Interview Summary
Hepatitis B virus (HBV) infection is a significant healthcare burden worldwide, and there are approximately 11 million people living with HBV in the WHO Western European region. Chronic hepatitis B (CHB) infection is associated with high rates of mortality due to cirrhosis and hepatocellular carcinoma (HCC). To help address this challenge and incorporate the latest developments in the field, in 2025, the European Association for the Study of the Liver (EASL) updated its guidelines for the diagnosis, treatment, and prevention of HBV infection. During interviews conducted by EMJ in January 2026, Maria Buti, Head of Hepatology and Internal Medicine at the Hospital General Universitari Vall d’Hebron, Barcelona, Spain; and Francesco Paolo Russo, Associate Professor of Gastroenterology at Padova University Hospital, Italy, highlighted the key changes in the new EASL guidelines and considered their implementation and impact. The move to cite functional cure as the ultimate goal of therapy, and to recommend quantitative hepatitis B surface antigen (qHBsAg) biomarker testing to aid in management decisions were viewed as significant updates. In addition, expansion of the nucleos(t)ide analogues (NA) treatment criteria, facilitating earlier and broader patient access to treatment, was seen as influential for patients, physicians, and healthcare policymakers alike, alongside an increased focus on shared decision making. The experts concluded by emphasising the importance of effective communication and education around these changes (across different countries and settings) to enable successful integration into patient-centred treatment pathways and improve the lives of people living with CHB.
INTRODUCTION: EASL GUIDELINE UPDATE FOR CHRONIC HBV
The treatment approach for CHB is evolving, with the WHO setting targets to eliminate HBV as a global public health concern by 2030.1 This promotes simplified and expanded treatment criteria to address the high mortality (and morbidity) rates associated with chronic infection.1,2 It also aims to reduce barriers to care, aligning with patient preferences for simpler, more accessible treatment.2 Another key area of discussion is the introduction of functional cure as the ultimate treatment goal beyond suppression of HBV DNA. While most patients will expect to receive lifelong therapy for CHB infection, this shift in thought to consider functional cure as an achievable outcome for future treatment strategies could be transformative for both patients and healthcare systems worldwide.3 For patients, it would not only improve quality of life from the symptoms and treatment perspective, but also in terms of the social and personal stigma that is prevalent in people living with CHB.3 However, despite these goals, in Western Europe in 2022, only 34% of people with HBV (HBsAg-positive population) were diagnosed, and just 31% of eligible patients received treatment, leaving many undiagnosed, untreated, and at elevated risk of severe liver complications.4
Against this background, and in light of continued developments in diagnostic and treatment strategies, the EASL released their updated guidelines on the management of HBV infection during their annual congress in May 2025. This included expanding the goals of therapy, extending treatment criteria, and looking to the use of biomarkers in screening and monitoring disease, promoting linkage to care. During a focused discussion, experts Maria Buti and Francesco Paolo Russo gave their informed opinions on the latest EASL guidelines and considered the implementation and impact of the recommended changes in the management of CHB.
CHANGES TO THE EASL GUIDELINES
Buti and Russo began by outlining what they viewed as the key updates in the 2025 EASL guidelines.5 “Overall, I would say that the biggest change in the new EASL guidelines is a shift to a more practical, patient-centred approach,” said Buti: “The guidelines move away from relying only on [the testing of] alanine aminotransferase (ALT) and HBV DNA levels [to direct treatment] and place much more emphasis on individual risk and biomarkers. An example is qHBsAg testing, which now has a clear role, helping to better understand immune control and to [guide] the selection of patients for different treatment decisions.” For example, Buti highlighted that the EASL guidelines now state that treatment with NAs should be informed by HBsAg levels and, crucially, that NAs may be stopped in patients with confirmed HBsAg loss.5 The EASL guidelines clearly cite HBsAg-loss (with the opportunity to stop NA treatment) as the ultimate goal of antiviral therapy for CHB and as a key component of functional cure.5
The experts agreed that another important change is related to the initiation of NA therapy. Russo explained that the EASL guidelines now recommend qHBsAg testing for all individuals with suspected, or who are at risk of, HBV infection.5 In turn, all HBsAg-positive individuals with detectable HBV DNA are now (in principle) candidates for antiviral NA therapy.5 Russo underlined that this recommendation also extended treatment to people who, due to accessibility issues in different countries and settings, cannot undergo the repeated blood tests necessary to monitor HBV DNA. Buti agreed: “Probably the most important change is in starting treatment. The criteria are now more proactive, and more patients can be treated. Overall, it’s less ‘wait and see’, with the guidelines more in favour of early risk stratification, timely treatment, and looking at functional cure as a main goal.”
