Meeting Summary
Zovegalisib (RLY-2608) is the first pan-mutant-selective PI3Kα inhibitor, binding a novel allosteric site distinct from approved non-mutant-selective orthosteric inhibitors. The first poster discusses efficacy and safety results from the first-in-human ReDiscover study, which evaluated zovegalisib plus fulvestrant in patients with PIK3CA-mutant, hormone receptor-positive/human epidermal growth factor receptor 2-negative (HR+/HER2-) advanced breast cancer following CDK4/6 inhibitor treatment. These findings provide the rationale for the registrational Phase III ReDiscover-2 study, and the second poster details its design, which compares zovegalisib plus fulvestrant versus capivasertib plus fulvestrant in patients with PIK3CA-mutated, HR+/HER2- locally advanced or metastatic breast cancer following progression on a CDK4/6 inhibitor.
Updated Efficacy of Mutant-Selective PI3Kα Inhibitor RLY-2608 in Combination with Fulvestrant in Patients with PIK3CA-Mutant HR+/HER2- Advanced Breast Cancer: ReDiscover Trial (Poster 65)
Sarah L. Sammons
PI3Kα pathway alterations are a major driver in HR+/HER2- breast cancer; approximately 40% of patients with HR+/HER2- breast cancer have PIK3CA mutations.1,2 Approved therapies are limited by toxicities associated with broad inhibition of the PI3K pathway, including hyperglycaemia, rash, diarrhoea, and stomatitis.3-9 Zovegalisib is the first oral, pan-mutant-selective PI3Kα inhibitor designed to overcome these limitations by selectively targeting mutant forms of PI3Kα and sparing the wild-type protein. Zovegalisib acts by binding to a novel allosteric site that is distinct from non-mutant-selective orthosteric PI3Kα inhibitors (i.e., alpelisib, inavolisib); the orthosteric site is conserved across wild-type and mutant forms of PI3Kα, while the allosteric site is selective for mutant forms of PI3Kα.10 In this poster, updated outcomes from the ReDiscover trial investigating zovegalisib plus fulvestrant were reported, which supported the development of a Phase III study comparing zovegalisib plus fulvestrant to the current standard of care.
ReDiscover is an ongoing, multi-arm, open-label, first-in-human study evaluating the efficacy and safety of zovegalisib plus fulvestrant in patients with PIK3CA-mutated advanced solid tumours. In the arm that enrolled patients with HR+/HER2- advanced breast cancer, eligibility required prior treatment with a CDK4/6 inhibitor, no prior exposure to PI3K, AKT, or mTOR inhibitors, and no more than one line of chemotherapy in the metastatic setting. Patients received zovegalisib (100–1,000 mg twice daily [BID] under fasting conditions) plus fulvestrant in 28-day cycles. Key objectives were investigator-assessed efficacy per Response Evaluation Criteria in Solid Tumours Version 1.1 (RECIST v1.1) and adverse events (AE) per Common Terminology Criteria for Adverse Events Version 5.0 (CTCAE v5.0).
As of 26th March 2025, 118 patients in this study arm were treated with zovegalisib plus fulvestrant, of whom 64 were treated with the recommended Phase II dose (RP2D) of zovegalisib 600 mg BID, fasted. Safety was assessed in all 118 zovegalisib-treated patients, and efficacy was assessed in 52 patients treated with zovegalisib at the RP2D without detectable PTEN or AKT E17K co-mutations. Approximately half the patients had a PIK3CA tumour mutation in the kinase domain, while the other half had a non-kinase domain mutation, reflecting the natural distribution of these variants. Patient demographics and baseline characteristics of those treated with zovegalisib 600 mg BID were consistent with a heavily pre-treated population, as all patients had received prior CDK4/6 inhibitors per protocol. A total of 43.8% had received two or more prior lines of therapy, 51.6% had prior fulvestrant or a prior oral selective oestrogen receptor degrader, 28.6% had tumours with ESR1 mutations, 28.1% had prior chemotherapy/antibody–drug conjugate, and 59.4% had visceral metastases.
At the 600 mg BID dose, median progression-free survival (mPFS) was 10.3 months (95% CI: 7.2–18.4), with a median follow-up time of 12.5 months (Figure 1). The 6-month and 9-month landmark progression-free survival (PFS) were 69.3% and 60%, respectively. Although cross-trial comparisons are inherently limited, the CAPItello-291 registrational trial reported an mPFS of 5.5 months in a post-CDK4/6 inhibitor population treated with capivasertib plus fulvestrant.7 As expected, the subgroup of patients treated in second line (N=30; mPFS: 11.0 months [95% CI: 7.3–22.0]; median follow-up: 12.0 months) showed longer mPFS than those treated in third line or later (N=22; mPFS: 9.2 months [95% CI: 5.4–not reached]; median follow-up: 14.1 months). Reductions in tumour dimensions were observed in 80.6% of patients with measurable disease, and 12 out of 31 patients achieved a confirmed objective response, for a confirmed objective response rate (ORR) of 38.7% (Figure 2). Tumour regression and objective responses were also observed among patients who received prior fulvestrant: six out of 15 patients achieved a confirmed objective response, for a confirmed ORR of 40.0%.
