Meeting Summary
Chronic inflammatory skin diseases share common features, including substantial morbidity and significant impact on quality of life (QoL). There are also similarities in the pathological mechanisms of these diseases, involving keratinocytes and pro-inflammatory cytokines. While traditional immunosuppressive/immunomodulatory treatments provide rapid symptom relief for many patients, they are associated with adverse effects, variable efficacy, and cumulative toxicity over the long term. Therefore, there is a need for efficacious, well-tolerated treatment options for these diseases.
This article summarizes selected new data on the impact and management of chronic inflammatory skin diseases, reported at national and international conferences in 2025 and 2026. The review includes three posters, presenting new information on the efficacy and safety of tralokinumab in atopic dermatitis (AD) with involvement of the head, neck, or hands; one poster reporting early reductions in itch and pain in chronic hand eczema (CHE) from pooled clinical trials of delgocitinib cream; and posters presenting evidence to support the characterization of generalized pustular psoriasis (GPP) as a chronic disease requiring long-term treatment, and findings from a qualitative study of the patient journey in pyoderma gangrenosum (PG).
Introduction
Chronic inflammatory skin diseases represent a heterogeneous group of dermatoses commonly characterized by persistent and relapsing inflammation, potentially involving the whole skin.1,2 These diseases share common features, such as substantial morbidity and significant impact on QoL.2 Examples include AD, CHE, psoriasis (including GPP), and neutrophilic dermatoses, such as PG.3,4
Although the epidemiology of chronic inflammatory skin diseases varies according to disease type,1 they share overarching pathological mechanisms, being driven by keratinocyte-mediated dysregulated immune responses and pruritus.2,5 Keratinocytes are the major cutaneous source of IL-36 cytokines, which play a role in psoriasis and are associated with disease mechanisms in other chronic inflammatory diseases, including AD and neutrophilic dermatoses.3 In addition, activated keratinocytes produce thymic stromal lymphopoietin (TSLP), which promotes T-helper 2 cell differentiation, leading to the production of pro-inflammatory cytokines, including IL-4 and IL-13.6 In turn, IL-4 and IL-13 drive allergic inflammation through the JAK/signal transducer and activator of transcription (JAK-STAT) pathway, a key pathogenic mechanism in AD, CHE, and psoriasis.7,8
Traditional immunosuppressive and immunomodulatory agents have been the mainstay of therapy in chronic inflammatory skin diseases for decades, controlling inflammation and providing rapid symptom relief for many patients.2 However, these medications are associated with adverse effects due to their non-specific suppression of the immune system; their efficacy can be variable, and long-term therapy can lead to cumulative toxicity.2 Therefore, there has been a growing need for more targeted therapies with improved efficacy and tolerability.2
New data surrounding the impact and management of chronic inflammatory skin diseases were reported at international dermatology conferences in 2025 and 2026, and a selection of these data is summarized below.
Atopic Dermatitis
AD affects approximately 11.1% of children and adolescents, and 6.3% of adults worldwide.9 Characterized by dry, red, thickening, and weeping skin, the defining symptom of AD is itch, which can be severe.7 An estimated 84% of patients with AD have involvement of the head, neck, or hands, and both disease severity and impact on QoL are significantly worse in these patients compared with those without involvement of these regions.10,11
FDA approval of the first targeted biologic treatment for moderate-to severe AD, dupilumab, represented a significant advance in the management of the disease.2 Dupilumab blocks IL-4 and IL-13 signaling through the IL-4 receptor α, modulating the Th2-mediated immune response. It has been shown to significantly improve skin clearance and reduce pruritus in clinical trials and real-world data, with a safety profile superior to traditional systemic therapies.2,10,12 More recently, biologics that target IL-13 directly (tralokinumab and lebrikizumab) have been approved for use in adult patients with moderate-to-severe AD. Clinical trials have shown a significant decrease in disease and pruritus severity, as well as improvements in QoL.2
Three recent poster presentations provided updates on the efficacy and safety of tralokinumab in patients with moderate-to-severe AD with involvement of the head, neck, or hands.
