BACKGROUND AND AIMS
The choice of menopausal hormone therapy (MHT) is traditionally guided by the need to alleviate climacteric symptoms by correcting the deficiency of oestrogens and progesterone. However, androgens are equally fundamental for maintaining metabolic, sexual, cognitive, and overall systemic health. Age‑related decline in androgen levels, as well as individual variability in androgen status, may influence treatment response and long‑term outcomes. Emerging research suggests that certain MHT regimens may differentially affect androgen profile, particularly sex hormone‑binding globulin (SHBG) and free/total testosterone levels. The aim of this study was to evaluate the potential effects of fixed‑dose oral MHT containing oestradiol/dydrogesterone (1/10 mg) on serum androgen‑related hormonal parameters in perimenopausal women, compared with transdermal oestradiol 0.1% plus oral dydrogesterone 10 mg or non‑hormonal management.1
MATERIALS AND METHODS
This was a prospective, multicentre, randomised, open, comparative study conducted across three parallel treatment groups. A total of 148 perimenopausal women were enrolled. Inclusion criteria were: perimenopausal state with amenorrhoea for at least 4 months, presence of climacteric symptoms, and absence of contraindications to MHT. Participants were randomised to one of the following: oral oestradiol/dydrogesterone, transdermal oestradiol plus oral dydrogesterone, or nonhormonal therapy (control group).
Serum hormonal parameters, including SHBG and total testosterone, were assessed at baseline and at 6‑ and 12‑month follow‑up visits. Statistical analysis included within‑group and between‑group comparisons with a significance threshold of p<0.05.
RESULTS
The analysis confirmed the relevance of considering androgen status in the selection of MHT. Absolute hyperandrogenism is relatively uncommon in peri‑ and postmenopausal women (prevalence ≤10%), whereas relative hyperandrogenaemia is more frequently encountered. SHBG measurement may serve as a practical approach to evaluating androgen status and predicting hormonal response to therapy.
Data from the multicentre trial demonstrated a statistically significant increase in SHBG levels in the group receiving fixed‑dose oral oestradiol/dydrogesterone. SHBG increased at 6 months (48.3 nmol/L) and further at 12 months (50.32 nmol/L) versus baseline (p<0.01; Figure 1). In contrast, the transdermal MHT group and nonhormonal control group showed no significant changes over time.
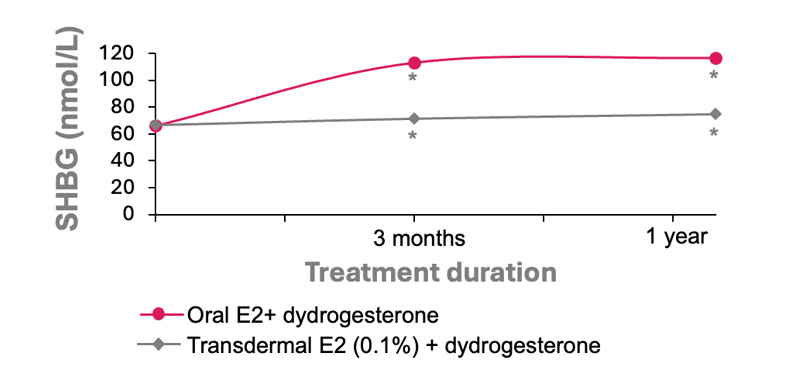
Figure 1: The dynamics of SHBG level in the oral MHT and transdermal MHT group.
*p<0.01.
E2: oestradiol; MHT: menopausal hormone therapy; SHBG: sex hormone‑binding globulin.
Despite the rise in SHBG, total testosterone levels remained stable in the oral MHT group throughout the 12‑month observation period (0.82 nmol/L versus 0.147 nmol/L, respectively; p=0.413), suggesting preservation of endogenous androgen levels. No other significant changes in hormonal profile were observed in any study arm.
These findings align with previous reports indicating that MHT regimens containing dydrogesterone do not induce a decline in testosterone levels, unlike some other progestogens.
CONCLUSION
Age‑related androgen decline may contribute to functional impairments affecting multiple systems. Therefore, selecting an MHT regimen that preserves androgen balance is of clinical importance. Dydrogesterone, a highly selective and biologically similar progestogen, demonstrated no suppressive effect on baseline androgen concentrations when used as part of oral MHT. These results support dydrogesterone‑containing fixed‑dose combinations as a rational therapeutic option for perimenopausal women, particularly when maintaining endogenous androgen levels is desirable.







