Abstract
Acne vulgaris is the most common skin condition affecting the American population. The present review evaluates the topical and systemic therapies available in the USA for the management of acne reporting their relevance, efficacy, tolerability, and safety. This review also discusses alternative treatments such as light therapy, diet, and probiotics. Further research on acne therapy is needed given the high prevalence, and thus, the immense economic burden that the condition poses in our society.
INTRODUCTION
Acne is the most common skin condition in the USA, affecting over half of its population.1 It is an inflammatory skin disease that predominantly affects adolescents and young adults. Multiple factors are involved in the pathogenesis of acne including skin hyperkeratinisation around the follicular infundibulum, increased sebum production, colonisation of Cutibacterium acnes, and activation of the innate immune system leading to an inflammatory response.2 Acne represents a significant economical and psychological burden for our society, and thus, the relevance of understanding its pathogenesis and seeking an adequate treatment that is cost-effective, safe, and widely accepted by patients is extremely important. This review will discuss the different available options for the treatment of acne across time, focussing on agents and modalities currently used and their level of evidence and safety. An English language search for literature on PubMed using the key terms “acne”, “treatment”, and each of the individual therapies was included according to personal experience. Relevant articles were reviewed pertaining to these treatments used in daily practice in the USA and are presented here.
TREATMENTS
Over the last century, new treatment options have emerged for the management of acne. Figure 1 illustrates how topical and systemic agents have advanced over time.3 There is a vast number of current available treatment options for this condition and the success of such treatments relies upon the use of an individualised approach focussing on the severity of acne presentation, treatment side effects, patient preferences, patient education, and establishing realistic expectations to treatment.4 The treatment of acne can be divided in three major categories, which include topical agents, systemic agents, and miscellaneous or complementary treatments.
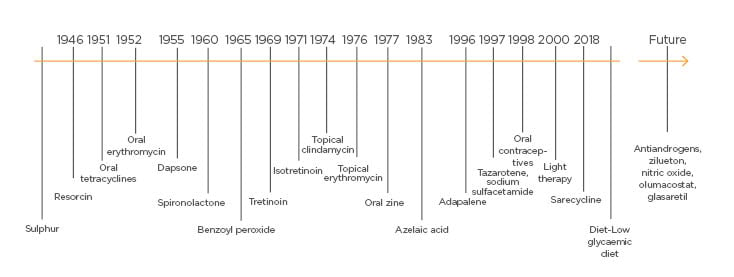
Figure 1: Treatment across time.
Acne treatment over the last 50 years.
TOPICAL AGENTS
Sulphur
Sulphur has been used for the treatment of acne since Ancient Egypt due to its antimicrobial and keratolytic properties.5 It is currently used mainly as an adjuvant to other therapies such as sodium sulfacetamide, which is also approved as a monotherapy treatment for acne in the form of washes, leave-on lotions, creams, masks, and foams in concentrations of 1–10%.5
Topical sulphur is tolerated very well with few side effects that may include skin dryness and malodour. Sulphur is a Category C medication; therefore, it should be only used in pregnant women when the benefits outweigh the risks.5
Resorcinol
Topical resorcinol for the treatment of acne has been used for centuries in low concentrations of 1–2% combined with other agents, such as a keratolytic agent. In higher concentrations of up to 40%, it has been used as a chemical peel for the treatment of hyperpigmentation, erythema, and scars.6
Topical Antibiotics
Efficacy of clindamycin and erythromycin is limited over time. Increased antibiotic resistance is reported with long-term treatments, particularly with erythromycin.7,8 The clinical guidelines for the treatment of acne in 2016 classified topical antibiotics as Grade A with a level of evidence of I–II.9
Such guidelines established the level of evidence of treatments according to the quality of selected studies that evaluated each therapy; similarly, the strength of recommendation was defined according to the best available evidence.9
TETRACYCLINES
Topical minocycline is a relatively new alternative for the treatment of acne lesions. Two randomised placebo-controlled Phase III studies evaluated the efficacy of minocycline for the treatment of acne during a 12-week period in 961 patients and concluded that treatment with minocycline was significantly superior in reducing the inflammatory lesion count (p0.05).10
Benzoyl Peroxide
Topical benzoyl peroxide is a comedolytic and antibiotic that effectively works against C. acnes.5 It has been used as an adjuvant medication to other topical antibiotics to reduce resistance to medication.11,12 Multiple studies have addressed its effectiveness alone or in combination with other agents such as topical antibiotics and retinoids. Shalita et al.13 evaluated the efficacy of benzoyl peroxide 6.0% wash and tretinoin 0.1% gel compared to tretinoin alone for the treatment of acne vulgaris in 87 patients during a 12-week period. They concluded that patients using the combination therapy had a significant reduction in the acne papule count (p=0.037) from Week 2, compared to patients in the monotherapy group who did not show a significant improvement in their acne until Week 12.13 Side effects were minimal and included local skin irritation.5 The clinical guidelines for the treatment of acne in 2016 classified it as Grade A with a level of evidence of I–II.9
Topical Retinoids