Tralokinumab Compares Well with Dupilumab in Moderate-Severe AD with Hand Involvement
April Armstrong, Department of Dermatology, University of California Los Angeles, USA, presented data from an anchored matching-adjusted indirect comparison (MAIC) study conducted using data from two 16-week Phase III trials: ADHAND (tralokinumab 300 mg every 2 weeks) and LIBERTY-AD-HAFT (dupilumab 300 mg [200 mg in adolescents <60 kg] every 2 weeks). Individual patient data from ADHAND and aggregate published data from LIBERTY-AD-HAFT were used, with placebo as the common anchor. Patient data from ADHAND were weighted to match age, sex, race, and baseline Hand Eczema Area and Severity Index (HECSI) score in LIBERTY-AD-HAFT.13
LIBERTY-AD-HAFT included 67 patients in the dupilumab arm and 66 in the placebo arm. The matched ADHAND sample included 76 patients in the tralokinumab arm and 23 in the placebo arm. The mean age in the matched active treatment arms was 36 years, the mean HECSI score at baseline was 46, 67% of patients were female, and 79% were White.13
Treatment with tralokinumab resulted in significantly more patients achieving ≥90% reduction in HECSI score (HECSI-90) versus dupilumab at 16 weeks (hazard ratio: 17.5; p=0.04), indicating a deeper response. In addition, patients treated with tralokinumab had a significantly greater improvement in their ability to perform routine activities (not including work), compared with dupilumab (hazard ratio: 16; p=0.005). All other endpoints compared (HECSI-70, HECSI percent reduction, reduction in itch, ≥4-point improvement in itch, reduction in pain, and Investigator’s Global Assessment [IGA] score of 0/1) favored tralokinumab, but differences over dupilumab did not reach statistical significance.13
Tralokinumab is Effective and Well-Tolerated in Moderate-Severe AD with Hand Involvement
Benjamin Ehst, Oregon Medical Research Center, Portland, USA, presented data from the 32-week double-blind, placebo-controlled Phase III ADHAND study with a 16-week, open-label treatment period of tralokinumab monotherapy in adults with AD. The study included a wash-out period for topical corticosteroids (TCS) and other AD medication prior to study start. Inclusion criteria included moderate-to-severe atopic hand eczema (AHE), Hand Eczema Symptom Diary (HESD) itch score of ≥4, inadequate response to topical prescription medications within the past year, and AD involvement of at least one body location other than hands/wrists. Participants were randomized 2:1 to receive tralokinumab 300 mg or placebo every 2 weeks. The primary endpoint was an IGA-AHE score of 0 or 1 at Week 16.14
Tralokinumab met the primary and all secondary endpoints of the study at Week 16. IGA-AHE 0/1 (clear/almost clear skin) was achieved by 40% of patients in the tralokinumab arm, versus 10.6% of those in the placebo arm (Figure 1A). Response rates continued to improve through Week 32, with 57.9% of patients continuing tralokinumab achieving IGA-AHE 0 or 1. HECSI-50, HECSI-75, or HECSI-90 were achieved by a significantly greater proportion of patients for tralokinumab versus placebo at Week 16. HECSI-90 was achieved by 41.7% versus 10.9% of patients treated with tralokinumab versus placebo, respectively (treatment difference: 30.8%; p<0.0001; Figure 1B). Further improvements were observed in all HECSI outcomes through Week 32. At least 4-point reductions in HESD itch score were achieved by 47.3% versus 20.7% of patients in the tralokinumab versus placebo arms, respectively, and in HESD pain score by 45.3% versus 13.3% of patients, respectively (each p<0.001). Further improvements in HESD itch and pain were observed through Week 32. QoL measures also improved, with a mean change in Dermatology Quality of Life Index (DLQI) from baseline to Week 16 of -60.9% in the tralokinumab arm versus -21.0% in the placebo arm, and in Hand Eczema Impact Scale (HEIS) score of -64.9% versus -28.5%, respectively (each p<0.001).14
