Meeting Summary
At The Liver Meeting® 2025, organized by the American Association for the Study of Liver Diseases (AASLD) and held in Washington, D.C., USA, hepatology experts Mandana Khalili, Professor of Medicine and Hepatologist at the University of California, San Francisco, USA; Paul Kwo, Professor of Medicine and Director of Hepatology at Stanford University, California, USA; and Norah Terrault, Professor of Medicine and Chief of Gastroenterology and Liver Diseases at the University of Southern California, Los Angeles, USA, discussed practical strategies for managing chronic hepatitis B (CHB), with a focus on indeterminate phase case studies within the context of the updated 2025 AASLD/Infectious Diseases Society of America (IDSA) treatment guidelines. The presenters discussed the treatment of patients who fall into the indeterminate phase, which is one of the most challenging aspects of CHB care. The indeterminate phase includes patients whose disease does not fit into any of the five phases of infection. Through a case-based discussion of a 38-year-old woman with borderline viral load and mildly elevated levels of transaminases, the panelists discussed how comprehensive phase characterization using tests such as quantitative hepatitis B surface antigen (HBsAg) measurement and non-invasive fibrosis assessment can guide treatment decisions. The presenters highlighted the importance of shared decision-making for the management of indeterminate phase CHB and concurrent management of metabolic dysfunction. The panelists also emphasized that caution is needed when considering discontinuation of antiviral treatments, given the high relapse rates in individuals who discontinue treatment. The session underscored that CHB is a dynamic disease that requires serial assessments and long-term monitoring, as well as individualized surveillance and treatment decisions based on age, sex, fibrosis stage, family history, and patient preferences. The session underscored the clinical utility of quantitative HBsAg testing for disease phase determination, risk stratification, and treatment monitoring, and emphasized that achieving functional cure should be the ultimate goal of CHB management.
Introduction
Chronic hepatitis B affects approximately 1.7 million people in the United States and represents a significant public health challenge because of its potential to cause severe liver disease, including cirrhosis and hepatocellular carcinoma (HCC).1,2 An educational session sponsored by GSK at The Liver Meeting® 2025, held in Washington, D.C., USA, from November 7–11, 2025, brought together distinguished hepatology experts and guideline authors to discuss practical, patient-centered strategies for managing CHB.3 The session was timely, as updated AASLD/IDSA guidelines for CHB treatment were published a few days before the meeting.2 The updated guidelines were discussed extensively during the session.
The educational session was led by three panelists: Mandana Khalili, Paul Kwo, and Norah Terrault.3 Through an interactive format combining audience polling and case-based discussion, the session addressed barriers and unmet needs in CHB management, including the role of quantitative HBsAg testing, focusing on the clinically challenging indeterminate phase of CHB infection. The panelists also discussed updated evidence-based recommendations for the management of CHB.3 This article synthesizes the key discussions from that session, providing clinicians with actionable insights for optimizing CHB care in their practice.
The Burden of Chronic Hepatitis B
Khalili, the session moderator, started the presentation by emphasizing the high disease burden of CHB.3 Approximately 1.7 million Americans live with CHB, although some estimates suggest that the true burden may be as high as 4.1 million.2,4,5 The prevalence of CHB in the United States is higher in foreign-born individuals than in those born in the country, and the majority of persons with CHB in the United States are of Asian ethnicity.4,5 In 2023, there were more than 17,000 new cases of chronic hepatitis B virus (HBV) infections in the United States, with approximately 46% occurring in individuals aged 30–49 years.6
In 2023, viral hepatitis accounted for approximately 1.1 million deaths globally.7 If left untreated, 15–40% of individuals with CHB may develop cirrhosis, liver failure, or HCC over their lifetime.8,9 CHB is the most common etiology for HCC worldwide, accounting for at least 50% of HCC cases globally.10 Mortality rates linked to CHB have been rising since 2019, primarily attributed to an increase in hepatitis-associated cancers.11