Topical retinoids include tretinoin, adapalene, and tazarotene. Their use in the treatment of acne has demonstrated satisfactory outcomes.14 An analysis of two multicentre, randomised, vehicle-controlled, Phase III studies evaluated the efficacy of topical tretinoin for patients with moderate-to-severe acne and demonstrated a significantly higher efficacy of tretinoin 0.05% in reducing acne lesion count compared to vehicle alone in 154 patients (p=0.001).14 U.S. Food and Drug Administration (FDA)-approved in 1996, adapalene has mainly been reported as a combined regimen with benzoyl peroxide with mean percentage changes of comedonal acne being superior than for those patients treated with adapalene as a single agent.15,16 Topical tazarotene was first approved in 1997 for the treatment of acne. A review of five Phase I studies in normal controls and two Phase III studies in patients with moderate-to-severe acne reported on the safety and efficacy of tazarotene 0.1% foam for acne vulgaris.17 Reported side effects are mainly limited to dryness and irritation.5 The clinical guidelines for the treatment of acne in 2016 classified them as Grade A with level of evidence of I–II.9
Salicylic Acid and Azelaic Acid
Both salicylic and azelaic acid are used as a topical keratolytic for the treatment of acne.5 Both agents have mild side events which are limited to local irritant reactions. The clinical guidelines for the treatment of acne in 2016 classified them as Grade B with a level of evidence II and Grade A with level of evidence I, respectively.9
Dapsone
Dapsone was first approved in 1955, and later its higher concentration (5%) was FDA-approved in 2008 for the treatment of acne.18 Topical dapsone 5.0% and 7.5% gel is used especially among female adults. In controlled studies, it significantly reduced the noninflammatory and total lesion counts in adult patients when compared to adolescents. Two double-blind, randomised Phase III studies evaluated the efficacy of dapsone 5.0% gel applied twice daily for the treatment of acne. At Week 12, patients on the dapsone group were found to have a significant reduction in their acne lesion count, particularly the adult patients compared to adolescents (p0.001).18 In clinical studies, it has demonstrated a safety and efficacious profile including in patients with glucose-6-phosphate dehydrogenase deficiency.19 Adverse events are minimal and include local reactions such as dryness and pruritus. The clinical guidelines for the treatment of acne in 2016 classified it as Grade A with a level of evidence of I–II.9
Nicotinamide
Recent reports and studies have investigated the efficacy of topical nicotinamide for the treatment of acne, but the current available data is too limited to create formal recommendations. Upon testing, either as single agent or in combination with antibiotics, nicotinamide has demonstrated an improvement of acne lesions.20,21 No major side effects have been reported. Future topical agents currently undergoing Phase II–III clinical trials include nitric oxide22 and olumacostat glasaretil.23
SYSTEMIC AGENTS
Birth Control Pills
Birth control pills reduce acne lesion counts and severity of lesions.24-26 Lucky et al.24 compared the efficacy of drosperinone 3 mg and ethinyl estradiol 20 μg for the treatment of acne vulgaris in a randomised, placebo-controlled study, and found that the therapy group had a significant improvement in the investigators overall improvement rating scale compared to those patients in the placebo group (p0.001). Norgestimate-ethinyl estradiol, drospirenone-ethinyl estradiol, norethindrone acetate-ethinyl estradiol, and ethinylestradiol/drospirenone/levomefolate calcium are FDA-approved for the treatment of acne.24-26 The most common side effects associated with them include weight gain, breast tenderness, mood changes, and vaginal bleeding;27 although rarely, thromboembolism and stroke have also been reported.27 Patients with uncontrolled high blood pressure or migraines with neurologic signs should not be started on birth control pills.28 The clinical guidelines for the treatment of acne in 2016 classified it as Grade A with level of evidence of I.9
Spironolactone
Spironolactone was first approved by the FDA in 1960. Spironolactone at doses 50–100 mg daily displays favourable results for the treatment of acne vulgaris through its antiandrogenic effects and reduction of sebum production.29-31 The most common reported side effects include menstrual irregularities, hyperkalaemia, and central nervous system symptoms such as lethargy, fatigue, dizziness, and headache.29,30 Physicians should be careful when prescribing this medicine concomitantly with potassium supplements, angiotensin-converting enzyme inhibitors, other potassium-sparing diuretics, digoxin, lithium, corticosteroids, and nonsteroidal anti-inflammatory drugs. Flutamide is a pregnancy Category C medication. The clinical guidelines for the treatment of acne in 2016 classified it as Grade B with a level of evidence of II–III.9
Systemic Antibiotics
Systemic antibiotics have been widely used for the treatment of acne vulgaris because of their antimicrobial and anti-inflammatory properties. Despite the increased risk of acquired antibiotic resistance, it is recommended to maintain a regimen of 6–8 weeks with a total duration of 3–6 months of therapy. The most commonly used antibiotics for the treatment of acne vulgaris include tetracyclines and macrolides.
