INTRODUCTION
Amyotrophic lateral sclerosis (ALS) is a progressive neurodegenerative disease that typically involves upper and lower motor neurons. It is clinically characterised by a progressive weakening of the muscles that starts either in the bulbar region or in the limbs and spreads, leading to death from respiratory failure in 3–5 years.
Lower urinary tract symptoms (LUTS) were previously thought to be infrequent features in ALS. When they occurred, they were largely attributed to reduced mobility since Onuf’s nucleus remains relatively spared in the disease. However, we and others have shown that LUTS are reported by around 40% of ALS patients and that urinary incontinence (UI) is found in ≤30% of them.1 Moreover, we have shown that these urinary symptoms are secondary to neurogenic bladder and therefore attributable to disease pathogenesis, although the underlying mechanisms remain unknown.
OBJECTIVES
Characterisation of LUTS among ALS phenotypes and their correlation with clinical characteristics may help us to understand the underlying pathophysiology. This could be important, since the different mechanisms leading to UI can have diverse impact on treatment and/or prognosis. This study aimed to describe differences in LUTS among the ALS phenotypes and their relationship with other clinical characteristics, including prognosis.
PATIENTS AND METHODS
Patients meeting criteria of classical ALS, progressive muscular atrophy (PMA), or primary lateral sclerosis (PLS) were recruited between May and November 2014 and followed-up until August 2016. Neurological and neuropsychological examinations were performed. Disability was assessed with the Revised ALS Functional Rating Scale (ALSFRS-R) score. Patients were categorised into bulbar or spinal according to the onset region. Muscle strength was measured using the Medical Research Council (MRC) rating scale. Executive and behavioural functions were also assessed.
Validated questionnaires of urinary symptoms (International Consultation on Incontinence Questionnaire – Short Form [ICIQ-SF], International Prostate Symptom Score [IPSS], and Overactive Bladder Awareness Tool [OAB-V8]) were self-administered. A complete pelvic and genital examination, dipstick test, prostate ultrasound, prostate-specific antigen levels (males), transabdominal ultrasonography, and urodynamic study were performed in clinically significant LUTS (csLUTS) patients who consented. Patients with csLUTS and abnormal urodynamic findings not related to pathology of the lower urinary tract were considered to have neurogenic bladder (NB).
Association between the different urinary symptoms scales, phenotype, and ALSFRS-R score was assessed using ordinal logistic regression models. Association of the clinical variables with presence of relevant urinary symptoms and NB was assessed using logistic regression models. We performed a multivariable survival analysis to study the effect of urinary symptoms in risk of death of the study population. We included, as covariates, other variables (age, bulbar onset, and phenotype) that affect survival in ALS patients. Death or tracheostomy were defined as endpoints.
RESULTS
Fifty-five ALS patients (37 classical ALS, 10 PMA, and 8 PLS) were included. Twenty-four patients reported csLUTS, and NB was demonstrated in 9 of them. LUTS were not influenced by age, phenotype, disability, cognitive or behavioural impairment, or disease progression, but female sex appeared to be a protective factor (odds ratio: 0.39, p=0.06). All NB patients were male and a subgroup, reporting LUTS early in the disease course, often showed other non-motor features and poor survival. Survival of csLUTS patients was similar to non-csLUTS (70.6 versus 62.9 months, p=0.69). More NB patients met the endpoint (death or tracheostomy) at last follow-up, and tracheostomyfree survival was shorter compared to patients without NB (70.6 versus 28.2 months respectively), although it was not statistically significant. We noticed that patients showing NB within 2 years from symptoms onset deteriorated rapidly and met the endpoint prematurely. In the multivariate survival analysis, age and urinary symptoms were the variables more strongly associated to survival, although statistical significance was not met (p=0.06 and p=0.08, respectively). See Figure 1 and Table 1.
