Abstract
Caseous mitral annular calcification (CMAC), sometimes called liquefaction necrosis of mitral annular calcification (MAC), is a rare variant of MAC, a chronic degenerative process that progresses with age. It is a degenerative abnormality of the fibrous tissue and typically involves the posterior annulus, appearing as a smooth mass with no flow or acoustic shadow artefacts. It can be differentiated from other cardiac masses by CT and MRI. Whilst benign in nature, it is associated with a range of pathologies, such as mitral valve dysfunction, arrhythmias, and systemic embolisation. Given the risk of systemic embolisation, surgery may be appropriate, but there is no clear consensus in the literature in patients who are asymptomatic. This case highlights a case of CMAC presenting with a retinal artery occlusion, and was managed conservatively.
Background
The mitral annulus is a key component of the mitral valve, ensuring appropriate function by facilitating complete closure of the leaflets during systole. With age, MAC can occur, potentially resulting in mitral valve dysfunction, infective endocarditis, and arrhythmias. CMAC is a rare variant of MAC, typically involving the posterior annulus. Whilst benign, the risk of complications, such as systemic embolisation, may indicate a surgical approach to management, even in patients who are asymptomatic.
Case Presentation
A 60-year-old female, presenting with sudden loss of vision in the lower part of her left eye due to a branch retinal artery embolus, was admitted from ophthalmology services. An outpatient transthoracic echocardiogram showed an abnormal mass on the posterior annulus of the mitral valve. Transoesophageal echocardiography identified a calcified posterior aspect of the mitral annulus with normal leaflet mobility and trivial regurgitation, and an echogenic mass attached to the ventricular aspect of the mitral annulus (at the level of posteromedial commissure), 9×4 mm in size. A cardiac CT showed a caseous mitral valve with evidence of rupture of the calcified shell. Discussion with the surgical multidisciplinary team resulted in a conservative approach, with follow-up echo for monitoring.
Conclusion
Whilst a benign and rare variant of MAC, CMAC is associated with a range of pathologies, including mitral valve disease, arrhythmias, and systemic embolisation. Currently, there is no standardised management approach for CMAC. Surgery is currently recommended in the context of already known surgically indicated pathologies, such as severe mitral valve disease or systemic embolisation. However, this conservatively-managed patient has had no further complications with stable echocardiographic appearance on repeat echo 3 months following initial exam.
Key Points
1. Caseous mitral annular calcification (CMAC) should be considered in the differential when assessing mitral annular masses, and can be differentiated with CT and MRI.
2. Whilst a benign and rare variant of mitral annular calcification, CMAC is associated with a range of pathologies, including mitral valve disease, arrhythmias, and systemic embolisation.
3. Currently there is no standardised management approach for CMAC. Surgery could be considered in good surgical candidates who are asymptomatic, given the reported prevalence of cardioembolic events associated with CMAC.
BACKGROUND
The mitral annulus is a key component of the mitral valve, ensuring appropriate function by facilitating complete closure of the leaflets during systole. With age, calcification of the annulus can occur, termed mitral annular calcification (MAC), and is a chronic degenerative process that can result in mitral valve stenosis and/or regurgitation, infective endocarditis, and arrhythmias. Caseous mitral annular calcification (CMAC) is a rare variant of MAC, typically involving the posterior annulus. Whilst a benign mass in nature, the risk of complications, such as systemic embolisation, may indicate a surgical approach to management, even in patients who are asymptomatic. This case highlights a patient who was identified to have CMAC following presenting with a retinal artery occlusion, and was managed conservatively.
CASE PRESENTATION
A 60-year-old female presented to local eye services, reporting a 4-day history of new ‘floaters’, followed by a sudden loss of vision in the lower part of her left eye. They were identified by the ophthalmology services to have an embolus in a branch retinal artery on fundus photography, and was referred to the acute inpatient medical team for further assessment.
The patient’s past medical history included a previous left knee replacement, BMI of 43, non-medicated hypertension, and vertigo.
Observations on admission were normal, with a respiratory rate of 17, O2 saturation 96% on air, blood pressure 118/65 mmHg, and heart rate of 75 beats per minute. The patient was afebrile, though on repeat observations they were noted as mild hypertensive with average blood pressure 130–140 mmHg systolic. Examination demonstrated no positive findings, with no evidence of irregular pulse or abnormal cardiac auscultation.
