Meeting Summary
Toripalimab is a humanised therapeutic monoclonal antibody that blocks the interactions of programmed cell death protein 1 (PD-1) with programmed death ligand 1 (PD-L1) and 2, thereby enhancing the immune system’s ability to eliminate malignant cells. It is approved in Europe as a first-line treatment for nasopharyngeal carcinoma (NPC) and advanced oesophageal squamous cell carcinoma (OSCC), in combination with chemotherapy. This article summarises Phase III clinical trial data from two presentations on toripalimab at the recent 2025 European Society of Medical Oncology (ESMO) Asia Congress.
Long-term overall survival (OS) data from the JUPITER-02 trial of toripalimab in combination with gemcitabine-cisplatin (GP) as first-line treatment for recurrent or metastatic NPC were presented by Qiu-Yan Chen from Guangzhou Medical University, China. After 6 years’ follow-up, toripalimab plus chemotherapy continued to demonstrate significant survival advantages over chemotherapy alone, with median OS reaching 64.8 months in the toripalimab arm compared to 33.7 months with GP.
Yan-Xing Chen from Sun Yat-Sen University Cancer Center, Guangzhou, China, presented data from the final OS analysis and biomarker evaluation of the JUPITER-06 study of toripalimab in combination with chemotherapy in patients with advanced or metastatic OSCC. Median OS was 17.7 months in the toripalimab plus cisplatin-paclitaxel (TP) arm, compared to 12.9 months with placebo plus TP, confirming the sustained survival benefit of toripalimab plus chemotherapy in the first-line treatment of advanced OSCC. Potential genomic biomarkers associated with improved outcomes from toripalimab-based immunochemotherapy were also identified.
Toripalimab in Nasopharyngeal Carcinoma: Long-Term Overall Survival Follow-Up of JUPITER-02
Toripalimab was the first immunotherapy to be approved for the treatment of NPC in Europe on the basis of the Phase III JUPITER-02 study.1,2 It is indicated in combination with GP for the first-line treatment of adult patients with recurrent, not amenable to surgery or radiotherapy, or metastatic NPC.3 NPC is a type of cancer that affects the nasopharyngeal mucosal epithelium and is driven primarily by latent Epstein–Barr virus infection.2 Additional risk factors include alcohol consumption and tobacco use.2 NPC constitutes a rare form of head and neck cancer in Western countries, but is markedly more prevalent in Asian regions, where over 80% of reported cases occur.4,5
JUPITER-02 was an international, multicentre, randomised, double-blind Phase III study conducted in NPC-endemic regions, including mainland China, Taiwan, and Singapore. It enrolled 289 patients, aged 18–75 years old, with primary metastatic or recurrent NPC after curative-intent therapy who were treatment naïve for recurrent/metastatic disease.2 Patients in this study were randomised 1:1 to receive toripalimab 240 mg (n=146) or placebo (n=143) in combination with GP once every 3 weeks (Q3W) for up to six cycles. This was followed by maintenance therapy with toripalimab or placebo Q3W until disease progression, intolerable toxicity, or a maximum of 2 years of treatment. Stratification was performed according to patients’ Eastern Cooperative Oncology Group (ECOG) performance (0 versus 1) and disease status (recurrent versus primary metastatic). The primary endpoint of the JUPITER-02 study was progression-free survival (PFS), as evaluated by a blinded independent review committee (BIRC), and OS was a key secondary endpoint.2,6