Phases of Chronic Hepatitis B
Through a patient case study, the panelists discussed the importance of accurately characterizing the phase of CHB infection, as this fundamentally guides management decisions. The presenters emphasized that CHB is a dynamic disease that evolves over a person’s lifetime, making regular monitoring essential.2,3 The guidelines specify that normal ALT is defined as <25 U/L for females and <35 U/L for males.2 These thresholds are lower than the upper limits used in many clinical laboratories.12
The updated AASLD/IDSA guidelines define five phases of CHB (Figure 1):2
- Immune tolerant phase: Characterized by hepatitis B e-antigen (HBeAg) positivity, very high HBV DNA levels (>10,000,000 IU/mL), and persistently normal alanine aminotransferase (ALT) levels (<25 IU/mL in women, <35 IU/mL in men).2HBeAg-positive immune active phase: Characterized by HBeAg positivity, elevated ALT (≥2× upper limit of normal [ULN]), and HBV DNA levels >20,000 IU/mL.2
- HBeAg-negative immune active phase: Occurs after HBeAg seroconversion, with elevated ALT (≥2× ULN) and HBV DNA >2,000 IU/mL2 despite HBeAg negativity.2
- Inactive phase: Defined by HBeAg negativity, normal ALT levels, and low HBV DNA (<2,000 IU/mL).2
- HBsAg-negative phase: Represents loss of HBsAg due to antiviral therapy or spontaneously in a person who was previously positive for HBsAg.2
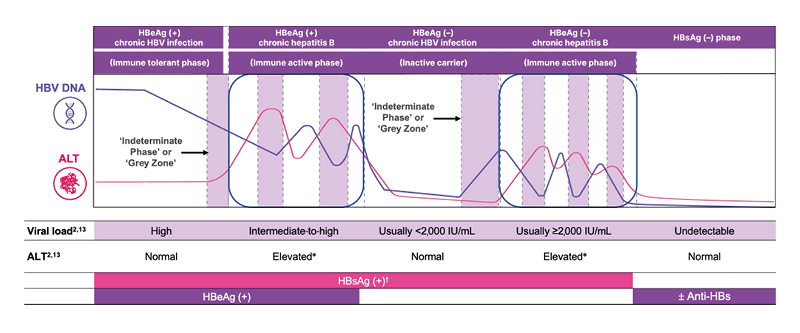
Figure 1: Phases of chronic hepatitis B infection.2,8,13
*Persistently or intermittently.
†Can also occur in patients who are HBsAg (–).
ALT: alanine aminotransferase; anti-HBe: antibody to HBeAg; anti-HBs: antibody to HBsAg; HBeAg: hepatitis B e antigen; HBsAg: hepatitis B surface antigen; HBV: hepatitis B virus.
The Indeterminate Phase of Chronic Hepatitis B
A large portion of the session focused on what the guidelines referred to as the “indeterminate phase” or “grey zone” of CHB, a term used to describe the disease state in patients who do not fit into any of the defined phases.3 The presenters emphasized that indeterminate phase CHB is common.3 Studies suggest that up to 30–50% of individuals with CHB fall into indeterminate phases at some point during their disease course.8 Patients in the HBeAg-negative indeterminate phase may have a high risk of HCC incidence and mortality compared to those in the inactive phase.14-16
Terrault explained that the indeterminate phase encompasses patients who are HBsAg-positive but have ALT and HBV DNA levels that do not meet thresholds for either immune active disease or inactive infection.2,3 According to the updated AASLD/IDSA guidelines, indeterminate phase CHB can predominantly manifest as normal (<ULN) or mildly elevated ALT (<2× ULN) with HBV DNA ≥2,000 IU/mL; or as elevated ALT (>ULN) with HBV DNA <2,000 IU/mL.2 However, the criteria for different disease phases, including the indeterminate phase, can vary depending on the guidelines being used.2,8,13
Khalili noted that decisions on the management of indeterminate phase CHB can be challenging, as treatment is not always recommended based on high HBV DNA levels alone.2,3,13 Previous guidelines recommend monitoring individuals in indeterminate phase CHB until they transition to a more clearly defined phase, highlighting the need for biomarkers to refine the disease phase and determine the need for treatment.17,18 However, accumulating evidence suggests that patients in the indeterminate phase are at higher risk of fibrosis progression and HCC than those in the inactive or immune tolerant phases.19,20
To illustrate the challenges in the management of indeterminate phase CHB, the panelists presented a case scenario involving a 38-year-old woman who was born in Vietnam and moved to the United States at the age of 15 years.3 She worked as a warehouse manager and was found to be HBsAg-positive on routine screening, with an initial HBV DNA of 1,500 IU/mL.3
When seen by a hepatologist 4 months later, the patient was asymptomatic but concerned about her diagnosis, as she was getting married soon and wanted to have children.3 Her laboratory assessments showed that she was positive for HBsAg, negative for HBeAg, and positive for anti-hepatitis B e (anti-HBe). She had an HBV DNA level of 2,500 IU/mL and an ALT level of 31 U/L (ULN: 25 U/L for females).3 Her platelet count was 160×10⁹ /L, and her BMI was 25.3 kg/m².3
