BACKGROUND AND AIMS
Following the PRECISION trial, prostate multiparametric MRI has been adopted as the first-line investigation for suspected prostate cancer internationally.1 However, international data demonstrates that, due to resource limitations, not every patient who requires a pre-biopsy MRI has been receiving one. The PRIME trial has recently demonstrated the non-inferiority of biparametric MRI to multiparametric MRI for clinically significant prostate cancer detection,2 and this is a step towards allowing more men to be scanned with existing resources.
With the global incidence of prostate cancer predicted to double in the next 20 years,3 the potential adoption of biparametric MRI,2 and the introduction of national MRI screening programmes,4 the demand for prostate MRI is set to rise substantially. Interpretation, however, has a steep learning curve, with optimal performance achieved by expert genitourinary radiologists.5 With a rising demand for medical imaging and a projected 40% radiologist shortfall by 2027,6 a prompt international solution is warranted.
PARADIGM aims to evaluate whether AI is non-inferior to radiologists in detecting clinically significant prostate cancer (Gleason Grade Group ≥2).7,8
METHODS
PARADIGM is an international, prospective, multicentre, non-inferiority, within-patient, level 1 evidence diagnostic study. Five hundred men will be recruited over 18 months. These men will undergo standard of care MRI with either 1.5 or 3.0 T and at least a pelvic phased array coil. The radiologist and a primary AI algorithm will report the MRI, blinded from each other. The radiologist will then be unblinded and produce a merged report, with the ability to overrule AI findings for safety. Suspicious lesions identified by either AI or the radiologist will undergo targeted biopsies, with optional perilesional and/or systematic biopsies. The primary outcome is the proportion of men with clinically significant cancer. Planned secondary outcomes include the proportion of men with clinically insignificant cancer (Gleason Grade Group 1), test performance characteristics of AI and radiologists, and health economics analysis.
RESULTS
Eighty-one centres from 25 countries (six continents) have expressed an interest in taking part and are undergoing pre-trial MRI quality control (Figure 1). PARADIGM will open to recruitment in October 2026.
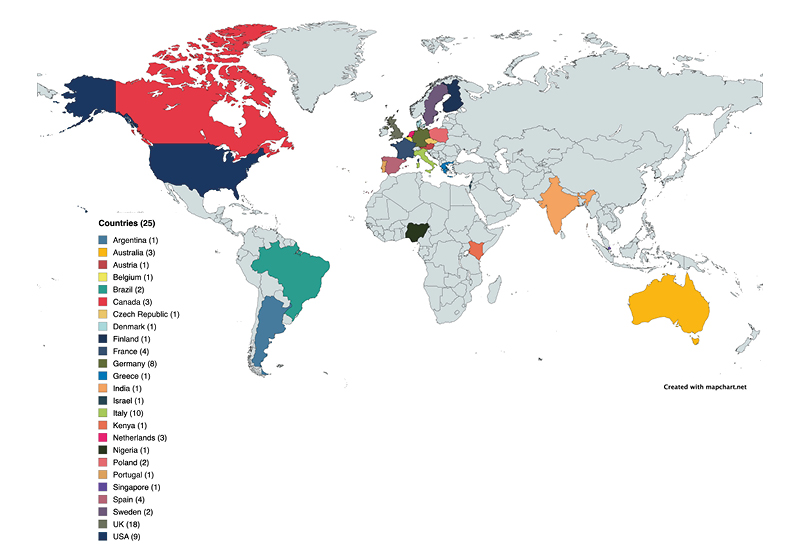
Figure 1: Eighty-one centres from 25 countries and six continents have expressed interest in taking part in the PARADIGM trial.
CONCLUSION
PARADIGM will provide the first prospective, Level 1 evidence on the diagnostic performance of AI in the detection of clinically significant prostate cancer on MRI.