In the original final OS (overall survival) analysis of JUPITER-02, carried out at 36.0 months follow-up, median OS was not reached in the toripalimab plus chemotherapy arm compared to 33.7 months for placebo plus chemotherapy, with a stratified hazard ratio (HR) for death of 0.63 (95% CI: 0.45–0.89; p=0.0083).2 The updated survival data from the JUPITER-02 study presented at the 2025 ESMO Asia congress provided long-term OS results after a total of 6 years of follow-up. At a data cut-off date of 24th June 2025, 68 months after the last patient enrolment into the trial, a total of 156 deaths had occurred.7 The median OS was 64.8 months in the toripalimab plus GP arm compared to 33.7 months in the placebo plus GP arm, with a stratified HR for death of 0.62 (95% CI: 0.45–0.85; nominal p=0.0027; Figure 1).6 As Chen outlined, these results equate to a 31.1-month improvement in median OS in patients treated with toripalimab plus chemotherapy as compared to chemotherapy alone. The significant OS benefit of toripalimab over chemotherapy was maintained across most key patient subgroups analysed. One exception was patients with a metastatic baseline disease stage per CRF (n=117), where the HR for toripalimab plus GP versus placebo plus GP was 1.13 (95% CI: 0.68–1.87; p=0.6373).6
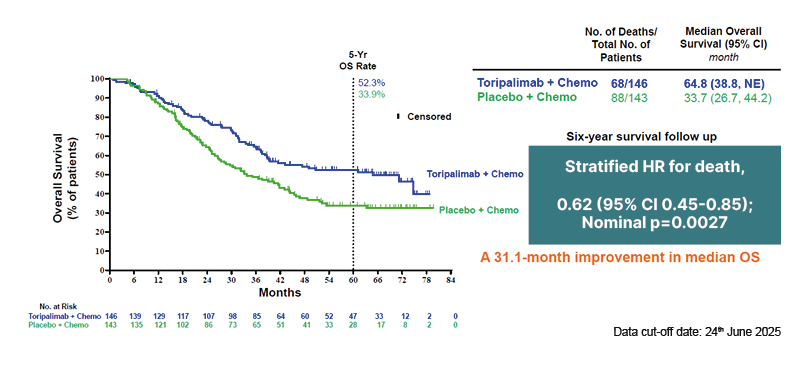
Figure 1: Overall survival in the intention-to-treat population of the Phase III JUPITER-02 study following long-term follow-up.6
A sensitivity analysis of the JUPITER-02 trial was also conducted to further evaluate the impact of post-progression immunotherapy on survival. In total, among patients experiencing disease progression during the course of the study, 46% from the toripalimab arm and 40% from the control arm received subsequent anti-PD-1/PD-L1 therapy.6 After adjusting for post-progression anti-PD-L1 therapy among the intent-to-treat population, the median OS was 61.0 months in the toripalimab arm, compared to 25.1 months in the placebo arm, with an HR of 0.52 (95% CI: 0.38–0.72). This compares to unadjusted OS rates of 64.8 months versus 33.7 months (HR: 0.62; 95% CI: 0.45–0.85; p=0.0027), respectively. The improved HR of the sensitivity analysis over the unadjusted analysis suggests that post-progression immunotherapy had a more favourable effect on survival in the placebo arm.6,7
In summary, toripalimab plus GP chemotherapy demonstrated significant survival benefits over GP alone as a first-line treatment for recurrent or metastatic NPC in this long-term follow-up of the JUPITER-02 trial. The median OS reached 64.8 months in the toripalimab arm, equivalent to a 31-month improvement over GP alone. Toripalimab plus GP is now approved in over 40 countries worldwide and, as Chen concluded, represents an important new option for the treatment of patients with recurrent or metastatic NPC.6,7
Toripalimab in Oesophageal Squamous Cell Carcinoma: Final Analysis and Biomarker Evaluation of JUPITER-06
Oesophageal cancer ranks as one of the most common malignancies globally and, like NPC, is particularly prevalent in Asian regions of the world.8 Histologically, oesophageal cancer can be classified as either squamous cell carcinoma or adenocarcinoma.9 The squamous histology is the predominant subtype globally and in Asia, where it accounts for 90% of all cases. In contrast, OSCC only constitutes around a third of all oesophageal cancer incidences in Western countries of the world.9
Toripalimab is approved for the first-line treatment of adult patients with unresectable advanced, recurrent, or metastatic OSCC in Europe in combination with cisplatin and paclitaxel.3 This approval in OSCC was based on an interim analysis of the JUPITER-06 trial (NCT03829969) in which toripalimab plus chemotherapy demonstrated significant improvement in both PFS and OS over chemotherapy alone, irrespective of patients’ PD-L1 expression levels.9
JUPITER-06 was a randomised, double-blind, placebo-controlled, Phase III trial carried out at multiple study sites across China. A total of 514 previously untreated patients with advanced or metastatic OSCC were randomly assigned in a 1:1 ratio to receive either toripalimab (240 mg) or placebo in combination with TP (paclitaxel 175 mg/m2 and cisplatin 75 mg/m2) Q3W for up to six cycles. As in the JUPITER-02 trial, initial study treatment was then followed by toripalimab or placebo maintenance Q3W continued until progressive disease, intolerable toxicity, withdrawal of consent/investigator’s judgement, or a maximum treatment of 2 years. The dual primary endpoints of the JUPITER-06 study were PFS by blinded independent central review (BICR) and OS. Secondary endpoints included investigator-assessed PFS, response rates, safety, and health-related quality of life.9,10