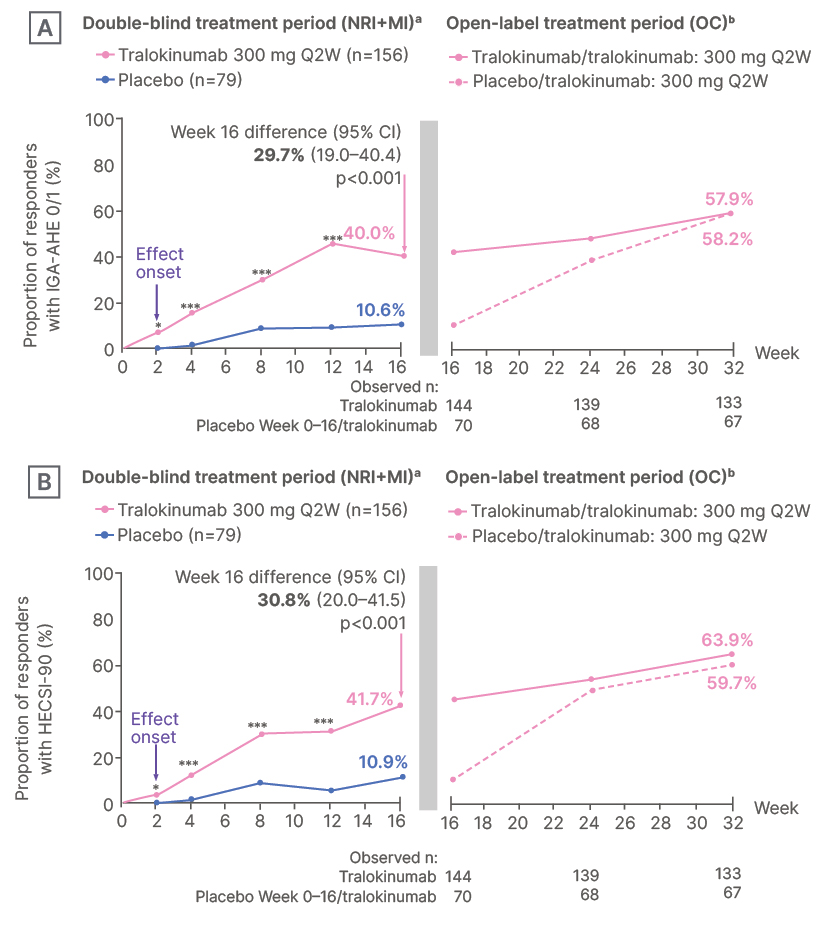
Figure 1: Proportion of responders with A) IGA-AHE 0/1 (clear/almost clear skin); and B) HECSI-90 through Week 32.14
*p≤0.01.
***p≤0.001.
aResults from a logistic regression model on the two cohorts of patients randomized before and after sample size
re-calculation are combined using a combination-test with equal weights. NRI was used for data after rescue, and data after discontinuation due to adverse events or lack of efficacy. Multiple imputation was used following other reasons for discontinuation, and for other missing data.
bData analyzed as observed cases.
HECSI: Hand Eczema Severity Index; IGA-AHE: Investigator’s Global Assessment for Atopic Hand Eczema; MI: multiple imputation; NRI: non-responder imputation; OC: observed cases; Q2W: every two weeks.
Overall, tralokinumab was well-tolerated. Adverse events were reported in 60.3% of patients treated with tralokinumab and 60.8% with placebo through Week 16, with serious adverse events reported in 1.9% and 1.3% of patients, respectively. The most frequently reported adverse events were nasopharyngitis, injection site reaction, AD, and COVID-19. Most events were mild or moderate in severity.14
The poster concluded that these results support tralokinumab as a treatment option for this high-burden, difficult-to-treat population of patients with AD.14
Tralokinumab Provides Long-Term Control of Head and Neck AD
ECZTEND was a 5-year open-label extension study of tralokinumab (300 mg every 2 weeks) with/without concomitant topical corticosteroids/calcineurin in participants ≥12 years with moderate-to-severe AD.15 Participants who completed one of multiple tralokinumab parent trials (ECZTRA 1–8 and the investigator-initiated study, TraSki) were included.15 An interim 3-year analysis of ECZTEND data demonstrated progressive and sustained improvements in head and neck (H&N) AD in participants treated for up to 1 year in the parent trials and up to 3 years in ECZTEND.16
Andreas Wollenberg, Department of Dermatology and Allergy, Augsburg University Hospital; Department of Dermatology and Allergy, Ludwig Maximilian University of Munich; Comprehensive Centre for Inflammation Medicine, University of Lübeck, Germany, presented end-of-treatment results from ECZTEND with up to 5 years of tralokinumab exposure (median 2.6 years) at Week 248. Of 1,647 participants enrolled, 68.4% completed treatment, 7.1% withdrew due to lack of efficacy, and 4.3% withdrew due to adverse events. The mean age of participants at parent trial baseline was 37 years, 57.7% were male, the median H&N Eczema Area and Severity Index (EASI) subscore (range: 0–7.2)16 was 2.8, and 87.5% of patients had an H&N EASI ≥1.17