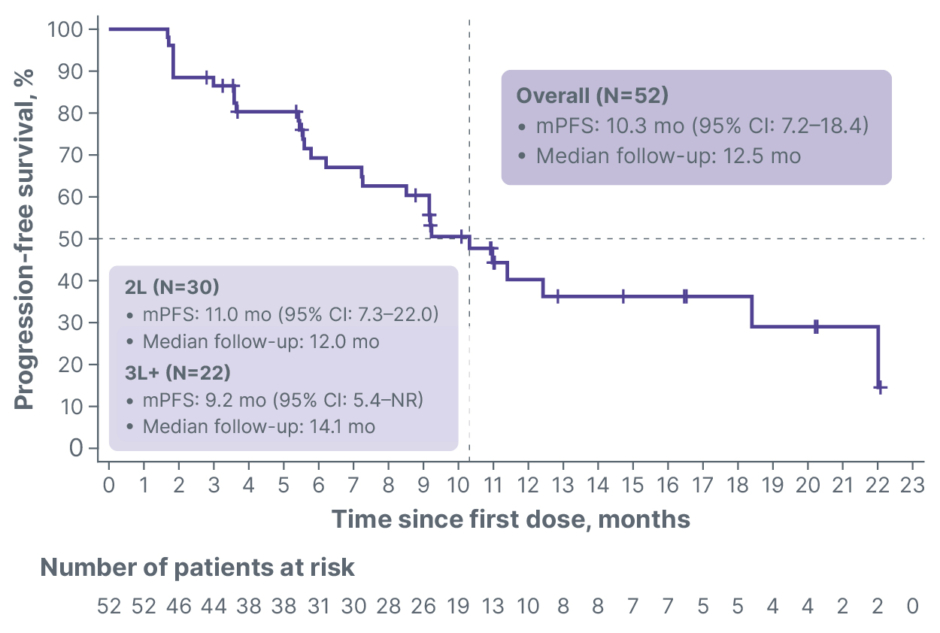
Figure 1: Overall median progression-free survival (N=52).
2L: second line; 3L+: third line or later; mPFS: median progression-free survival; mo: month; NR: not reached.
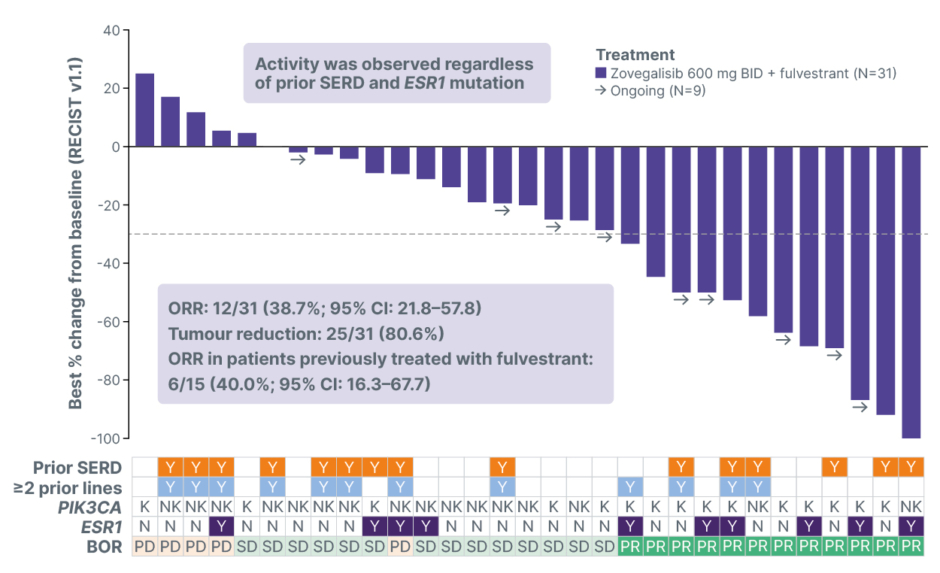
Figure 2: Radiographic tumour reduction and response per RECIST v1.1 (N=31).
Not shown: CR in patient with non-measurable disease.
