INTRODUCTION
The disaccharide galactose-α-1,3-galactose (alpha gal) is a typical component of glycoproteins in mammals.1 Since primates, including humans, have lost the enzyme required for alpha gal production, it is immunogenic and large amounts of natural IgM and IgG antibodies to it are produced by sensitised B cells in humans. On the other hand, IgE antibodies specific to alpha gal are never produced in humans except for in patients with a delayed immediate-type reaction to red meat and drugs containing alpha gal.2 It has been demonstrated that development of alpha gal syndrome is closely associated with tick bites,3 suggesting that tick bites promote production of IgE antibody to alpha gal, although the mechanism remains poorly understood. To address this issue, we stratified patients by historical frequencies of tick bites and investigated the association between serum anti-alpha gal IgE levels and basophils.
METHODS
A total of 56 patients who presented at Shimada Municipal Hospital with tick bites were enrolled in this study after providing informed consent. Patients were categorised into four groups: patients with 1 tick bite (1°), patients with 2 tick bites (2°), patients with 3 tick bites (3°), and patients with ≥4 tick bites (≥4°). The study was performed according to the Declaration of Helsinki, and the study protocol was approved by the Shimada Municipal Hospital Ethical Committee.
RESULTS
Because the alpha-gal epitope is structurally related to the blood type B antigen, production of the specific IgE antibody was suppressed in patients who had the B antigen.4 We found that the specific IgE antibody levels were significantly increased in association with tick bite frequency in patients with A or O blood types, but not with B or AB blood types (Figure 1A). Because the B antigen can disturb the processes of anti-alpha gal IgE antibody production, patients with B or AB blood types were excluded from further investigations.
Patient groups were stratified by historical tick bite frequencies and the chronological variations of the antibody levels among the groups (1–2 days, 3–5 days, 6–10 days, or ≥10 days after bites) were compared (Figure 1B). We found three distinctive patterns: marginal levels in the patients with 1°, a gradual increase in the patients with 2°, and a sharp increase in the patients with ≥3°.
We also compared chronological variations of the skin-infiltrating basophils among the groups. Although a marginal increase in the number of basophils was found in the patients with 1°, the number of basophils increased gradually in the patients with 2° and sharply in the patients with ≥3°, followed by a decrease there after (Figure 1C).
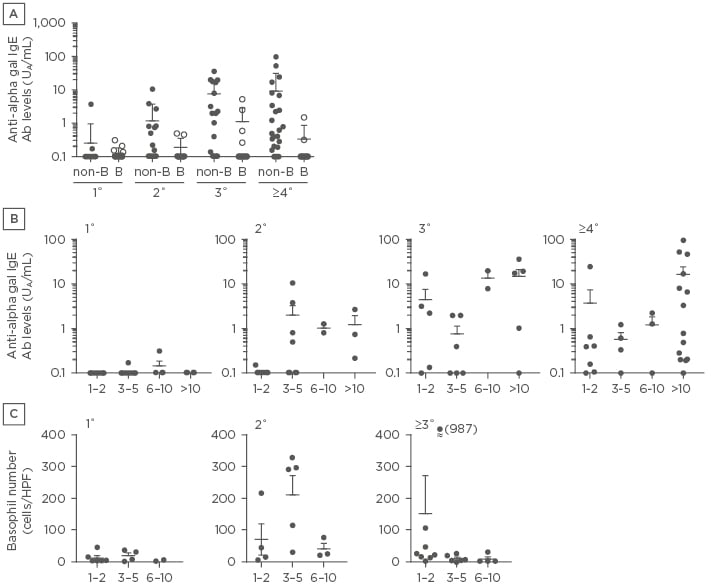
Figure 1: A) Anti-alpha gal IgE levels in patients with different tick bite frequencies who have the non-B (blood types A or O) or B blood types (blood types B or AB). B) Chronological variations of anti-alpha gal IgE levels. C) Chronological variations of basophil numbers in bitten skin.
1°: patients with 1 tick bite; 2°: patients with 2 tick bites; 3°: patients with 3 tick bites; ≥4°: patients with ≥4 tick bites; Ab: antibody; Alpha gal: galactose-α-1,3-galactose; HPF: high power field.
We propagated the skin-infiltrating T cells from the lesions of the patients by a previously described method5 and investigated the cytokine production of these cells using intracytoplasmic staining with antibodies to cytokines, including IL-4, IL-5, IL-13, and IFN-γ, after stimulation with PMA. We found that Th2 cytokine production of skin-infiltrating T cells was augmented with increased bite frequency.
DISCUSSION
We found chronological variations of anti-alpha gal IgE levels and basophil kinetics in the patients with 1°, 2°, and ≥3° tick bite histories, respectively. Furthermore, we observed that tick bite histories were associated with Th2 cell infiltration in bitten skin. Because basophils have an antigen-presenting function to promote Th2 differentiation and IgE production, repeated tick bites causes accumulation of Th2 cells in bitten skin to provide a Th2 cytokine milieu, which is critical for class switching to IgE of sensitised B cells.