Tralokinumab demonstrated sustained improvements in H&N AD with tralokinumab treatment for up to 5 years; median H&N EASI subscore was 0.4 at ECZTEND trial start (N=1,639) and 0.1 at treatment end (n=84). Improvements observed for H&N EASI subscores were comparable to those observed for other body regions. Among participants with H&N EASI ≥1 at parent trial baseline, the proportions of patients achieving EASI-75, EASI ≤1, EASI ≤2, and EASI 0 remained high during ECZTEND, reaching 84.4%, 89.6%, 97.4%, and 46.8% at end of treatment. The authors concluded that, together, these findings indicate that tralokinumab is an efficacious long-term option for patients with AD with H&N involvement.17
Chronic Hand Eczema
CHE is defined as hand eczema that persists for more than 3 months or recurs at least twice annually.18,19 A common inflammatory skin disease with a self-reported physician-diagnosed prevalence of 9.6%,20 CHE has a multifactorial pathophysiology, with several etiological and clinical subtypes.18 CHE can have a substantial impact on QoL. In the multinational CHECK survey, among respondents with moderate-to-severe CHE who were using TCS (n=312), 83% reported moderate-to-very severe itch, 63% reported moderate-to-very severe pain, and 59% reported moderate-to-very severe sleep disturbance in the past 24 hours, despite treatment.21 The RWEAL study, a multinational, retrospective online chart review, indicated that an estimated 31% of patients with moderate-to-severe CHE experience an inadequate response or contraindication to TCS, and 31% progress to phototherapy and or systemic therapy, underscoring the need for effective and well-tolerated treatment options for this difficult-to-treat disease.22
Delgocitinib cream is the first and only FDA-approved topical pan-JAK inhibitor for the treatment of adults with moderate-to-severe CHE in adults who have had an inadequate response to TCS, or for whom their use is not advisable.23 Approval was based on clinician-assessed efficacy data from the Phase III DELTA 1 and DELTA 2 studies, which enrolled adults with CHE with HESD itch scores ≥4;23 90.9% of patients also had baseline HESD pain scores of ≥4.24 Pooled data from the studies suggested delgocitinib cream (20 mg/g twice daily) efficacy was superior to cream vehicle, and was well-tolerated over 16 weeks.25
New data from the pooled DELTA 1 and 2 studies were reported at the recent Winter Clinical Hawaii Conference and are summarized here.
Delgocitinib Cream is Associated with Early Reductions in Itch and Pain in Adults with Moderate-to-Severe CHE
While the primary endpoint of the DELTA studies was achievement of a clinician-assessed IGA-CHE score of 0/1 (clear or almost clear),25 patient-reported measures of itch and pain, such as HESD subscores, can permit evaluation of treatment impact before changes in visible signs of CHE. Brian Kim, Kimberly and Eric J. Waldman Department of Dermatology, Icahn School of Medicine at Mount Sinai, New York City, USA, presented a pooled post-hoc analysis of the DELTA studies to evaluate the changes in HESD itch and HESD pain scores over time.26
A total of 638 participants in the delgocitinib cream arm and 321 in the cream vehicle arm were included in the analysis. Baseline demographics and disease characteristics were similar between arms. In adults with moderate-to-severe CHE, delgocitinib cream provided rapid, clinically meaningful improvements in patient-reported measures of both itch and pain.26