When audience members were polled about the CHB disease phase in this patient, the majority initially categorized her disease phase as immune tolerant. However, Kwo pointed out her viral load and laboratory testing results at presentation do not neatly fit into any of the five phases of CHB.3 He explained that the patient was negative for HBeAg, ruling out the immune tolerant phase.3 Her viral load of 2,500 IU/mL and her mildly elevated ALT level place her in the indeterminate, “grey zone” phase.3
The Role of Imaging and Biomarkers in Chronic Hepatitis B Assessment
According to the CDC screening guidelines, serological testing for HBsAg, anti-hepatitis B surface antigen (anti-HBs), and total anti-hepatitis B core (anti-HBc) is recommended to assess immunity and past infection.2,17,21,22 However, the panelists emphasized that establishing the phase of CHB infection and determining appropriate treatment strategies require assessment beyond serological testing.3 Kwo noted that serial monitoring with additional imaging and biomarker assessments is necessary to accurately characterize disease phase and guide management decisions.3
The latest AASLD/IDSA guidelines recommend several imaging and assessment strategies to evaluate disease severity and stage fibrosis, including vibration-controlled transient elastography (VCTE), to assess for and stage fibrosis, offering better performance than serum markers; serum fibrosis markers (e.g., fibrosis-4 index) when elastography is unavailable; and liver biopsy, which is infrequently used but the most accurate for grading liver inflammation and fibrosis.2
The guidelines also note that quantitative HBsAg may be considered for guiding decisions about discontinuing nucleos(t)ide analogue therapy (threshold <100 IU/mL).2
For the patient in this case scenario, imaging assessments included VCTE, which showed liver stiffness of 6.8 kPa (suggesting minimal fibrosis) and controlled attenuation parameter (CAP) of 275 dB/m (indicating hepatic steatosis); and abdominal ultrasound, which revealed mildly echogenic liver consistent with fatty liver, with no evidence of cirrhosis, nodularities, or portal hypertension.3 Additional laboratory testing showed that the patient was negative for HIV RNA and for hepatitis C virus and hepatitis D virus antibodies with a quantitative HBsAg value of 1,200 IU/mL.3
The Role of Quantitative Hepatitis B Surface Antigen
The panelists discussed the importance of quantitative HBsAg in the evaluation and management of CHB.3 According to Kwo, the quantitative HBsAg level of 1,200 IU/mL in the case scenario further supports that the patient is between the inactive and active phases of CHB.3 Quantitative HBsAg levels below 1,000 IU/mL are generally associated with the inactive phase and a lower risk of HCC development, whereas higher levels suggest more active infection.23-25 Very low HBsAg levels (≤100 IU/mL) are predictive of spontaneous HBsAg seroclearance, which is a key component of functional cure.3,13
Quantitative HBsAg may be used as a biomarker for CHB to monitor HBV natural infection by distinguishing true HBeAg-negative chronic infection from active CHB or by helping clinicians predict spontaneous HBsAg seroclearance, with HBsAg levels <100 IU/mL being significantly associated with spontaneous HBsAg loss.13,26-28 According to European Association for the Study of the Liver (EASL) 2025 guidelines, quantitative HBsAg testing may be valuable for determining disease phase, with 85–94% accuracy for identifying inactive infection in individuals with HBV DNA <2,000 IU/mL + HBsAg <1,000 IU/mL, and highest specificity across HBV genotypes in those with HBsAg <100 IU/mL + HBV DNA <2,000 IU/mL.13 The EASL 2025 guidelines recommend that HBsAg levels using quantitative HBsAg testing should be determined every 12 months.13
Quantitative HBsAg may also be helpful in predicting prognosis in individuals with CHB.29 It may be used to inform follow-up and treatment decisions, especially in indeterminate CHB, and to assess the risk of HCC and determine HCC screening needs.27,30,31 The EASL 2025 guidelines note that combining quantitative HBsAg with HBV DNA levels provides better prognostic information than either marker alone.13
In addition, quantitative HBsAg measurements may be useful for monitoring treatment response and predicting response to antiviral therapies.2,27,28 HBsAg titers may help clinicians identify patients with CHB who may be eligible for clinical trials and respond to investigational medicines.2,18,27
