Abstract
During the COVID-19 pandemic, frequent handwashing and disinfection have exacerbated or caused skin diseases. This case report shows the simultaneous development of allergic contact dermatitis to Kathon CG (DuPoint, Wilmington, Delaware, USA), Euxyl K400, and fragrance mix I because of the frequent use of moist (baby) wipes during the pandemic outbreak. The sensitisation to multiple allergens was determined by patch testing, whereas the disease resolved after usage discontinuation of moist wipes. Notably, two COVID-19 pulmonary events anticipated the clinical sensitisation. This case demonstrates that changes in attitude and behaviour with concern to exposure intensity to moist wipes can induce a clinical response to many allergens.
Key Points
1. The escalation of hygiene measures during the COVID-19 pandemic led to frequent hand washing and disinfection procedures as a way to reduce the spread of coronavirus. Many considered the use of moist wipes an equally healthy alternative to handwashing when other means were not available.2. There are several reports on hand erythema, scaling, burning, and fissures; all of which were classified as signs of toxic-irritant hand dermatitis. This case report shows multiple sensitisation to fragrances and preservatives.
3. The importance of specification of products’ ingredients on labelling is necessary to facilitate the discovery of potential allergens and to reduce the risk of side effects.
INTRODUCTION
COVID-19, the pandemic of the 21st century, emerged in Wuhan, China, and swiftly became a global phenomenon.1 As a result of the COVID-19 outbreak, the intensification of hygiene measures led to frequent hand washing and disinfection procedures.2-5 Such hygiene practices have caused or exacerbated skin diseases, especially among healthcare workers. These include irritant, atopic, and allergic contact dermatitis (ACD).3-6 The most common complaints were redness, dryness, itching, cracking, burning, flaking, peeling, and lichenification.7 This case shows a multiple sensitisation to fragrances and preservatives because of the frequent use of moistened wipes during the pandemic outbreak in a patient who had two COVID-19 pulmonary events.
CASE PRESENTATION
Disease History, Examinations, and Treatments
A 66-year-old White male complained about erythema and itching of upper extremities during the recent month. The objective examination revealed diffuse erythema on dorsal surfaces of forearms sensible to vitro-pressure and confluent oedematous infiltrative lesions. The authors observed similar but less evident lesions in the lower legs. The patient appeared like indurated psoriatic lesions; however, the subject has been addressed to an allergist after the exclusion of psoriasis by a dermatological examination. The initial suspect was a psoriasiform drug reaction because of the treatment of additional pathologies mentioned below.
The patient had rheumatologic arthritis for a couple of decades. The therapy comprised TNF-α antagonist (etanercept biosimilar) for the recent half-year and magnesium supplements for the last month. The patient also took antihypertensive and coronary artery disease medicaments such as ezetimibe/simvastatin, cardioaspirin, and bisoprolol over the last 15 years; also, six stents were inserted. Tamsulosin was also taken for prostatic hypertrophy. The patient preferred lactose- and gluten-free diets. There has been no change related to the detergents used. The patient had no prior allergic pathologies, nor a history of other family members with allergies.
The increased levels of rheumatoid factor, C-reactive protein, and erythrocyte sedimentation rate confirmed an inflammatory and autoimmune response (data not shown). Ultrasound examination of upper extremities revealed minimal oedema on the right radio-carpal articulation, minimal effusion on the periarticular areas, and around attachment sites of tendons, with hypoechogenic images in their distal segments. The subject reported two severe COVID-19 events, and the radiologic examination showed diffuse ground-glass opacities in the subpleural pulmonary zones.
The patient was informed and given official consent about procedures of examination, diagnostic methods, and treatment medications (including risks and benefits). The discontinuation of recently introduced medicaments (from other caregivers) revealed no changes in health status. Then, the successful treatment with methylprednisolone (locally, twice daily) and decreasing dose of prednisone (orally, initial daily dose 20 mg) for 2 weeks suggested the presence of ACD. It was only at this point that the patient informed the authors about the present prominent lesions at the perianal and perineal zones similar to those observed before in the upper extremities (not under recent treatment with methylprednisolone [hl]Table 1[/hl]). The imperative patch test (European Baseline Series, Chemotechnique Diagnostics, Vellinge, Sweden) revealed a sensitisation to fragrance mix I, Euxyl K400 (phenoxyethanol and methyldibromo glutaronitrile), and Kathon CG (methylisothiazolinone/methylchloroisothiazolinone [MI/MCI], produced by DuPoint, Wilmington, Delaware, USA), both 48 and 96 hours after their application (Table 2) and Figure 1). Balsam of Peru revealed a suspect 48 hours after application. Thus, an ACD is confirmed. The simultaneous patch testing for bisoprolol, aspirin, tamsulosin, and ezetimibe/simvastatin did not show sensitisation.