Measurable relief with delgocitinib cream was observed as early as Day 1 for itch; among patients who received delgocitinib cream, a ≥2-point reduction in HESD itch score was observed in 25.3% (95% CI: 22.1–28.8) at Day 1 (versus 19.1% [15.1–23.7] vehicle; p<0.05). A ≥2-point reduction in HESD itch was achieved by 50.5% of participants treated with delgocitinib cream, versus 34.4% with cream vehicle at Day 13 (p<0.001). More participants treated with delgocitinib had a ≥4-point reduction in HESD itch at Week 16 compared with vehicle (47.2% versus 21.5%, respectively; p<0.001; Figure 2A), and, of these participants, 58.7% versus 41.2% achieved HESD itch subscore of 0/1 (minimal to no itch), respectively.26
In terms of pain, measurable relief with delgocitinib cream was observed as early as Day 3; a ≥2-point reduction in HESD pain was observed in 33.8% (95% CI: 30.2–37.6) at Day 3 (versus 26.2% [21.6–31.3] vehicle; p<0.05). A ≥2-point reduction in HESD pain was achieved by 33.8% of participants treated with delgocitinib cream versus 26.2% with cream vehicle at Day 11 (p<0.01). More participants treated with delgocitinib had a ≥4-point reduction in HESD pain at Week 16 compared with vehicle (48.9% versus 25.2%, respectively; p<0.001; Figure 2B), and, of these participants, 65.4% versus 50.7% achieved HESD pain subscore of 0/1, respectively.26
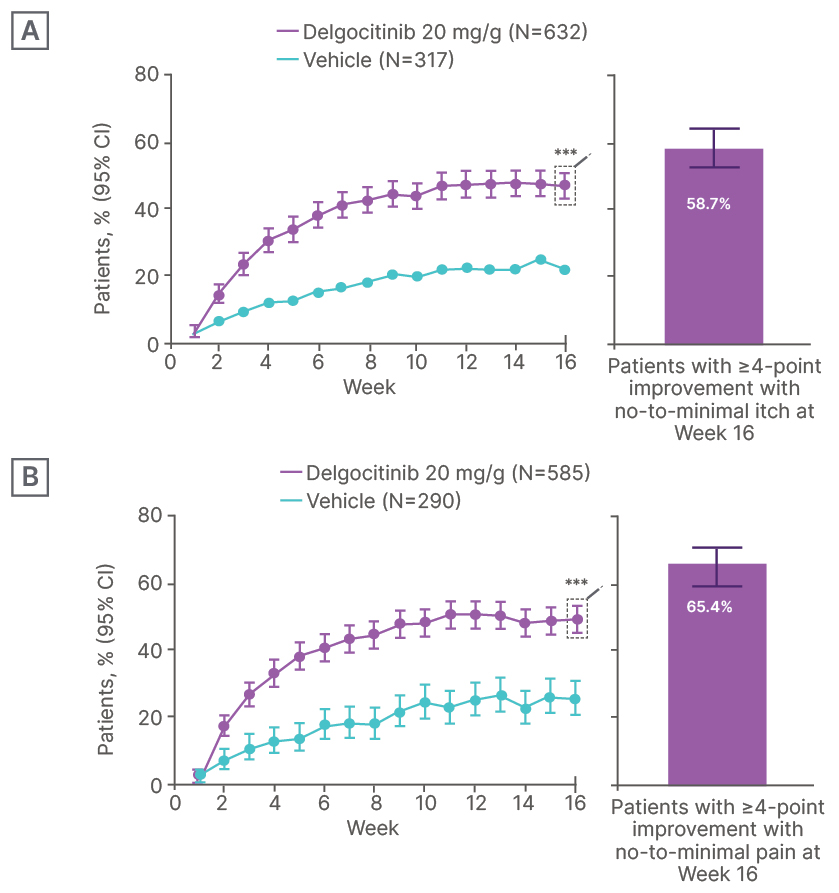
Figure 2: Patients with ≥4-point improvement in A) HESD itch and B) HESD pain (weekly average).26
***p≤0.01.
HESD: Hand Eczema Symptom Diary.
[/hl]Generalized Pustular Psoriasis[/hl]
GPP is a rare, potentially lethal, clinical variant of psoriasis characterized by persistent symptoms and recurrent flares of rapidly disseminating erythema, sterile pustules, scales, itching, and skin pain, often with systemic symptoms.3,27 GPP has a detrimental impact on QoL, whether patients are experiencing a flare or not, and patients regularly report anxiety and depression.28
Prior to the availability of spesolimab, treatment options for GPP were considered suboptimal and were associated with high costs, due to treatment cycling and frequent hospitalization of patients.28 Spesolimab is an anti-IL-36 receptor monoclonal antibody approved by the FDA in 2022,29 based on the Effisayil®2 (LEO Pharma AS, Ballerup, Denmark) trial,30 and is licensed for the treatment of GPP in adults and pediatric patients ≥12 years of age and ≥40 kg.29,30 It is administered intravenously to treat patients experiencing a flare and subcutaneously to those not experiencing a flare.29 Spesolimab is also being investigated for broader applications beyond GPP, including PG.31
GPP is a Chronic Disease Requiring Targeted, Long-Term Treatment
As a rare disease, there is a paucity of knowledge regarding GPP, with few clinical studies and management guidelines.32 To address this issue, a comprehensive literature search was conducted for English-language publications from 2015–2025 that addressed GPP in PubMed, Embase, and Google Scholar. A total of 126 references, of which 56 reported real-world evidence and 40 reported clinical trial data, were included in a narrative review. Joseph Merola, University of Texas Southwestern Medical Center, Dallas, USA, presented data from a subset of these references to describe the evidence supporting the characterization of GPP as a chronic disease requiring long-term management with targeted therapy.33
Evidence from both real-world and clinical trial data suggests that GPP is a chronic disease with ongoing symptoms and long-term patient burden. Key findings of the literature review included that, in patients with GPP:33
- Symptoms are persistent, extending beyond periods of flare.