Final OS outcomes from the JUPITER-06 trial after a median follow-up time of 14.2 months were presented at ESMO Asia. At this final analysis, a significant improvement in OS was observed for the toripalimab arm over the placebo arm: 17.7 months versus 12.9 months (HR: 0.72; 95% CI: 0.58–0.88; p=0.002), 3-year OS rates were 29.7% and 19.9%, respectively (Figure 2). This significant survival benefit for toripalimab plus chemotherapy over chemotherapy alone was maintained across all key patient subgroups analysed.10
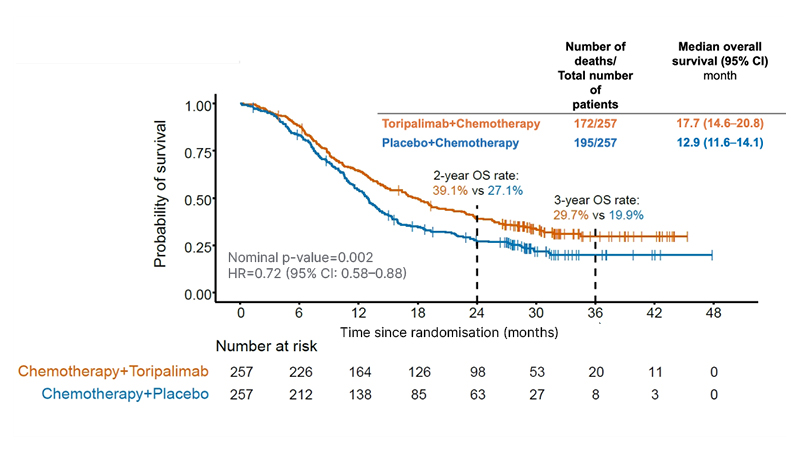
Figure 2: Overall survival in the intention-to-treat population of the Phase III JUPITER-06 study at the final analysis.10
HR: hazard ratio; OS: overall survival.
Toripalimab demonstrated a manageable safety profile in the original analysis of JUPITER-06, with a similar incidence of Grade ≥3 treatment-emergent adverse events between the two study arms.9 In this final analysis of JUPITER-06, no new safety signals were identified with the addition of toripalimab to chemotherapy over the extended follow-up period.11 The most commonly reported treatment-emergent adverse events with an incidence of 30% or more in any treatment arm were anaemia, leukopenia, neutropenia, nausea, fatigue, vomiting, decreased appetite, alopecia, thrombocytopenia, and peripheral neuropathy.10
As part of the JUPITER-06 study, whole exome sequencing was performed to characterise genomic profiles in 486 patients, the aim being to explore potential genomic biomarkers predictive of long-term survival. Neither total tumour mutation burden (TMB) nor clonal TMB correlated with long-term survival benefit in the toripalimab arm. However, when OS was evaluated by copy number alteration-corrected TMB (ccTMB), patients in the toripalimab arm with higher copy ccTMB exhibited a significant improvement in 3-year OS rate compared to those with lower ccTMB: 43.6% versus 24.6%, respectively.10,11
Similarly, the established genome-based immuno-oncology classification (EGIC) scheme effectively identified long-term survivors on anti-PD-1 plus chemotherapy. More durable survival benefit was seen in patients with the EGIC1 classification, with a median OS of 30.4 months on toripalimab plus TP compared to 11.2 months for placebo plus TP, and 3-year OS rates of 48.9% versus 16.8%, respectively.10,11
Considering the profiles of targetable alteration events, switch/sucrose non-fermenting (SWI/SNF) complex alterations were found to be associated with improved long-term outcomes under toripalimab combination therapy. Focusing specifically on oncogenic risk alterations, activating alterations in both the cell cycle pathway and wingless-related integration site (WNT) pathways were linked to reduced immunochemotherapy benefit. Cyclin-dependent kinase 4/6 (CDK4/6) inhibitors and porcupine (PORCN) inhibitors were identified as potential combination partners to further enhance treatment efficacy and improve outcomes in patients with these pathway activations, based on evidence from the Cancer Genome Interpreter database.10,11
In summary, this pre-specified final OS analysis of the JUPITER-06 study presented at ESMO Asia confirmed the sustained survival benefit obtained with the addition of toripalimab to chemotherapy in first-line advanced or metastatic OSCC, with no new safety signals identified during the extended follow-up period. ccTMB and the extended EGIC scheme were identified as potential promising predictive biomarkers for long-term survival with anti-PD1-based combination therapies.10,11
| Adverse events should be reported. Reporting forms and information can be found at: https://yellowcard.mhra.gov.uk/ or search for MHRA Yellow Card in the Google Play or Apple App Store. Adverse events should also be reported to Drug Safety at LEO Pharma by calling +44 (0) 1844 347333 or emailing medical-info.uk@leo pharma.com. |
MAT-91745