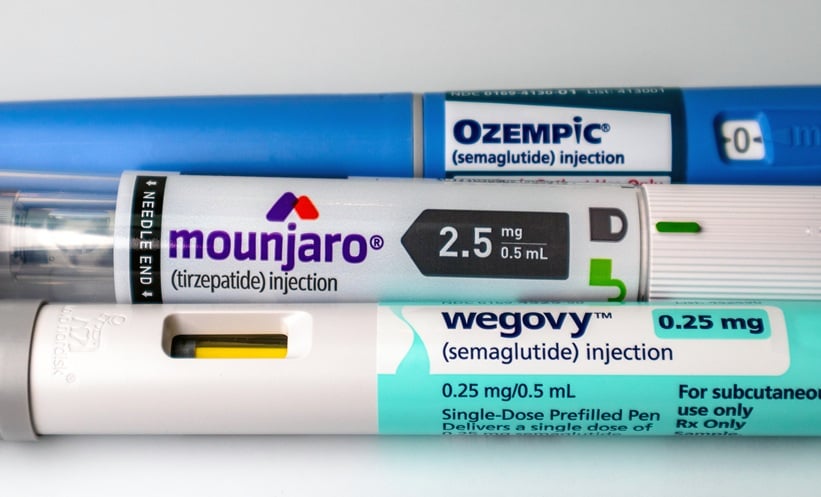
WARNING letters have been issued to 30 telehealth companies for making false or misleading claims about glucagon-like peptide-1 (GLP-1) products offered on their websites, the US Food and Drug Administration (FDA) announced on Tuesday.
The FDA has now sent more warning letters to pharmaceutical and telehealth firms to remove misleading advertisements in the past 6 months than over the entire preceding decade.
Primary violations identified in the letters included making claims implying sameness with FDA-approved weight loss drugs and obscuring product sourcing by marketing drugs under the telehealth firm’s name or trademark, without clearly stating who compounded them.
This is the second group of warning letters sent since a September crackdown on misleading direct-to-consumer pharmaceutical advertisements was launched following US President Donald Trump’s order for tighter oversight of how companies promote prescription drugs.
Marty Makary, FDA Commissioner, said: “It’s a new era.
“We are paying close attention to misleading claims being made by telehealth and pharma companies across all media platforms – and taking swift action.
“Compounded drugs can be important for overcoming shortages or meeting unique patient needs – but compounders should not try to compound drugs in a way that circumvents FDA’s approval process.”
Non-FDA-Approved GLP-1 Drugs
This follows last month’s announcement that the FDA plans to “take decisive steps” in restricting non-FDA-approved GLP-1 receptor agonists.
As a result, telehealth provider Hims & Hers pulled its new compounded semaglutide pill from the market just mere days after launch.
Novo Nordisk also took legal action against Hims & Hers, alleging that the telehealth provider was unlawfully mass marketing unapproved versions of Novo Nordisk’s FDA-approved semaglutide medicines (Wegovy and Ozempic).
The Danish drugmaker reported: “These knock-offs are putting patient health and wellbeing at risk.”
The FDA announced that the restriction of GLP-1 active pharmaceutical ingredients intended for use in non-FDA-approved compounded drugs is aimed at safeguarding consumers from drugs for which the FDA cannot verify quality, safety, or efficacy.
Featured image: K KStock on Adobe Stock