- Pain, itching, pustules, and redness are common between flares.
- Non-flaring patients may have elevated expression of genes associated with inflammation.
- Flares are often long-lasting (1 week–3 months), recur frequently (1–4 times per year), and require prolonged hospitalization (12–75% of flaring patients are hospitalized, for an average duration of 4.9–47 days).
- Anxiety, depression, and chronic comorbidities are common.
- GPP can affect the ability to exercise, be intimate, wear shoes, socialize, and attend work/school, and may impact finances, sleep, and mobility.
- The average age of onset is 34–57 years, and many patients live with GPP for >10 years.
- Approximately two-thirds of patients (73%) have mutations in IL-36RN, the gene that encodes the IL-36 receptor.
Findings also highlighted that, prior to the availability of spesolimab, non-targeted biologic therapies and small molecules were used off-label to treat GPP with little success. Spesolimab was the first biologic therapy to target the IL-36 receptor, demonstrating rapid clearance of flares and sustained long-term improvement in chronic symptoms and QoL in participants with GPP in the EFFISAYIL® 2 study. Imsidolimab, another IL-36 monoclonal antibody, is currently in Phase III trials in GPP.33
The authors suggest the study supports the need for guidelines that recommend long-term treatment of chronic GPP with approved targeted therapy.33
Pyoderma Gangrenosum
PG is a rare, inflammatory, neutrophilic skin disorder characterized by painful, rapidly progressing ulcers.34,35 The chronic nature, unpredictable course, and associated pain of the disease, along with a lack of definitive treatment, can significantly impact patients’ physical, emotional, and psychosocial wellbeing.33 Due to the rarity of PG, data describing the emotional challenges faced by patients and their caregivers are limited.36
Real-World Emotional Impact and Unmet Needs in PG
Afsaneh Alavi, Department of Dermatology, Mayo Clinic, Rochester, Minnesota, USA, presented findings from a qualitative research study that explored the journey of patients with PG, with a focus on the emotional burden at multiple disease stages, and the critical need to provide more support to patients with PG.36
Adults with PG and their caregivers were recruited through healthcare professional (HCP) referrals, patient advocacy groups, and virtual panels on PG across the USA, China, France, and Japan. A total of 22 patients and four caregivers participated in the study, completing online tasks followed by an interview. Most participants were female (74%), aged 26–55 years (78%), and had self-reported moderate-to-severe PG.36
Key findings included:36
- PG initially presents as a ‘small bump’ that grows aggressively into an ulcer.
- PG ulcers cause excruciating pain, and the disease impacts daily routines, hobbies, clothing choices, ability to socialize, relationships, and ability to work.
- Patients also experience anxiety, depression, low self-esteem, and isolation.
- HCPs can struggle to diagnose and advise patients due to a lack of objective markers for diagnosis and information on the disease; patients are often misdiagnosed, and diagnosis can be delayed.
- Patients experience anxiety and fear related to the risk of relapse.
- Relapses are often unexpected and can be very distressing for patients.
Together, these findings emphasize the substantial emotional impact of PG on the study’s patients and caregivers and highlights their challenges throughout the patient journey. Addressing delayed diagnosis and misdiagnosis through improved disease education and offering mental health support are crucial to empowering patients and improving their outcomes, the authors concluded.36
Key Takeaways
Tralokinumab is an efficacious long-term option for AD with H&N involvement; clinical trial data demonstrate sustained improvements in patients ≥12 years of age over up to 5 years of treatment.16 Tralokinumab is also a potentially valuable treatment option for moderate-to-severe AD with hand involvement; clinical trial data show the agent is effective and well-tolerated in adults in this population, with early onset of efficacy observed after 2–3 weeks of treatment,14 and that tralokinumab may elicit a deeper response than dupilumab.13
In clinical trials, twice-daily delgocitinib cream provided rapid, clinically meaningful improvements in both patient-reported itch and pain in adults with moderate-to-severe CHE, as early as Day 1 and Day 3, respectively, supporting the efficacy of the non-steroidal topical cream in this population.25
GPP and PG are rare, chronic inflammatory skin diseases associated with a considerable long-term patient burden. There is a need for improved HCP education and guidelines to support HCPs in managing the emotional and clinical needs of patients with these conditions.33,36






