BACKGROUND
One year after the identification of the novel severe acute respiratory syndrome 2 (SARS-CoV-2) infection in Wuhan, China, and the outbreak of the virus worldwide, the pandemic state persists, and the management of COVID-19 remains burdensome, with the number of people infected daily increasing progressively in most countries and the death rate being alarmingly elevated.1 Since the elevated rate of infectivity of the virus, the authorisation of histological examination has been a harsh process, with high-risk of contagiousness even in qualified medical personnel.2 However, thanks to the recently published histological reports, more about the pathogenic mechanism underlying viral-derived tissue damage has been understood. In this report, the authors describe a patient hospitalised for COVID-19 who developed necrotic acral lesions that were biopsied.
CASE REPORT
An 83-year-old female came to the emergency department because of acute respiratory distress, which required oxygen therapy. An oronasal swab was performed to identify SARS-CoV-2 RNA and the test resulted positive. Due to her rapidly deteriorating clinical condition, the patient was admitted to the infectious disease ward. Five days after her inpatient stay, she developed vesicular lesions on the lower limbs, which quickly became necrotic (Figure 1). A skin biopsy was performed, and the histopathology report evidenced a dermal-epidermal inflammatory infiltrate made of lymphocytes, histiocytes, neutrophils, and eosinophils, which displayed either an interstitial and periannexial distribution or a perivascular one with endothelial swelling and detachment.
The epidermis demonstrated spongiosis, erosion, and vesicles with mild keratinocyte acantholysis, and inflammatory cells (lymphocytes, histiocytes, and multinucleate giant cells (Figure 1).
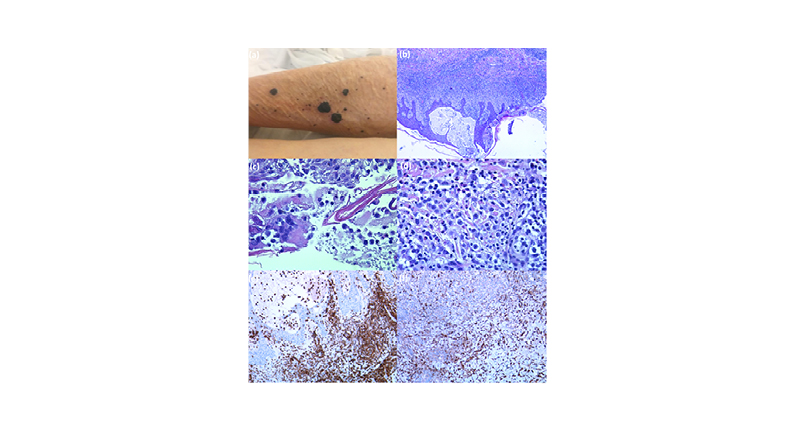
Figure 1: Gross and microscopy appearance of the necrotic lesions of a patient with COVID-19.
A) Multiple necrotic lesions on the lower limb of the patient. There are four main lesions and numerous other lesions with smaller diameters. B) Histological examination showing an intraepidermal vesicle and dense inflammatory infiltrate within the dermis (H&E 10x). C) Histology of the necrotic lesions. The lumen of the blister contains histiocytes and multinucleated giant cells (H&E 40x). D) Histology of the necrotic lesions. Many eosinophils associated with lymphocytes and histiocytes in the superficial dermis (H&E 40x). E) Numerous dermal and intraepidermal inflammatory cells are T lymphocytes as demonstrated with anti-CD3 rabbit monoclonal primary antibody 2GV6 (100x immunohistochemistry with haematoxylin counterstain using Ventana Ultraview DAB detection Kit in a Ventana BenchMark Ultra Processor® [Ventana Medical Systems, Tuscon, Arizona, USA). F) Histology of the necrotic lesions. The lymphocytes are mixed with numerous macrophages as demonstrated with anti-CD68 (KP-1) antibody (100x immunohistochemistry with haematoxylin counterstain using Ventana Ultraview DAB detection Kit in a Ventana BenchMark Ultra Processor®).
CD: cluster of differentiation; H&E: haematoxylin and eosin staining.
The inflammatory infiltrate was composed mainly by T cells (CD3+, CD4+, and CD8+), histiocytes (CD68+), with rare B cells (CD20+) and activated blasts (CD30+) (10–20%). Moreover, many mitotic figures were evident (Ki67). No natural killer cells were detected (CD56+) and no Epstein–Barr virus-LMP1 viral protein was present. In addition, myeloperoxidase, CD34, and Mart1 were negative. This morphologic report led to the identification of the lesions as SARS-CoV-2-related papulovesicular eruptions.
DISCUSSION AND CONCLUSIONS
Mounting scientific evidence has emerged regarding the manifestations of SARS-CoV-2 infection on the skin. Regarding histological reports, while several cases of cutaneous manifestations have been described, relatively few cases were subjected to biopsy because of the numerous limitations imposed in medical centres. Current data reports different histological pictures related to specific clinical aspects.3
The maculopapular lesions show lymphocytic exocytosis, with thrombosed vessels filled with neutrophils and eosinophils; conversely, dermatitis is characterised by infiltrated perivascular lymphocyte, focal elements of suprabasal acantholysis, dyskeratotic cells, and swollen vessels with mixed lymphocyte infiltration in the dermis.3 Vesicular (varicella-like) eruptions show dyskeratotic cells with acantholysis, intraepidermal vesicles, and suspected viral inclusions within multinucleate cells.3
Compellingly, a retrospective analysis carried out on 23 patients with COVID-19 and with cutaneous manifestations showed that, at histopathological level, microvascular and endothelial damage were evident. Infiltrating perivascular lymphocytes, thrombosis, and swollen vessels were present and C5b deposits were predominant.4 Of note, the activation of the complement system and coagulation cascade had already been identified as responsible for vascular clinical manifestations such as thrombosis and necrosis. However, in the authors’ case, the presence of abundant infiltrating lymphocyte, dilated vessels, and epidermal vesicles filled with inflammatory infiltrators without obvious elements of thrombosis may suggest a combination of direct viral and immune mediated damage.
Further studies are needed to better understand the skin involvement in COVID-19, in terms of clinical characteristics, evolution, and correlation with the severity of the disease, and the pathological mechanism responsible for cutaneous damage. From the data published so far, it can be assumed that the visible skin lesions are the result of a combination of the direct action of the virus and the immune signalling cascade induced by it.