Tetracyclines, including doxycycline, minocycline, tetracycline, and sarecycline, have been used for a long time with favourable results for the treatment of acne.32-34 Doxycycline is considered as first-line therapy for acne among systemic antibiotics. The most common side effects include gastrointestinal upset, photosensitivity, and hyperpigmentation.4,35 The clinical guidelines for the treatment of acne in 2016 classified them as Grade A with level of evidence of I–II.9 Tetracyclines are pregnancy Category B medications, and as such they are teratogenic.
Macrolides, including azithromycin, erythromycin, and clarithromycin, are a safe alternative to doxycycline for the treatment of acne, especially in those patients with poor tolerance or resistance to the treatment with doxycycline.36 A meta-analysis of randomised controlled trials was performed to analyse the efficacy of azithromycin compared to oral doxycycline in patients with moderate-to-severe acne vulgaris. Six studies were included and found no significant difference between the two groups in terms of lesion count improvement after therapy (p=0.27).36 Special consideration should be given to long-term antibiotic resistance where macrolides have a higher risk than clindamycin.37 The 2016 clinical guidelines for the treatment of acne classified them as Grade A with a level of evidence I.9 Trimethoprim has been used as an alternative in some patients. The 2016 clinical guidelines for the treatment of acne classified it as Grade B with a level of evidence of II.9
Isotretinoin
Isotretinoin is an FDA-approved oral retinoid for the treatment of acne vulgaris. Its mechanism of action covers the four major pathogenic factors involved in the development of acne and it is superior in disease control when compared with other medications in randomised clinical trials.9,39-41 Peck et al.39 performed a double-blind placebo-controlled study comparing the effects of isotretinoin initiated with 0.5 mg/kg/day with placebo over a 4-month period and reported a significant difference in acne improvement for the therapy group compared to placebo (p0.001 at 1 month and p0.008 at 2 months). Patients are commonly treated with 0.5–1.0 mg/kg/day until a cumulative dose of 120.0–150.0 mg/kg is reached.41 Reaching the cumulative dose is necessary for patients to see a long-lasting effect with less risk of recurrence. Relapses have been described more commonly in those who were treated in their early teens.42 More recently, a newer form of isotretinoin, called isotretinoin-lidose, has been released.43 This newer form increases the absorption levels of isotretinoin independently of the patient’s fasting status, thus maintaining the same safety and efficacy properties as the previous form.43,44 As a Category X medicine, isotretinoin is teratogenic and thus contraindicated in pregnancy, lactation, and in patients with severe hepatic and renal dysfunction.45 It should always be prescribed concomitantly with a reliable birth control method in females with child-bearing potential and everyone should be registered in the iPLEDGE program.9 Although caution should be taken in patients with suicidal tendencies and mental illness, studies have been inconclusive for a clear association among them.41,46-49 The clinical guidelines for the treatment of acne in 2016 classified it as Grade A with a level of evidence of I–II.9
Flutamide
Flutamide is a nonsteroidal antiandrogen medication infrequently used in dermatology for the treatment of acne. A randomised study compared the effects of flutamide with cyproterone acetate-ethinyl estradiol during a 6-month period and concluded that both treatments significantly decreased the acne severity scores (p0.001).50 The clinical guidelines for the treatment of acne in 2016 classified it as Grade C with a level of evidence of III.9
Zinc
Since 1977, zinc has been used for the treatment of acne after finding low levels of zinc in patients with the disease.51 The data on the efficacy of zinc for the treatment of acne has since been mixed.52-54 Zinc acts through several mechanisms such as regulation of protein, lipid, and nucleic acid metabolism and gene transcription; maintenance of an adequate immune activity; antimicrobial effects against C. acnes; and suppression of sebum production.55 Another systemic therapy currently undergoing Phase II–III clinical trials is zileuton.56
LIGHT THERAPY
Both photodynamic therapy (PDT) and lasers have demonstrated promising effects for the treatment of acne vulgaris, yet no long-term data from rigorous clinical studies is available. Their mechanism of action is mainly attributed to bactericidal and anti-inflammatory effects secondary to a reduction in the production of macrophage cytokines and a decreased sebum production due to thermic effects on the sebaceous glands.57
Photodynamic Therapy