However, the guidelines emphasize that quantitative HBsAg should not replace HBV DNA measurement; rather, HBsAg titers and HBV DNA levels may be used in combination to differentiate the phases of CHB and guide management decisions.2,13 The AASLD/IDSA 2025 guidelines also note a quantitative HBsAg level of <100 IU/mL as one of six criteria to consider stopping treatment; however, it does not guarantee the absence of risk of progression, and that patient monitoring is required after withdrawal of treatment.2
Importance of Shared Decision-Making
The speakers discussed the importance of shared decision-making in the management of CHB, particularly for patients in indeterminate phase disease.3 The new AASLD/IDSA guidelines suggest consideration of antiviral therapy using a shared decision-making approach, rather than monitoring, for adults with HBsAg-positive, HBeAg-negative chronic HBV infection without cirrhosis and in the indeterminate phase, as was the case discussed in this session.2
Factors to consider in shared decision-making regarding antiviral treatment initiation include age (risk increases with age >40 years), sex (males at higher risk), platelet count (low-normal counts may suggest more advanced fibrosis), fibrosis stage (advanced fibrosis warrants treatment), family history of HCC, patient preferences and concerns, and implications of long-term treatment commitment.2 The shared decision-making approach should also consider the risks, benefits, and costs of treatment.2
Terrault noted that, in addition to providing information about the disease and the potential benefits and risks of treatment, discussions with patients should also aim to understand their goals, personal values, preferences, and concerns.2,3 Clinicians and patients may consider reevaluating that decision at each follow-up visit if treatment has not been initiated, as patient values and preferences may change.2
Management of Co-existing Conditions
The case scenario also prompted discussion around the management of CHB in individuals with concurrent metabolic dysfunction-associated steatotic liver disease (MASLD) and the importance of assessing metabolic health in all patients with liver disease.3 Metabolic assessment of the patient case indicated elevated levels of triglycerides (340 mg/dL) and normal comprehensive metabolic panel, including glucose.3 Kwo noted that the finding of hepatic steatosis in the case scenario added complexity to the management of CHB, and that 35–42% of patients with CHB have concurrent MASLD.3,32,33
While the AASLD/IDSA guidelines do not provide recommendations for the management of CHB specifically in patients with metabolic dysfunction, the EASL guidelines emphasize the importance of managing both conditions.13 Although antiviral therapy indications remain the same as for patients without metabolic disease (based on HBV DNA, ALT, fibrosis stage), metabolic disease modifies prognosis, and even patients in indeterminate CHB may progress faster if metabolic risk factors are present.13 Therefore, in addition to treating HBV with nucleos(t)ide analogues, the EASL guidelines also recommend addressing metabolic disease through lifestyle, weight reduction, and diabetes control.3,13
The case scenario prompted discussion about the management of CHB during pregnancy.3 The updated AASLD/IDSA guidelines provide recommendations for the prevention of mother-to-child transmission of HBV.2 For pregnant individuals with HBV DNA >200,000 IU/mL, the guidelines recommend initiating tenofovir disoproxil fumarate (TDF) or tenofovir alafenamide (TAF) at gestational Week 28 to prevent transmission.2 For mothers with HBV DNA levels >200,000 IU/mL, TDF or TAF prophylaxis should be combined with subsequent infant vaccination, if hepatitis B immune globulin use in the infant is unavailable.2
For pregnant individuals with CHB who discontinue antiviral therapy after delivery, guidelines recommend HBV DNA and ALT monitoring every 1–3 months for up to 6 months to detect potential flares, and treatment reinitiation in case of flare (ALT ≥5× ULN).2 Breastfeeding is safe while taking TDF or TAF, and does not increase the risk of HBV transmission if infants receive appropriate immunoprophylaxis.2
The Management of Immune Tolerant Phase Chronic Hepatitis B
The session also covered management of the immune tolerant phase.3 Patients who consistently meet all criteria for immune tolerant disease (HBeAg-positive, ALT <ULN, HBV DNA >10⁷ IU/mL) have a low risk of HCC.2,34,35 However, older age (>40), lower HBV DNA, higher ALT, and lower platelet counts may indicate transition out of the immune tolerant phase and higher HCC risk.2