BID: twice daily; BOR: best overall response; CR: complete response; K: kinase; N: no; NK: non-kinase; ORR: objective response rate; PD: progressive disease; PR: partial response; RECIST v1.1: Response Evaluation Criteria in Solid Tumours version 1.1; SD: stable disease; SERD: selective oestrogen receptor degrader; Y: yes.
Zovegalisib had a favourable safety profile consistent with mutant-selective PI3Ka inhibition. Treatment-related AEs (TRAE) were generally low-grade and reversible. The most common TRAEs were hyperglycaemia (44.1% any grade; 2.5% Grade 3), nausea (42.4%; 0.8%), fatigue (44.9%; 8.5%), creatinine increased (39.8%; 1.7%), and diarrhoea (33.1%; 1.7%). There were only two cases of Grade 3 hyperglycaemia with zovegalisib 600 mg BID, and most events were Grade 1, requiring no medical management. There were no Grade 4 or 5 TRAEs. The favourable overall tolerability profile was associated with the ability to maintain dose intensity, which is critical for driving long-term clinical benefit. The median dose intensity at the RP2D was 92%; dose interruptions, when they occurred, were brief, and only two out of 64 patients discontinued treatment due to TRAEs.
In conclusion, results from the ReDiscover study showed that zovegalisib plus fulvestrant demonstrated durable efficacy in patients with advanced HR+/HER2- breast cancer across PIK3CA genotypes without concurrent PTEN or AKT co-alterations, confirming potent pan-mutant targeting. Tumour reduction was observed in 80.6% of patients and ORR was 38.7% across PIK3CA genotypes. Encouraging PFS was observed in patients with PIK3CA-mutated HR+/HER2- advanced breast cancer previously treated with CDK4/6 inhibitors; mPFS was 10.3 months across mutation types and 11.0 months in patients receiving zovegalisib as second-line treatment. Zovegalisib was well tolerated, with limited class-related AEs. The promising efficacy and safety data observed in this first-in-human study suggest that zovegalisib has broad therapeutic potential in PIK3CA-mutant, HR+/HER2- advanced breast cancer.
ReDiscover-2, a Phase III Study of Zovegalisib (RLY-2608) + Fulvestrant Versus Capivasertib + Fulvestrant as Treatment for Locally Advanced or Metastatic PIK3CA-Mutant HR+/HER2- Breast Cancer Following Recurrence or Progression On or After Treatment with a CDK4/6 Inhibitor (Trial in Progress; Poster PS5-08-25)
Hope S. Rugo
The ReDiscover first-in-human study validated zovegalisib as the first oral, pan-mutant-selective PI3Kα-specific inhibitor for patients with HR+/HER2- advanced breast cancer previously treated with a CDK4/6 inhibitor and differentiated itself from existing PI3K pathway inhibitors. The study enrolled a heavily pre-treated patient population, with nearly half of patients having two or more lines of prior therapy in the metastatic setting and over half receiving prior selective oestrogen receptor degrader(s). Zovegalisib exhibited improved tolerability, reducing the class‑related AEs typically observed with non-mutant-selective PI3K pathway inhibitors, like capivasertib (and others). This led to a high observed mPFS of 10.3 months in patients with PIK3CA-mutated HR+/HER2- advanced breast cancer previously treated with a CDK4/6 inhibitor, and a mPFS of 11.0 months in patients who received zovegalisib as second-line treatment. Based on the encouraging results from ReDiscover, the global Phase III Re-Discover-2 trial was initiated in 2025. Further, the FDA has granted Breakthrough Therapy Designation to zovegalisib in combination with fulvestrant for the treatment of adults with PIK3CA-mutant HR+/HER2- locally advanced or metastatic breast cancer following recurrence or progression on or after treatment with a CDK4/6 inhibitor.
ReDiscover-2 is a global, multicentre, open-label, randomised Phase III study comparing the efficacy and safety of zovegalisib plus fulvestrant to capivasertib plus fulvestrant in adult patients with PIK3CA-mutated HR+/HER2- locally advanced or metastatic breast cancer (Figure 3). Approximately 540 patients will be enrolled and randomised 1:1 to receive zovegalisib (400 mg BID with food) plus standard-dose fulvestrant or capivasertib (400 mg BID, 4 days on and 3 days off, with or without food) plus standard-dose fulvestrant. Results from a separate food study demonstrated that zovegalisib 400 mg BID administered with food provided exposures similar to zovegalisib 600 mg BID dosed fasted (data on file). Randomisation will be stratified by PIK3CA mutation type (kinase versus non-kinase), visceral disease (yes versus no), and geographic region (Region 1 [USA, Canada, Western Europe, and Australia] versus Region 2 [Latin America and Eastern Europe] versus Region 3 [Asia]). The primary endpoint is PFS assessed by blinded independent central review in patients with PIK3CA kinase domain tumour mutations and in all patients. The key secondary endpoint is overall survival; secondary endpoints include safety and tolerability, ORR, duration of response, Clinical Benefit Rate (CBR), and quality of life. Study visits for assessments of safety (including AEs, vital signs, laboratory tests, and electrocardiograms), pharmacokinetics, and circulating tumour DNA will be conducted periodically throughout study treatment.
