BACKGROUND AND AIMS
Asthma exacerbations (AE) are defined as acute or subacute worsening of symptoms from the patient’s usual status and are responsible for most of the morbidity in patients with asthma.1 Allergens and especially viral-associated respiratory tract infections (RTI) are common triggers for AE. Viral RTI account for approximately 40–60% of all AE.2 Animal studies suggest that during viral-associated AE, an increase of so-called alarmins in the circulation can be found, namely IL-33, thymic stromal lymphopoietin, and IL-25. In humans, an increase of serum cytokines, such as IL-10 and IL-18, and a decrease of serum periostin has been described during AE.3,4 Immunological mechanisms underlying AE have not yet been completely unravelled. In this study, the authors aimed to gain insight into the immunological dynamics of moderate-to-severe exacerbations.
METHODS
Patients from the Breathe study, a double-blind, randomised-controlled trial to determine the effects of a bacterial lysate in patients with Global Initiative for Asthma (GINA) 4 asthma and recurrent AE, were enrolled in the study. Patients were randomised to either 2-year winter season bacterial lysate or placebo treatment. Study visits took place every 3 months, and an extra study visit was planned promptly at the start of an AE. Every 6 months and at the start of during-exacerbation visits, blood was drawn. T2 phenotype was based on T2 inflammation, determined by GINA 2020.5 Nasopharyngeal swab Amies medium was analysed with multiplex viral PCR, blood leukocyte differentiation was calculated with DxH, plasma cytokines were measured with ELISA, and flow cytometry was used to analyse blood T-cell subsets and intracellular cytokines. Baseline and exacerbation data were compared. Data are shown in median (interquartile range).
RESULTS
Thirty-six patients were included in this study. Asthma Control Questionnaire (ACQ) score increased during an AE. In 43.0% of the exacerbations a nasopharyngeal virus was detected, and pathogenic bacteria were detected in 16.8% of the exacerbations. Blood neutrophils did not alter between AE and baseline, while numbers of eosinophils tended to decrease (4.31 [1.39–6.32] versus 1.35 [0.42–2.88] x103/mL; p=0.09). Plasma cytokine analysis showed an increase of IL-10 during an AE (1.95 [1.95–8.85] versus 5.61 (1.95–14.33) pg/mL; p=0.01). Preliminary T-cell subset analysis (n=22) showed a decrease in Th2 and Th17 cell numbers (Figure 1). Intracellular CD4+ T-cell cytokine analysis revealed an increase of percentages of IL-5 (0.83 [0.60–1.32] versus 1.91 [1.51–4.50]; p<0.01) and IL-9 (0.89 [0.50–2.65] versus 2.44 [0.91–3.01]; p<0.01) and a decrease of percentages of IFNγ (9.85 [6.75–14.93] versus 5.18 [3.62–9.28]; p<0.01). These intracellular effects were most profound in virus-associated exacerbations.
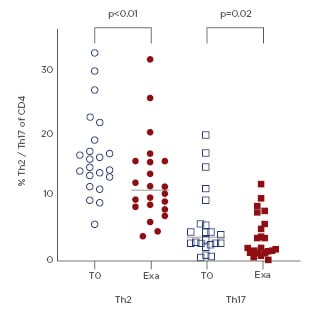
Figure 1: Percentage Th2 and Th17 of T-cells.
Exa: exacerbation; T0: stable timepoint.
CONCLUSION
In this study, >40% of AE were virus-associated. An increase of IL-5 and IL-9 and a decrease of IFNγ was seen, which is in line with previous literature.3,4 Moreover, the increase of IL-5 and IL-9 and the decrease of the number of eosinophils combined with a decrease of blood Th2 and Th17 cells during an AE might hint to migration from systemic inflammation to more local inflammation.








