NOVEL heart failure (HF) research has identified a surprising protective signal produced directly by heart muscle cells that may help slow the development of cardiac fibrosis, a key driver of declining heart function.
In a new experimental study, researchers found that transforming growth factor beta 3 (TGFB3) produced by cardiomyocytes acted as a natural brake on fibrotic remodelling in the failing heart. Loss of this signal significantly worsened structural damage and impaired cardiac performance in preclinical models of HF.
Cardiac Fibrosis Drives Heart Failure Progression
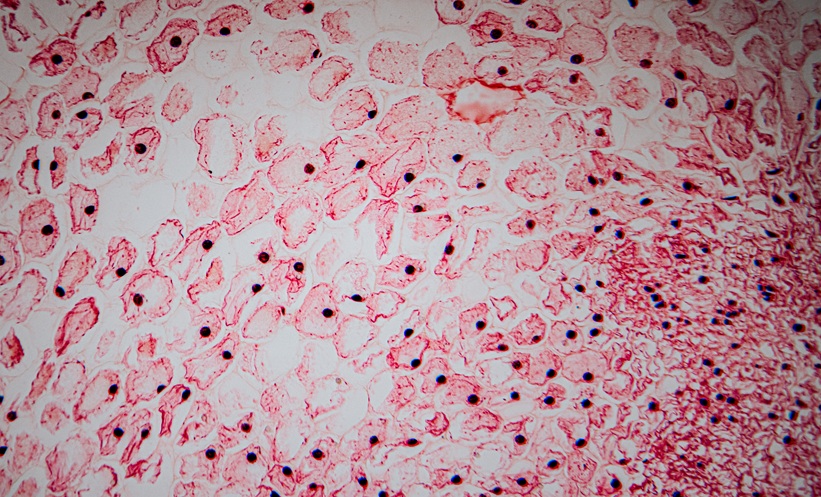
HF represents the final stage of many cardiovascular diseases and affects tens of millions of individuals worldwide. A hallmark of HF progression is cardiac fibrosis, a process in which excessive connective tissue accumulates in the heart, stiffening the myocardium and limiting its ability to pump effectively. Although several signalling pathways are known to promote fibrosis, the cellular origins and regulatory mechanisms of different transforming growth factor beta (TGFB) family members have remained unclear.
Researchers therefore analysed heart tissue from both patients with HF and mouse models of pressure overload to determine the role of TGFB3. Histological and molecular analyses indicated that TGFB3 expression originated primarily from cardiomyocytes and was induced by angiotensin II. In addition, circulating TGFB3 concentrations in patients correlated positively with plasma proBNP levels, a recognised biomarker of HF severity.
Cardiomyocyte TGFB3 Regulates Cardiac Fibrosis Signalling
To clarify the functional significance of this pathway, the team generated mice lacking TGFB3 specifically in cardiomyocytes. When exposed to pressure overload, these animals developed markedly worse cardiac dysfunction and significantly greater cardiac fibrosis compared with control mice.
Mechanistically, TGFB3 acts by competing with the more strongly profibrotic ligand TGFB1 for receptor binding. This competition reduces phosphorylation of Smad3, a key intracellular signalling molecule that drives fibrotic gene expression.
Loss of TGFB3 Accelerates Heart Failure Progression
In the absence of cardiomyocyte TGFB3, the study observed increased expression of profibrotic mediators including connective tissue growth factor (CTGF) and SERPINE1. These molecular changes promoted excessive extracellular matrix deposition and accelerated structural remodelling of the myocardium during HF progression.
The investigators suggested that cardiomyocytes therefore play a previously underappreciated role in regulating fibrosis through endogenous antifibrotic signalling. Rather than simply being passive victims of fibrotic injury, heart muscle cells appeared to actively modulate the balance between protective and pathological pathways.
Although the findings were based largely on preclinical models, the authors noted that the correlation between circulating TGFB3 and HF severity in human samples supported the clinical relevance of the pathway. Future studies will be needed to determine whether therapies that enhance cardiomyocyte-derived TGFB3 signalling could help limit cardiac fibrosis and preserve cardiac function in patients with HF.
Reference
Xuan J et al. Cardiomyocyte-derived TGFB3 attenuates cardiac fibrosis and preserves cardiac function in heart failure. Sci Rep. 2026; DOI:10.1038/s41598-026-42367-5.
Featured image: Jolanta on Adobe Stock