Initial haematology and biochemistry results were as follows: haemoglobin: 149; white cell count: 8.6; neutrophil: 5.18; calcium: 2.5; creatinine: 70; estimated glomerular filtration rate: 77; alanine transaminase: 18; bilirubin: 12; total cholesterol: 6.3; non-high-density lipoprotein cholesterol: 4.9; low-density lipoprotein: 4.1; and HbA1c: 39. CT head demonstrated small vessel disease but no evidence of acute infarction. Chest X-ray was normal.
Following inpatient assessment, the patient was discharged for outpatient transient ischaemic attack clinic review, provided with aspirin and statin therapy with outpatient echo, and carotid Dopplers and Holter were arranged. Outpatient carotid and vertebral Doppler were unremarkable with minimal disease, and outpatient 7-day Holter demonstrated sinus rhythm with no evidence of atrial fibrillation.
However, outpatient transthoracic echocardiogram (TTE) demonstrated an echogenic nodular anomaly closely related to the posterior mitral annulus with a mobile linear structure attached, possible vegetation or extension of caseous calcification (Figure 1). Normal left ventricular dimensions with visually normal systolic function were noted, with a visually estimated left ventricular ejection fraction 55–60%. In addition, the report stated a visually normal right heart size and normal systolic function, mildly dilated ascending aorta with no other significant valvular abnormalities.
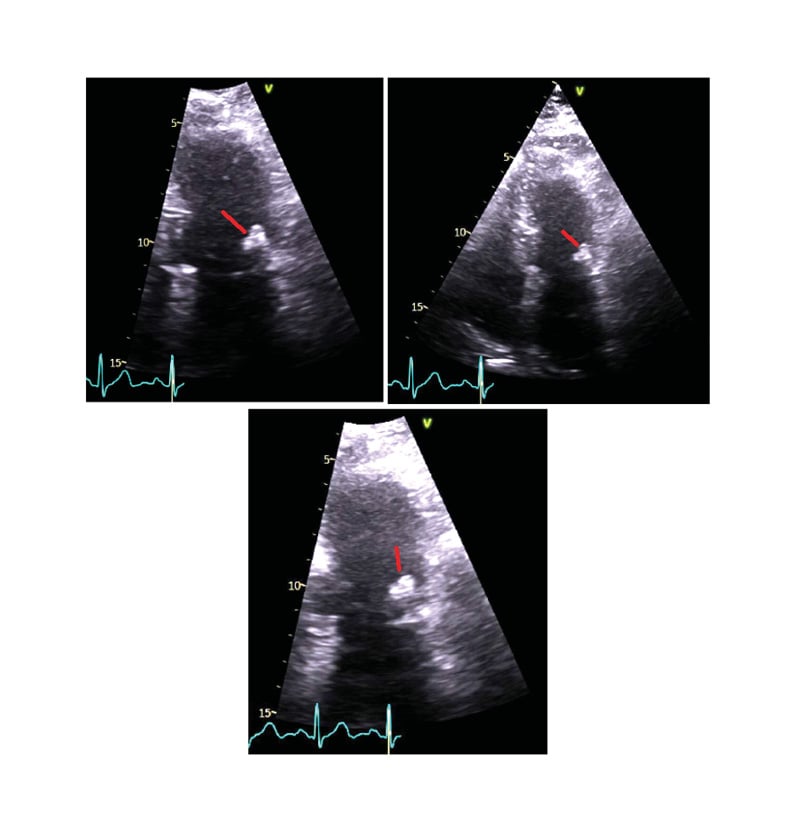
Figure 1: Transthoracic echocardiogram images.
The patient was admitted from outpatient echo following the report for urgent inpatient transoesophageal echocardiogram (TOE) and assessment for infective endocarditis. However, further blood investigations and microbiology cultures were all unremarkable, with no evidence of raised infection markers or positive microbiology results.
TOE demonstrated an echogenic posterior aspect of the mitral ring with normal mitral leaflet mobility with trivial mitral regurgitation. An echogenic mass was seen attached to the ventricular aspect of the mitral annulus at the level of posteromedial commissure, 9×4 mm in size, that was initially thought to represent a fibroma, or less likely an old vegetation (Figure 2). There was no thrombotic lesion attached to it.