The updated AASLD/IDSA guidelines suggest antiviral therapy for persons in the immune tolerant phase if they are older than 40 years or have significant liver inflammation (≥Grade 2) or fibrosis (≥F2) on biopsy or non-invasive tests.2 For persons younger than 40 years, observation and monitoring are generally preferred.2 Treatment may be considered for individuals younger than 40 through shared decision-making if the patient has risk factors such as family history of HCC, adherence concerns, or high transmission risk.2
The guidelines emphasize the importance of distinguishing patients with true immune tolerance from those who are transitioning to immune active disease.2 This may require serial monitoring of ALT and HBV DNA levels every 6 months.2
Cessation and Reinitiation of Antiviral Therapy
The speakers discussed factors to take into account when considering cessation of nucleos(t)ide analogue therapy in CHB.3 The new AASLD/IDSA guidelines recommend against withdrawing nucleos(t)ide analogue therapy in HBeAg-negative adults without cirrhosis who have achieved sustained undetectable HBV DNA until HBsAg loss is achieved.2 Terrault explained that although some studies suggest that treatment withdrawal may increase the likelihood of HBsAg clearance, the potential risks, including ALT flares, virological relapse, hepatic decompensation, and death, are substantial.2,3
For patients who strongly desire to stop therapy despite these risks, the guidelines outline strict criteria that must all be met.2 These criteria include the absence of history of cirrhosis, hepatic decompensation, HCC, or extrahepatic complications; negative HBeAg status with undetectable HBV DNA for ≥2 years; quantitative HBsAg <100 IU/mL; no HIV or hepatitis D virus coinfection; and willingness to adhere to frequent monitoring.2
AASLD/IDSA 2025 guidelines recommend that individuals who discontinue treatment should be closely monitored and ALT as well as HBV DNA checked every 1–3 months for the first 6 months, then every 3 months for up to a year, and subsequently at 3–6-month intervals depending on results.2 Treatment must be restarted immediately if HBV DNA rises ≥10,000 IU/mL, ALT exceeds ≥5× ULN, bilirubin surpasses 2.5 mg/dL, or if there are any signs of hepatic decompensation.2 Treatment may also be restarted at the patient’s request, in the presence of extrahepatic complications, or if standard treatment criteria are again met (HBV DNA >2,000 IU/mL with ALT >2× ULN).2
The panelists emphasized that quantitative HBsAg measurement could help clinicians identify appropriate candidates for treatment cessation and predict outcomes after withdrawal.3 The 2025 AASLD/IDSA guidelines specify that quantitative HBsAg <100 IU/mL is among the criteria that must be met before considering cessation, as this threshold is associated with higher likelihood of sustained off-treatment response and lower risk of virological relapse.2 However, even with quantitative HBsAg <100 IU/mL, close monitoring remains essential as this does not guarantee the absence of relapse risk.2
Conclusions
The GSK-sponsored session on chronic hepatitis B at AASLD 2025 was part of the Industry Programming Guide Sponsored Theater and featured an expert panel discussion of an indeterminate phase patient case.3 The updated AASLD/IDSA guidelines provide a more patient-centered approach that acknowledges the heterogeneity of CHB presentations and the importance of shared decision-making.2
The session highlighted that up to 50% of patients with CHB may be in the indeterminate phase, and that patients in indeterminate phase CHB are at higher risk of fibrosis and HCC compared with those in the inactive or immune tolerant phase.8,19,20 The expert panelists discussed that for patients in indeterminate phase CHB, clinicians engage in discussions about the potential risks and benefits of treatment, considering individual factors such as age, sex, fibrosis stage, and patient preferences.2
Serological tests, including quantitative HBsAg measurements, are becoming increasingly important in guiding clinical decisions for patients with CHB.2,13,36 Quantitative HBsAg testing provides valuable information for disease phase determination, risk stratification, treatment response monitoring, and identification of candidates for treatment cessation.2,13,36 Similarly, recognition and management of concurrent metabolic dysfunction is essential, as the intersection of CHB and MASLD becomes more common.2,13
Khalili concluded the session by reminding attendees that achieving functional cure, characterized by sustained HBsAg loss with undetectable HBV DNA off treatment for at least 6 months, should be the ultimate goal of CHB management, as it may reduce the risk of liver-related complications.2,3,13,31