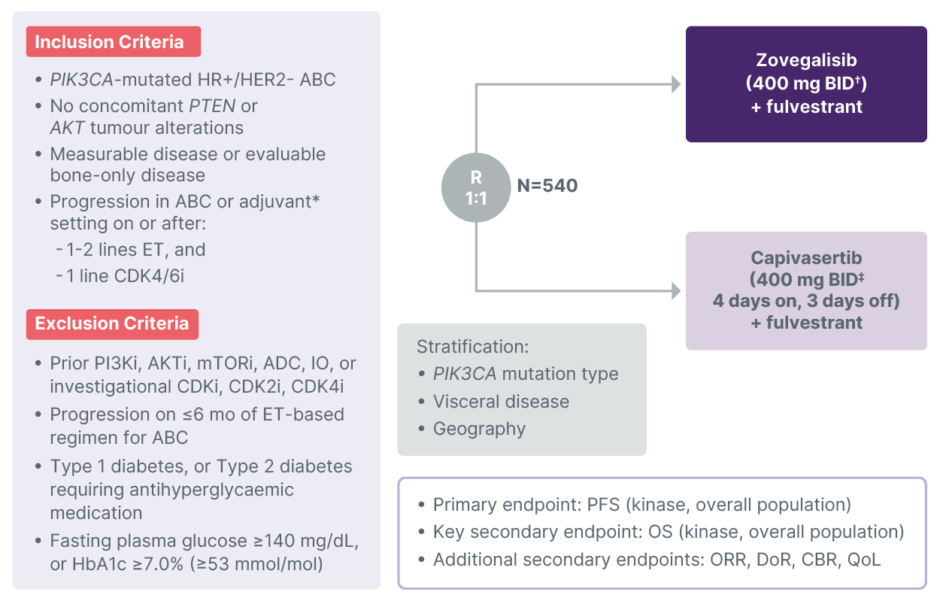
Figure 3: ReDiscover-2 (RLY-2608-102): Phase III registrational trial for post-CDK4/6 inhibitor HR+/HER2- advanced breast cancer with a PIK3CA mutation (NCT06982521).
*Disease progression during or within 12 months of completing adjuvant therapy.
?Zovegalisib administered with food.
‡Capivasertib administered with or without food.
ABC: advanced breast cancer; ADC: antibody–drug conjugate; BID: twice daily; CBR: Clinical Benefit Rate; DoR: duration of response; ET: endocrine therapy; HR+/HER2-: hormone receptor-positive/human epidermal growth factor receptor 2-negative; i: inhibitor; IO: immuno-oncology therapy; ORR: objective response rate; OS: overall survival; PFS: progression-free survival; QoL: quality of life; R: randomisation.
Key eligibility criteria include: ≥18 years of age with Eastern Cooperative Oncology Group (ECOG) performance status of 0–1; confirmed diagnosis of HR+/HER2- locally advanced or metastatic breast cancer with radiological or objective evidence of recurrence or progression; presence of one or more primary oncogenic PIK3CA mutations without evidence of AKT or PTEN alterations; measurable disease per RECIST v1.1 or evaluable bone-only disease; previous treatment for HR+/HER2- advanced breast cancer with at least one and no more than two lines of endocrine therapy (prior fulvestrant is allowed) and only one prior line of CDK4/6 inhibitor therapy; HbA1c <7.0% (<53 mmol/mol); and fasting plasma glucose <140 mg/dL. Patients with Type 1 diabetes, or Type 2 diabetes requiring antihyperglycaemic medication are excluded. Prior treatment with PI3K/AKT/mTOR pathway inhibitors or CDK‑targeting investigational agents is not allowed. ReDiscover-2 plans to open in >250 study centres globally; study enrolment began in July 2025 and recruitment is ongoing. For further information, contact: [email protected].
Summary
The first-in-human ReDiscover study of zovegalisib demonstrated encouraging antitumour activity across a range of PIK3CA genotypes and a favourable safety profile when given with fulvestrant in patients with PIK3CA-mutated, HR+/HER2- advanced breast cancer treated with a CDK4/6 inhibitor. The pivotal Phase III ReDiscover-2 study of zovegalisib plus fulvestrant versus capivasertib plus fulvestrant in patients with HR+/HER2- locally advanced or metastatic breast cancer with PIK3CA mutation may provide a new standard of care in this underserved patient population.