The case was discussed with the regional cardiothoracic multidisciplinary team with an outcome of repeat TTE in 3 months time, which if demonstrated change for re-discussion at the multidisciplinary team for consideration for surgical intervention.
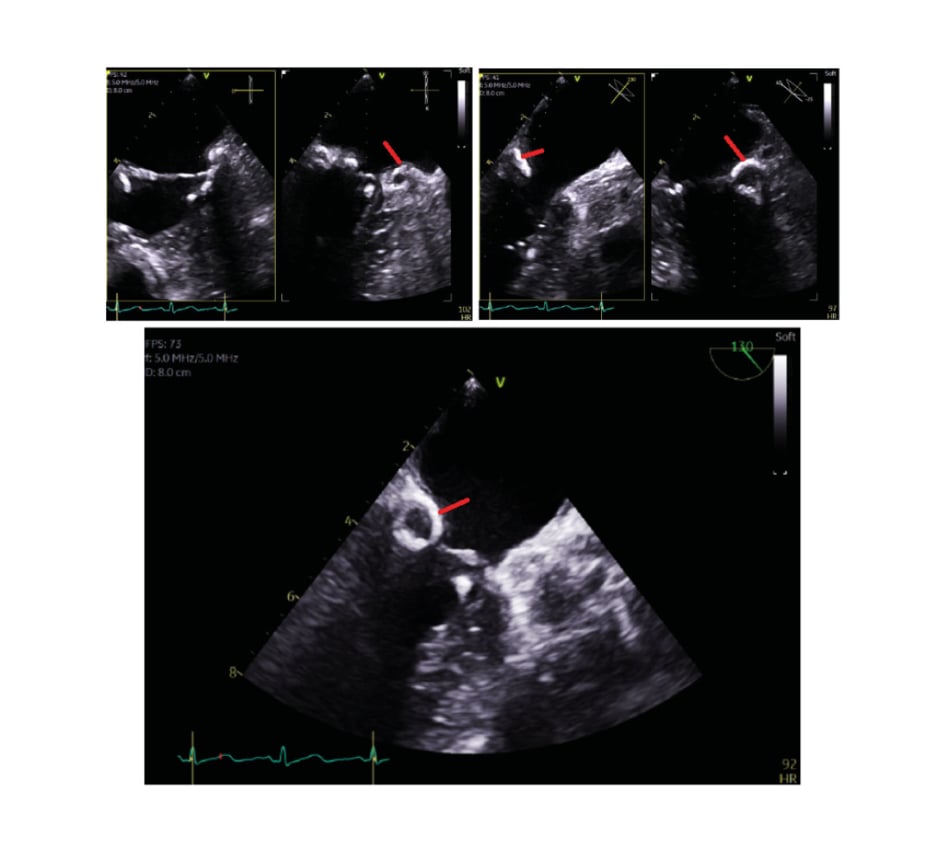
Figure 2: Transoesophageal echocardiography images.
An outpatient CT coronary angiogram revealed severe calcific atheroma, but was unable to fully evaluate the coronary arterial lumens (Figure 3). The CT also showed mixed calcific and soft tissue related to the mitral valve annulus. Caseous degeneration was diagnosed with evidence of rupture of the calcified shell.
Repeat TTE demonstrated the known caseous calcification of the posterior mitral annulus as previously reported, though the previous mobile attachment noted was not readily identified, possibly seen in the subcostal window with no significant increase in size.
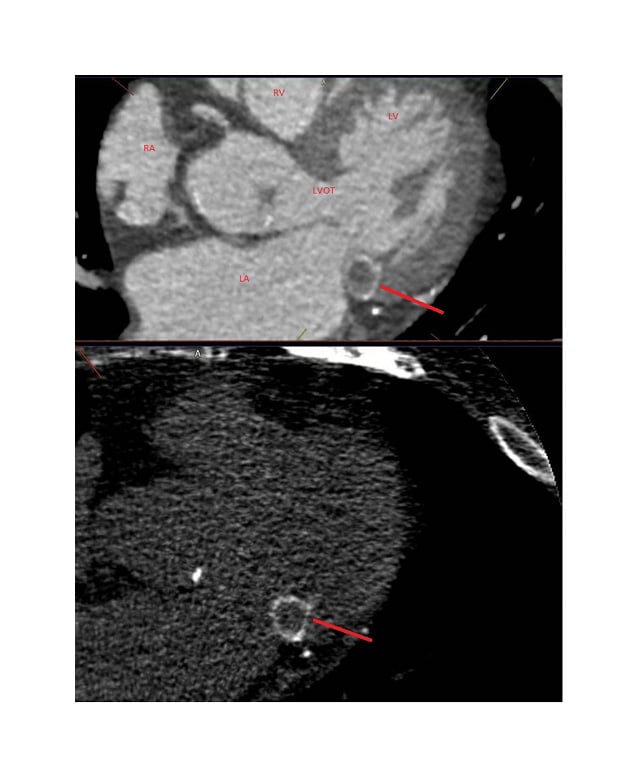
Figure 3: CT coronary angiogram image.