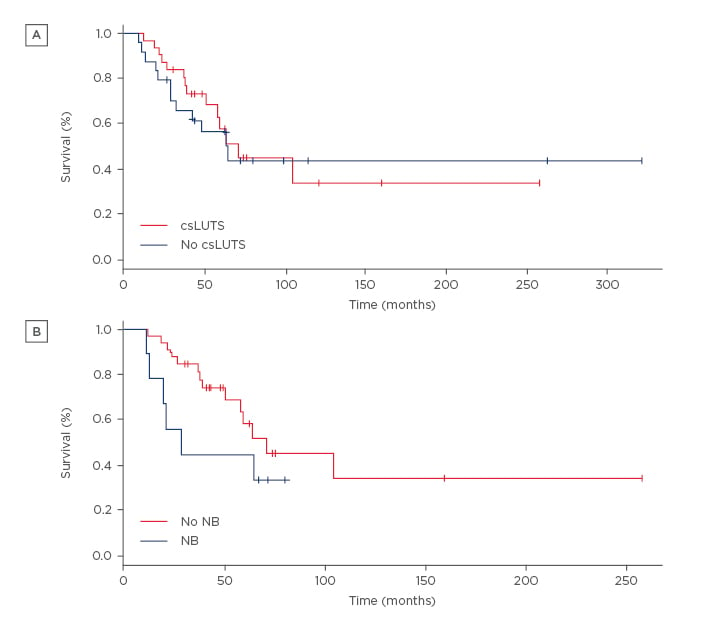
Figure 1: Cumulative survival of amyotrophic lateral sclerosis patients with or without clinically significant lower urinary tract symptoms (A) and with or without neurogenic bladder (B).
csLUTS: clinically significant lower urinary tract symptoms; NB: neurogenic bladder.
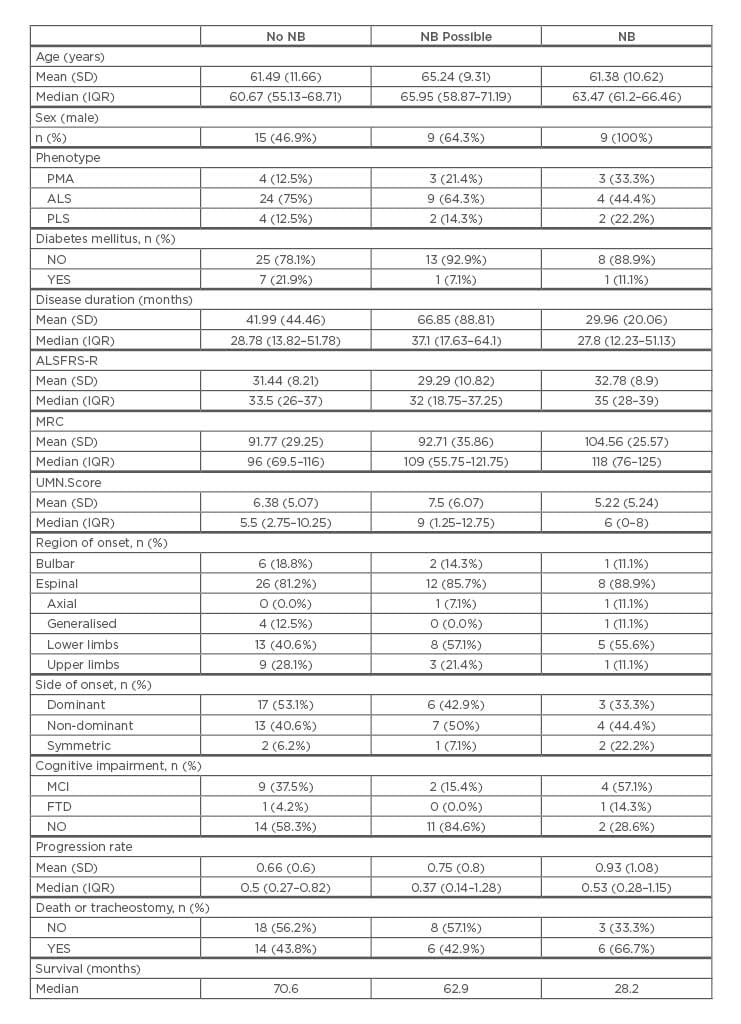
Table 1: Clinical characteristics of patients according to the presence or absence of neurogenic bladder.
NB: neurogenic bladder; SD: standard deviation; IQR: interquartile range; ALS: amyotrophic lateral
sclerosis; PMA: progressive muscular atrophy; PLS: primary lateral sclerosis; MCI: mild cognitive
impairment; FTD: frontotemporal dementia; ALSFRS-R: Revised ALS Functional Rating Scale; UMN:
upper motor neuron; MRC: Medical Research Council.
CONCLUSION
LUTS are more frequent in male ALS patients but are not related to age or clinical characteristics. When reported early, LUTS could be a sign of rapid disease spread and poor prognosis. Further prospective longitudinal and neuroimaging studies are warranted to confirm this hypothesis.