Supplementing these recommendations, the 2025 EASL guidelines also advocate a shared decision-making approach in treatment planning for CHB, which Russo viewed as vital: “Shared decision-making is something that we consider essential. We encourage collaboration between clinicians and patients. This is an open discussion about the benefits and limitations of therapy that is fundamental for addressing potential concerns, building trust, and promoting informed decision-making, which can improve adherence and overall outcomes.”
VALUE OF FUNCTIONAL CURE
The 2025 EASL guidelines describe functional cure as the ultimate treatment goal in CHB, beyond the primary goal of suppression of HBV DNA.5 Functional cure is defined as undetectable HBV DNA and undetectable HBsAg that continues at 24 weeks following cessation of all treatment, with or without hepatitis B surface antibodies.5 Drilling down further into the impact of this guideline update, the experts examined the value of functional cure of CHB to different stakeholders.
From a patient perspective, Buti clarified: “With current treatments, functional cure, defined as HBsAg loss, is achieved in very, very few patients. So, the majority remain on NAs, which means lifelong therapy.” However, the experts emphasised that, although rare, having a functional cure with HBsAg loss cited as the ultimate goal of therapy is still a significant change for patients. Russo observed: “It is very important, and will change the management of those patients who [have the chance of reaching] a functional cure.” Buti added: “We can tell our patients about the long-term clinical benefits [of functional cure], a reduced risk of cirrhosis and HCC,6 and I think the reduced risk of HCC, in particular, is really important for patients. It would also mean no more lifelong therapy, so less daily stress with medication, less stigma, and improved quality of life.” Russo concurred: “We can explain to patients that functional cure is associated with the best prognosis. This helps a lot because viral hepatitis still carries a stigma for patients. Sometimes they don’t want to go to the specialist or general practitioner because they know that they are infected with viral hepatitis. So, reaching functional cure really would be the best way to help the patient.”
Considering the physician’s view, Buti commented: “For the first time, we, as physicians, can give our patients a clear endpoint, and the possibility of a finite treatment duration. Up to now, we have told our patients that there is suppression of viral replication, that the virus is under control, but that it’s important to continue treatment. Now, we can tell our patients that there is the option of a cure. This is an important game changer.” Russo agreed, adding: “Never before have we had the possibility of telling patients that a functional cure could be the final outcome, which might get a more positive response from the patients themselves. Also, for those patients with very mild disease, the prospect of a functional cure means we could even consider discharging them from the specialist outpatient clinic at some point and returning them to their general practitioner. Of course, we would have to be sure that there is no advanced liver disease in these patients, but this could really change the way we manage patients with HBV in the future.”
From the policymaker perspective, Buti observed: “Healthcare systems and policymakers always like a finite treatment duration, and a functional cure means that we can stop treatment, and reduce long-term health costs, with less monitoring and lower risk of [medication-related] complications. So, achieving functional cure is in line with the WHO [HBV] elimination goals.1,7 Clearly, the possibility of a functional cure means using resources more efficiently, and this is always extremely important for healthcare systems and policymakers.” Russo agreed: “We will get fewer [medication-related] complications, fewer inpatients, and fewer complications of advanced liver disease, which, of course, is something that the policymakers will also be very happy to accept.”
ROLE AND ADOPTION OF QUANTITATIVE HBSAg TESTING
Considering the newly expanded role of qHBSAg testing in the updated EASL guidelines, with the additional insights afforded by quantitative testing (as opposed to qualitative, yes/no outcomes). Buti commented, “qHBsAg testing is becoming a key tool in the management of patients with CHB. First, it helps us understand which patients are at higher risk of disease progression; the higher the levels, the higher the likelihood of developing complications, particularly HCC.5,6 Secondly, it is helpful in monitoring treatment response. Initially, it was used [as an indicator] for stopping PEG-IFNα, but now we know that low levels of HBsAg can help to identify patients treated with NAs who could achieve a functional cure, which is the goal of treatment. So qHBsAg testing has these two important roles: identifying patients with a high risk of disease progression, and monitoring treatment response. In the future, it will also be very useful in the selection of patients for new treatments.”