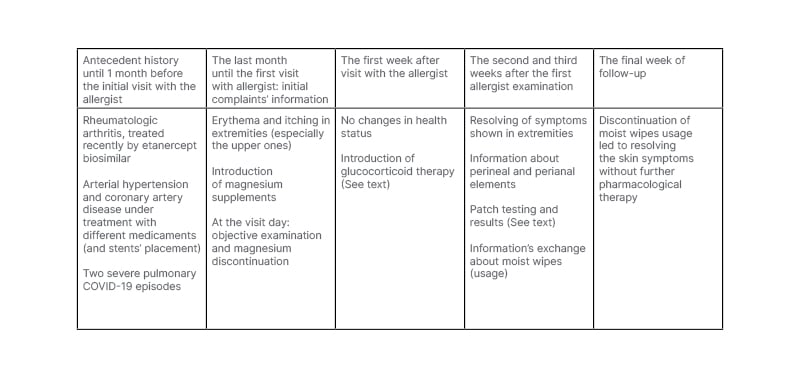
Table 1: The timeline of historical and current information.
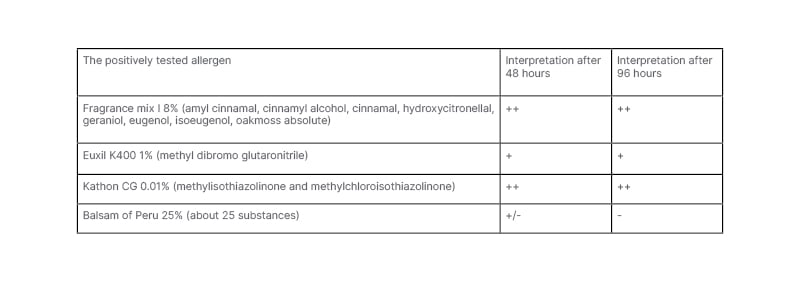
Table 2: The positively tested allergens
Figure 1: The positive patch testing (++) 96 hours after application (methyldibromo glutaronitrileon the left above; methylisothiazolinone and methylchloroisothiazolinone on the left below; and fragrance mix I on the right).
The authors informed the patient that the positively tested compounds are commonly present in the moisturised (baby) wipes. Afterward, the patient mentioned their frequent use for hygienic care of hands, forearms (even as a refresher), and perianal and perineal zones during the COVID-19 pandemic (even 1 day before the test interpretation). The source was not patched on the patient because the presence of the allergens was confirmed on the wipes used. According to the allergist’s recommendations, the patient agreed to discontinue the usage of moist wipes and avoid other potential contactants and, 1 week later, the skin appearance had normalised without pharmaceutical treatment.
Differential Diagnosis
In the authors’ case, morphological appearance and distribution mimicked psoriatic lichenification or lichen planus-like reactions, which belong to atypical manifestation patterns together with erythema multiforme-like reactions, bullous, nodular, granulomatous reactions, lymphomatoid reactions, etc.8 The history of pharmaceutical treatment suggested a cutaneous adverse drug reaction that, depending on its severity, may also mimic ACD. Based on the distribution pattern to contact localisation, simulators of ACD include psoriatic lesions induced by mechanical or chemical triggers, irritant contact dermatitis, dermatitis artefacta, Norwegian scabies, etc.8 Their exclusion happened in accordance with the (in)effectiveness of mentioned medical measures.
