BACKGROUND
Cutaneous ulceration presents many challenges to the dermatologist and multidisciplinary team due to a broad differential diagnosis, difficult management decisions, and delayed wound healing.1 New ulceration should always prompt consideration of infection, with tissue samples rather than swabs often required, along with extended and cold cultures.2,3 Reported here is a case of mycobacterial infection mimicking pyoderma gangrenosum (PG) and Crohn’s disease (CD) in which tissue culture prevented potential disseminated infection with the use of immunosuppressant biologic agents.
CASE REPORT
A 78-year-old male presented to the emergency department with a 4-week history of an enlarging ulcer on the left upper thigh. On examination, a 10×5 cm area of ulceration was noted, with a violaceous undermined edge (Figure 1). Despite the appearance, the patient reported the ulcer to be nontender. The patient had been treated for suspected cellulitis at this site 3 weeks prior with 1 week of intravenous flucloxacillin followed by 1 week of oral treatment. In addition, he had been diagnosed with CD 4 months previously and received one dose of the gut-specific α4β7-integrin blocker vedolizumab. An improvement in stool frequency was noted, but he remained persistently anaemic with a haemoglobin of 95 g/L (130–170 g/L) dependent on regular red cell transfusion.
Routine bloods revealed a C-reactive protein of 26.4 mg/L (<5 mg/L) with a normal serum protein electrophoresis and negative autoantibody screen. The working diagnosis was pyoderma gangrenosum secondary to CD and possible exacerbation by vedolizumab.4
Incisional biopsy for histopathology demonstrated a dense neutrophilic infiltrate supportive of PG; however, multinucleated giant cells forming noncaseating granulomata were also noted (Figure 1). Ziehl–Neelsen and auramine–rhodamine staining for mycobacteria were negative. Prolonged culture for atypical organisms was requested.
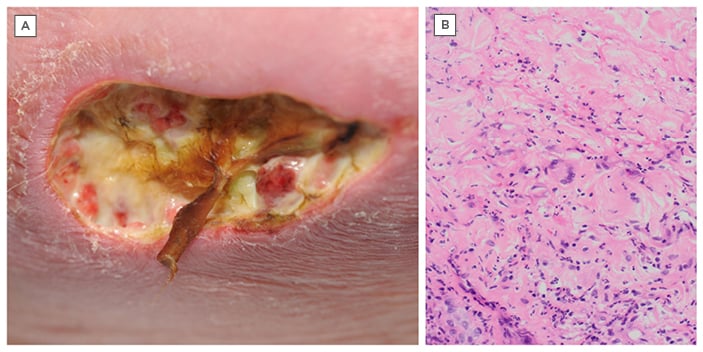
Figure 1: A) Clinical photograph of the left upper inner thigh. B) Histopathology photograph (haematoxylin and eosin staining 200x magnification) of inflammation with multinucleated giant cells.
Multidisciplinary input was sought and treatment initiated with oral prednisolone 0.5 mg/kg with screening bloods for biologics sent. A decision was made to commence infliximab to treat the CD and PG concurrently. Pseudomonas aeruginosa was subsequently isolated on swabs and due to increasing exudate, treatment with intravenous piperacillin/tazobactam and ciprofloxacin was commenced. While the exudate improved, the ulcer edge continued to appear violaceous and active.
Seven days before a planned admission for infliximab, Mycobacterium bovis was isolated from skin culture. The source of infection was not found. On review of gastrointestinal histology, no definitive features of CD had been identified and so primary colonic mycobacterial infection was suspected. The patient completed 9 months of antimycobacterial treatment consisting of rifampicin/isoniazid and ethambutol for 2 months followed by rifampicin/isoniazid alone for 7 months. He achieved complete healing of the ulcer site with resolution of all bowel symptoms and a return to activities of daily living unassisted.
CONCLUSION
PG is a diagnosis of exclusion with a differential diagnosis including malignancy, vasculitis, infection, and other rarer causes.2 Fatal disseminated M. bovis infection following Bacillus Calmette–Guérin vaccination has previously been reported in an infant born to a mother taking infliximab.5 Infliximab has been shown to be the anti-TNFα agent most commonly associated with mycobacterial infection.6 This case underpins the value of routine tissue culture in both presumed PG and atypical ulceration, a step that can often be forgotten. It is easy to experience diagnostic bias when faced with clinical PG-like ulceration albeit in the absence of pain. In this present patient, without tissue culture, disseminated mycobacterial infection may have ensued.