Despite the guideline recommendations and clear value of qHBsAg testing for disease management, there are barriers to its widespread adoption, with test availability varying between countries and individual laboratories. Russo noted that the heterogeneity in the application and availability of qHBsAg testing is limiting the use of HBsAg as a clinical marker and suggested that laboratory and clinical professionals should be educated about the important role that HBsAg quantification can play in deciding when to start and stop treatment for CHB. Buti added: “Many clinicians know about [qHBsAg], but they don’t integrate it into their routine in clinical practice. So, I think it’s important to increase awareness, educate physicians on its clinical value, and incorporate this biomarker into standard care pathways. Also, in some countries where there is no public health system, reimbursement can be an issue. It is always very important that the health systems, particularly the insurance companies, cover the [cost of] the test. For me, these are the two main barriers to adoption of qHBsAg testing: awareness and accessibility.”
Illustrating these issues in practice, the experts moved on to consider findings from a recent survey that assessed the implementation and awareness of qHBsAg testing in 91 clinical microbiology laboratories across Spain.8 Only 37% of these laboratories reported performing the qHBsAg test, and, of those who did not test, 86% did not refer their samples to external laboratories, with 46% citing lack of clinical demand.8 Shortages of equipment and resources were reported as other barriers to the widespread adoption of qHBsAg testing in Spain.8 Reviewing these findings, the experts noted that qHBsAg testing was available in the majority of academic hospitals, but was less common in other centres. However, according to Buti, the perception of low clinical demand is potentially a greater barrier than availability: “For me, what is really important is not if the test is available or not, it is if the clinicians are aware of the importance of qHBsAg testing, and that they push the laboratory to provide the test. So, while I think there is still room to improve the introduction of qHBsAg testing, it’s crucial that clinicians understand the value of this test in guiding treatment and monitoring for functional cure.” Russo agreed, “[the survey findings] confirmed that, unfortunately, there is heterogeneity in laboratory testing. In Italy, it is very similar to Spain. We have to teach those specialists who are not aware of the importance of HBsAg quantification why they should push their microbiology laboratory to have this test.”
THE IMPACT OF EXPANDING NA TREATMENT INITIATION CRITERIA
As outlined above, the 2025 EASL guidelines have expanded the eligibility criteria for NA treatment, and the addition of qHBsAg thresholds, along with individual risk factors, has widened the pool of patients available to treat.5 NAs work by preventing viral replication, so reducing the level of viral DNA in the blood, and early therapy may prevent complications from ongoing viral replication, such as cirrhosis.5 Buti commented: “I think the impact of expanding the NA treatment initiation criteria is particularly important because it allows us to treat patients earlier, before significant liver damage. Up to now, the tendency has been to treat patients when they have significant fibrosis, and with significant fibrosis comes a risk of developing HCC. So, if we treat patients with low or minimal liver damage earlier, we can better prevent these complications. We can also give more patients an opportunity to achieve functional cure and treat patients who have comorbidities that are associated with a higher risk of disease progression. I think expanding the NA treatment initiation criteria is extremely beneficial.” Russo added that the expanded treatment criteria would also facilitate treatment and optimise costs in countries where it is not feasible to manage patients through regular blood tests for HBV, adding that this will also support NA initiation in patients who already have CHB: “The WHO guidelines5 already suggest treating most patients, as do the Chinese guidelines.9 Some papers suggest that starting NAs in patients with chronic infection doesn’t give them the best outcome in the long term.10 However, multiple longitudinal and modelling studies demonstrate that CHB confers a continuous lifetime risk of cirrhosis and HCC, even below current treatment thresholds, supporting simplified and expanded treatment strategies.2,11 So, it’s something that is often discussed, but if [expanding NA treatment initiation criteria] can help in reaching functional cure, then I think we should consider this possibility.”
AMENDING THE PATIENT PATHWAY
As illustrated by the Spanish survey on qHBsAg testing (described above),8 issuing guideline recommendations is only one step towards evolving clinical practice. For real-world improvements, it is essential that these recommendations are integrated into standard care pathways and diagnostic workflows worldwide. The experts considered what changes may be required in the patient pathway to follow through with guideline implementation. Buti observed: “Those of us [in large academic centres] already use these biomarkers, qHBsAg, and the NA treatment criteria, and we also look to functional cure. However, it’s important to integrate these changes into the [standard] patient pathway, particularly for centres that are not treating large numbers of patients. For example, incorporating qHBsAg testing at baseline may seem like more testing, but, in the majority of patients, it can be done without an extra serum sample.” Russo concurred: “Of course, we will need implementation, mainly in those smaller centres where [they don’t currently use] qHBsAg testing, but, in our practice, the guidelines and how to follow-up patients have already been much discussed. Some small changes will be needed. For example, we suggest checking HBsAg at least every 12 months, and, in the hub centres, we can check it even more frequently. The clinical role of the specialist in the management of patients with CHB will also change.” Buti added: “Re-evaluating treatment initiation is also really important, but this needs education, particularly of those physicians who are not currently treating, or who are reluctant to start treatment until the patient has advanced or significant fibrosis. So, I think these changes need to be associated with more education.”
