician and remains in the stomach for approximately 16 weeks before a release valve opens, allowing it to pass naturally. A second swallowable gastric balloon can be administered 8 weeks after the first has passed, enabling two treatment cycles.
Swallowable Gastric Balloon Demonstrated Sustained Weight Loss
Approval was supported by data from the pivotal AUDACITY trial, an open-label, multicentre, randomised controlled study involving 550 participants. Subjects were randomised 1:1 to receive two cycles of the swallowable gastric balloon plus moderate-intensity lifestyle therapy, or lifestyle therapy alone.
At 48 weeks, 58% of participants in the treatment group lost more than 5% of their total body weight, meeting the responder rate co-primary endpoint (p=0.0089). The mean difference in total body weight loss between groups at 48 weeks was 3.77%, resulting in a 2.69% superiority margin. This fell below the pre-specified 3% superiority margin required to meet the comparative co-primary endpoint (p=0.1616), partly due to higher-than-expected weight loss in the control group.
At 40 weeks, around the time the second balloon had passed, the 4.22% mean difference in total body weight loss between groups exceeded the 3% superiority margin.
Serious adverse events occurred in 3.1% of participants receiving the swallowable gastric balloon, reported as the lowest rate in a pivotal FDA trial of a liquid-filled intragastric balloon for weight loss. In real-world use outside the USA, commonly reported adverse events included nausea, vomiting, and abdominal pain, while serious events included small intestine obstruction, pancreatitis, and gastric outlet obstruction.
Despite this, Shelby Sullivan, MD, Professor of Medicine and Director, Endoscopic Bariatric and Metabolic Program Center for Digestive Health at Dartmouth-Hitchcock Health, and Lead Investigator of the AUDACITY trial, stated:
“I believe the results of the AUDACITY trial demonstrate a favourable benefit-risk profile for people with obesity. If approved, the Allurion Balloon could fill a significant gap in the treatment of obesity and serve as a valuable alternative to GLP-1 medications.”
Implications for Obesity Management
Obesity remains a major health burden and driver of cardiometabolic disease worldwide, and demand for non-surgical, non-pharmacological therapies continues to grow. The swallowable gastric balloon may serve as a standalone intervention or complement existing treatments, including glucagon-like peptide-1 receptor agonists.
As the AUDACITY trial did not meet its comparative co-primary superiority margin at 48 weeks, longer-term data and real-world monitoring will be important to clarify durability of effect and safety. For clinicians managing patients with obesity, the swallowable gastric balloon represents a novel addition to the expanding weight management toolkit.
References
MedPage Today. FDA approves a balloon that patients swallow to lose weight. Available at: https://www.medpagetoday.com/endocrinology/obesity/120029. Last accessed: 25 April 2026.
Allurion. Allurion announces positive topline results from AUDACITY trial. Available at: https://investors.allurion.com/news/news-details/2025/Allurion-Announces-Positive-Topline-Results-From-AUDACITY-Trial/default.aspx. Last accessed: 25 April 2026.
Ienca R et al. The procedureless elipse gastric balloon program: multicenter experience in 1770 consecutive patients. Obes Surg. 2020; DOI:10.1007/s11695-020-04539-8.
Featured image: Generative AI on Adobe Stock