The use of PDT for the treatment of acne has been recently described.58 Studies have compared its efficacy with oral antibiotics and a significantly higher improvement has been shown in the PDT group. A randomised study performed by Nicklas et al.59 in 46 patients reported that patients treated with PDT had a significant reduction in the noninflammatory lesion count (p=0.013) and total lesion count (p=0.038) after 6 weeks of therapy compared to those treated with doxycycline 100.0 mg/day plus adapalene gel 0.1%. A small clinical trial demonstrated improvement of acne lesions after PDT therapy with minimal side events, decreased sebum production, and decreased inflammatory markers on the lesions.58 Different photosensitisers (aminolevulinic acid, methyl aminolevulinate, and indole-3-acetic acid) and light sources (red light, pulsed-dye laser, intense-pulsed light, long-pulsed dye laser, and green light) could be used. The best outcomes are with the combination of aminolevulenic acid and red light.58
Laser Therapy
Laser therapy was previously limited to post acne scarring, but since the early 2000s some lasers have been FDA-approved for active acne lesions demonstrating success in the treatment of inflammatory acne, especially with nodular lesions (p0.05).60 The 1,550 nm erbium glass fractional laser, 1,450 nm diode laser, and 800 nm diode lasers have had promising results in clinical trials.61-63 The most commonly reported side effect included mild erythema of the treated area.60 Gold-plated, silica-core, light-absorbing microparticles with light and laser therapy have recently been an area of special interest because of their antisebum effects. Vacuum-assisted light therapy is also gaining interest for the treatment of acne, claiming to improve light penetration and thus allowing for a better outcome when performed prior to light therapy.
CHEMICAL PEELS
The use of superficial and medium-depth chemical peels for acne is very popular in the dermatological practice because of its favourable results, low-cost, and safety.64,65 Exfoliation, keratolysis, decreased sebum production, anti-inflammatory and antibacterial properties, and comedolytic effects are all mechanisms of action attributed to chemical peels.66 The most popular chemical peels used for acne therapy are salicylic acid, glycolic acid, lactic acid, and trichloroacetic acid. The clinical guidelines for the treatment of acne in 2016 classified them as Grade B with a level of evidence of II–III.9 A special consideration should also be taken for patients undergoing treatment with tetracyclines, birth control pills, and isotretinoin, in whom there may be a higher risk of side effects.
ALTERNATIVE TREATMENTS
In the search for more natural therapy with less side effects, increased use of complementary and alternative medicine, probiotics, and diet have been emerging in society. Such approaches still lack strong evidence-based recommendations. Tea tree oil, green tee derivates, and resveratrol are some of the most common natural products used for acne displaying anti-inflammatory and sebo-suppressive properties.67,68 The clinical guidelines for the treatment of acne in 2016 classified complementary and alternative therapies (tea tree oil, herbal, and biofeedback) as Grade B with level of evidence of II.9 Diet and probiotics have also gained attention for acne treatment.9,69,70 A Cochrane review concluded that a low glycaemic load diet, although only supported by small trials with low-quality evidence, had positive results in the treatment of acne.69 A clinical trial examined skin samples of acne lesions and similarly concluded that the sebaceous glands were smaller and less inflammation was present in patients with a low glycaemic diet, resulting in improvement of their acne.71 Dairy, and dairy-derived products such as whey supplements, are also associated with increased risk of acne. A systematic review and meta-analysis described an increased odds ratio for acne in patients with a high dairy-based diet.72 Evidence-based data is still limited with regards to the impact of diet and acne, and results should be analysed very carefully because of small samples, lack of long-term follow-up, heterogeneity, and bias among studies. Probiotics also play a role in therapy of acne by reducing inflammation, sebum content, and C. acnes colonisation.70
CONCLUSION
Acne is one of the most common medical conditions in the USA and a very common reason of consult in dermatology. Treatment should be guided according to the severity of presentation and the patient’s expectations. Topical and oral antibiotics along with topical retinoids and/or benzoyl peroxide for mild-to-moderate acne, and isotretinoin as the standard of care for severe lesions, are in use to reach remission, but also to prevent scarring and other complications. Hormonal therapy for female patients should be considered with every case, but may not be suitable for all. Alternative or less-used treatments present us with options to refine care for the acne patient, but most often have to be used with more traditional therapies. More research is needed given the existent gaps in knowledge. When to start, which agent to choose, and duration of treatment are often questions both the practitioner and the patient have when considering acne therapy. Treatment challenges for transgender patients, pregnant women, and patients of differing ethnicities; more long-term safety data for systemic agents; and additional options for men are the biggest gaps in the current knowledge. It is mandatory for practitioners to continuously seek for a more efficacious, safe, and cost-effective therapeutic option for their patients.