DISCUSSION
General Considerations
Since the outbreak of COVID-19, authorities have preached the importance of personal hygiene, including handwashing and disinfection.5 The COVID-19 pandemic has led to a significant increase in the incidence of signs of irritant hand eczema, since most of the affected subjects reported erythema, scaling, burning, and fissures that were classified as predominant signs of toxic-irritant hand dermatitis rather than contact allergy.3 Of the populations who had never had hand eczema, a consistent proportion of studied subjects developed the pathology after returning to day care, showing a significant association with the frequency of handwashing or disinfection.4,5
The general population considers the use of moist wipes an equally healthy alternative to handwashing when other means are not to be disposed of. Yet, the first ACD case caused by moist toilet wipes was reported in 1980 and was related to a fragrance.9 The incidence of ACD to their ingredients has exploded over the last decades of the 20th century.9-12
According to the literature, the most frequent allergenic ingredients in moist wipes are preservatives and fragrances.9 Among them, the allergy caused by preservatives shows more prominence than fragrance-induced allergy. Like our case, allergic contact perianal dermatitis because of a specific fragrance, cinnamic alcohol, was reported in a woman.9 Both Fragrance mix and Balsam of Peru contain some common substances like different cinnamates or eugenol, which often can lead to double positivity.13 In our case, the early positive response to both allergenic substances reflects this commonality, even not as a principal response.
Yet, the most problematic allergens in moist toilet wipes are preservatives. In the past, the most common allergen was the complex MI/MCI, also known as Kathon CG.9,11,14,15 There were high sensitisation rates to MI/MCI in Europe, because of its widespread use in cosmetic products.9-11,16-18 The negative publicity on the cosmetics preservative MI/MCI has made many cosmetic manufacturers look for safer alternatives.14,15 The most popular substitute is Euxyl K400. Unfortunately, this preservative also induces allergic reactions to cosmetics and to ‘moist toilet wipes’, being thus, a growing problem.10-12,14,15 According to the literature, the perianal and perineal occurrence of eczema should suggest an allergy to methyldibromo glutaronitrile and MI because of exposure to moistened toiled tissues.12,14,16-18 Together with perianal and perineal zones, the regular usage of moist wipes by positive patients induces almost allergic and irritative symptoms in the hands and lower legs.11,16,18 The lower lesions’ intensity on the lower legs suggests an occasional contact with culprit allergens. Besides the reaction to baby wipes and moist towelettes, a minority of MI-positive patients reacted to MI in shampoos, conditioners, deodorants, moisturisers, etc.18
Case Details
Considering the moist wipes as a healthy hygienic instrument, the authors’ patient used them for refreshing the affected forearms, without suspecting the deterioration of the situation. Patch testing revealed the simultaneous allergic sensitization at least to a fragrance (contained in the fragrance mix I), and at least in two different preservatives (contained in MI/MCI and Euxyl K400). To the authors’ best knowledge, this is the first report of a triple sensitisation that causes ACD to allergens mentioned above. The sensitisation happened only after extensive use of moist wipes because of the COVID-19 pandemic. Like other reports, their usage discontinuation resolved the ACD.16,17 Both fragrances and preservatives are of evident relevance to ACD, as suggested by patch testing results, and the knowledge that many sorts of moist wipes (used in the authors’ country Albania) contain fragrances.
Other differential diagnoses we considered include COVID-19 infection-related immunological cutaneous eruptions and TNF-α-induced psoriasiform eruption. Severe COVID-19 has an immense effect on the immune and neuropsychological systems and, therefore, can trigger additional pathological situations. Rheumatoid arthritis and, especially, the use of TNF-α antagonists may induce psoriatic manifestations.19,20 However, such manifestations cannot be resolved quickly after usage cessation of moist baby wipes.
Originating from a case report, these findings show the effectiveness of patch tests and allergens avoidance, as well as the limitation of the missed study sample. The final diagnosis (ACD) replaced psoriasiform drug reaction after partially successful treatment with glucocorticoids when the patch testing resulted positive for the allergens mentioned above. Afterward, the patient and their allergist exchanged information about the allergens’ exposure and hygienic attitudes concerning moist wipes. The consecutive discontinuation of their usage led to skin normalisation (both the resolving of erythema and itching confirmed by the patient too). Collectively, these facts were the ultimate proof of ACD.
CONCLUSIONS
This case shows that the use of moist wipes causes a simultaneous clinical sensitisation to at least three quite different allergens, which can affect all the body zones exposed to. The changes in hygienic attitudes because of the COVID-19 pandemic and an inevitable increase in exposure intensity to them can initiate a clinical response. Consequently, the specification of products’ ingredients on their labels is necessary to facilitate the discovery of a potential allergen and to reduce the risk of side effects caused by culprit allergens.