Single antiplatelet therapy was initiated given the evidence of non-obstructive coronary artery disease, but anticoagulation therapy on risk–benefit balance was not initiated. The patient has had no further admissions suggestive of ongoing complications due to this pathology.
DISCUSSION
The mitral valve has a marginal fibrous ring known as the mitral annulus. This is an extension of the cardiac fibrous skeleton to which the mitral valve leaflets are attached. Its function is to reduce the surface area during systole to facilitate complete closure of the leaflets.
MAC, most common in elderly females, is a chronic degenerative process that progresses with age and can result in mitral valve disease (both stenosis and regurgitation), infective endocarditis, atrial arrhythmias, and high-grade block.1 It is more commonly found in patients with systemic hypertension, mitral valve prolapse, raised left ventricular systolic pressure, aortic valve stenosis, chronic kidney disease, secondary hyperparathyroidism, atrial fibrillation, and risk factors for the development of coronary artery disease.2
CMAC, sometimes called liquefaction necrosis of MAC, is a rare variant of MAC. It typically involves the posterior annulus, appearing as a smooth mass with a surrounding hyper-echogenic rim, with no flow or acoustic shadow artifacts.3 The mass typically consists of calcium, fatty acids, and cholesterol, and though benign can cause significant clinical concern, such as mitral stenosis or regurgitation, left ventricular outflow obstruction, or systemic embolisation, such as in this case study.4
The prevalence of CMAC amongst echocardiographic studies has been reported as 0.640% of patients with MAC, and 0.068% of the general population, though incidence amongst an autopsy series was 2.700%, possibly suggesting prevalence may be higher.5 Most cases are incidentally found on echocardiograms or during catheterisation with fluoroscopy of the annulus. However, cases can present following investigation for embolic causes, or as work-up for possible cardiac tumours, possibly presenting as new murmurs.
Echographically, CMAC appears as a round or semilunar bright echogenic mass in the peri-annular region of the mitral valve. Typically, it contains a central echolucent area reflecting liquefaction, but with no acoustic shadowing artefacts such as in MAC, which presents with a posterior echo shadow.3 On 3D echo imaging, it appears similarly.6 Whilst TTE can be helpful in identifying CMAC, it is well known to be limited in accuracy and diagnostic value in some patients due to poor acoustic windows, and therefore TOE can provide high-quality visualisation.
Differentiation of CMAC from other cardiac masses, usually tumour or thrombus, can be done with CT or MRI. On CT, CMAC typically appears as well-defined oval or crescent shaped calcified mass, with peripheral calcification with high Hounsfield units, and lacks contrast enhancement. The central hypodensity is considered to be due to the liquefactive material within the centre of the mass.6 On cardiovascular MRI, T1/T2 steady-state free precession and post-gadolinium sequences will be required, and the calcium is low signal on all sequences, the centre area is more fluid, so low on T1 and high on T2 and steady-state free precession without avid vascularity.
Currently, there is no standard treatment protocol for CMAC. Given the benign nature of CMAC, some authors have recommended a conservative management for asymptomatic cases.7 However, a literature review by Dietl et al.9 suggested elective surgical resection in good operable candidates in asymptomatic cases, given the risk of embolic strokes unrelated to atrial fibrillation. Dietl et al. reported prevalence of cardioembolic events associated with CMAC as 19.2% amongst literature they reviewed.8,9 Though, if the embolism is more consistent with a fat embolism, then anticoagulation is unlikely to be effective. However, surgery is generally considered in good surgical candidates, where the clinical situation is in the context of other established surgical indications, such as severe mitral valve disease or systemic non-embolisation, especially if embolisation is in the context of established anticoagulation.