BACKGROUND AND AIMS
Enfortumab vedotin plus pembrolizumab (EV+P) is the preferred first-line (1L) regimen for patients with advanced urothelial carcinoma (aUC).1,2 Despite the robust efficacy of EV+P demonstrated in the landmark EV-302 clinical trial, 10% of patients experienced primary progression (PD) on 1L EV+P, highlighting the need to identify those at risk for early treatment failure.2 The authors hypothesised that the presence of mixed variant-predominant or pure variant histology (VH) in conjunction with previously described poor prognostic factors in aUC, such as Eastern Cooperative Oncology Group Performance Status (ECOG PS) ≥1, presence of liver metastases, haemoglobin (Hgb) level <10 g/dL, and elevated neutrophil-to-lymphocyte ratio (NLR), would be associated with inferior outcomes, including early PD.3-6
MATERIALS AND METHODS
UNITE is a multi-site retrospective study across 17 sites in the USA of patients with aUC treated with targeted agents, including EV. Patients who received at least 1 dose of 1L EV+P were included. Observed response rate (ORR) was assessed in patients with post-baseline imaging. Progression-free (PFS) and overall survival (OS) were calculated from EV+P start to PD or death. Early PD was defined as radiographic or clinical PD occurring within ≤12 weeks from EV+P start. Baseline clinicopathologic factors, somatic alterations by next-generation sequencing (NGS), and human epidermal growth factor receptor 2 (HER2) immunohistochemistry (IHC) score were assessed, and in an exploratory analysis, compared between patients with and without early PD using Fisher’s exact and Mann–Whitney U tests. In the overall population, associations between a priori selected baseline factors and outcomes were evaluated using univariable (UVA) and multivariable (MVA) logistic and Cox regression models.
RESULTS
Among 387 patients treated with 1L EV+P, 52 experienced early PD. NGS was available in 238 patients (32 with early PD) and HER2 IHC score was available in 83 patients (eight with early PD). At EV+P start, median age was 73 years (range: 40–99); 73% of patients were male; 81% of patients were White; 77% had ECOG PS 0–1; 74% had lower tract primary tumour; and 16% had liver metastases. Histology included pure UC in 65% of patients, mixed UC-predominant in 22% of patients, and VH in 12% of patients. Median follow-up was 12.2 months (95% CI: 11.2–15.0). ORR was 57.4% in 339 evaluable patients.
Early PD occurred in 13.4% of patients. Median PFS and OS were 12.2 months (95% CI: 8.8–17.0) and 22.8 months (95% CI: 18.6–56.2), respectively. VH, ECOG PS ≥2, lower Hgb, and higher NLR were associated with early PD (all p<0.05). Somatic alterations by NGS and HER2 IHC score were not associated with outcomes including early PD. On UVA, ECOG PS ≥2 and NLR ≥5 were associated with lower ORR and shorter PFS and OS, while VH, presence of visceral or liver metastases, and Hgb <10 g/dL were associated with shorter PFS and OS (all p<0.05). On MVA (Table 1), ECOG PS ≥2 and NLR ≥5 were independently associated with lower ORR; VH with shorter PFS; and ECOG PS ≥2, Hgb <10 g/dL, and NLR ≥5 with shorter PFS and OS.
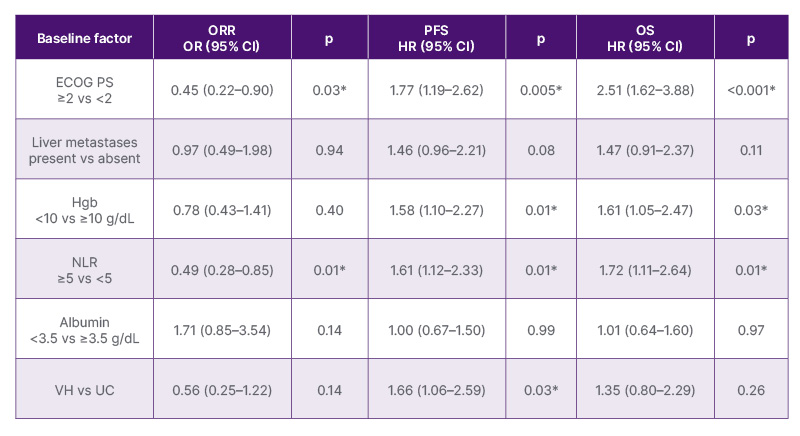
Table 1: Multivariable analysis of a priori selected baseline factors and outcomes in patients with advanced urothelial carcinoma treated with first-line enfortumab vedotin plus pembrolizumab.
*Statistically significant (p<0.05).
ECOG PS: Eastern Cooperative Oncology Group Performance Status; Hgb: haemoglobin; HR: hazard ratio; NLR: neutrophil-to-lymphocyte ratio; OR: odds ratio; ORR: observed response rate; OS: overall survival; PFS: progression-free survival; UC: urothelial carcinoma; VH: mixed variant-predominant or pure variant histology; vs: versus.
CONCLUSION
Inferior outcomes, including early PD, on 1L EV+P occurred more frequently in patients with VH and adverse prognostic features previously identified in platinum- and immune checkpoint inhibitor-treated cohorts. The persistence of these prognostic factors across therapeutic classes suggests that poor outcomes in aUC are largely driven by treatment-agnostic, tumour-intrinsic biology. Integration of circulating tumour DNA, histologic, and molecular correlates may refine future risk stratification models.