Buti continued to consider another, earlier step in the patient pathway: “We have talked a lot about increasing treatment, expansion of NA criteria, qHBsAg testing, and how it’s useful to better manage our patients, but we haven’t touched on screening. I think screening for HBV is really important, because the majority of our patients are completely asymptomatic.” Russo agreed: “Viral hepatitis, and chronic liver disease in general, are very difficult to diagnose in the early phase, so screening, and of course [identifying] the target population to be screened, is a main goal that we need to reach.” “If we don’t test, we cannot treat,” said Buti, “and I think the approach that has been used a lot for hepatitis C now has to move to CHB. That means test and treat.”
EDUCATION AND COMMUNICATION
Throughout the interview, Buti and Russo emphasised the importance of education to ensure implementation of the key EASL guideline changes, including communicating the value of functional cure. Summarising their thoughts on this, Buti stated: “For me, education is critical, but not only the education of physicians. We also need to engage patients and educate the healthcare system and policymakers. So, we need a three-level approach. We can educate physicians in meetings, in workshops, in webinars, looking for active participation. This is relatively easy to achieve, but we also need to explain the new guidelines, the importance of functional cure, and how functional cure can reduce the risk of negative clinical outcomes to patients. In Spain, we have several patient organisations that have been very active for hepatitis C and could also be influential for HBV. In Catalonia, they are very active in promoting ‘Linkage to Care’ (the bridge between testing and treatment, care and support). HBV is also frequent in migrants/the non-Spanish population here, and patients overall need to understand the value of testing, early diagnosis, and functional cure. Then, finally, policymakers also have an important role in this three-level approach. The healthcare system has to cover the cost [of implementing these latest changes], particularly the qHBsAg testing, so they need to know its value in terms of monitoring and in selecting patients for treatment.” Russo supported this view, adding that: “In the future, there may be new treatments that can help reach functional cure, which, [together with] qHBsAg biomarker testing for deciding who should be treated and when that treatment has a good outcome, will be a real game changer […] and, as mentioned, this will help on three levels: with patients, with physicians, and with policymakers.”
In summary, Buti and Russo highlighted that broadening the NA treatment criteria, use of qHBsAg testing, and introduction of functional cure as the ultimate treatment goal are important and influential changes in the care of patients with CHB, introduced in the updated EASL guidelines 2025. However, implementation of these changes in practice will require education of patients, physicians, and healthcare policymakers, in order to translate guidance into meaningful improvements in clinical outcome and quality of life for patients with CHB (Figure 1).
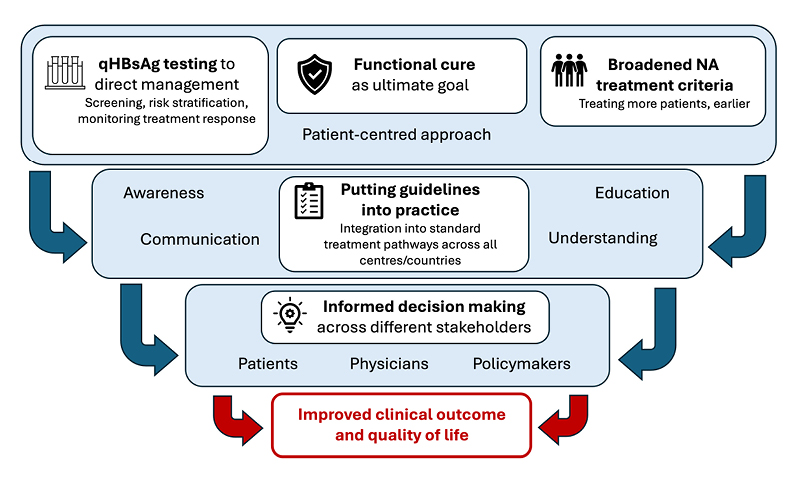
Figure 1: European Association for the Study of the Liver (EASL) 2025 guideline updates: key discussion points.
NA: nucleos(t)ide analogue; qHBsAg: quantitative hepatitis B surface antigen.
NX-GBL-HBU-ABST-260001 | DOP March 2026