Abstract
Hidradenitis suppurativa, or acne inversa, is a chronic, recurrent inflammatory disease of the skin and subcutaneous tissue, and is characterised by painful, recurrent nodules that results in abscesses and formation of chronic draining sinus tracts and scarring. It has been traditionally described as a disease of the apocrine gland. Present evidence, however, suggests it is a disease of the follicular epithelium and the pathogenesis is complex and multifactorial, implicating genetics, microbiome, physiological, and environmental factors. Recently, several cases of apparent hidradenitis suppurativa have been reported in the literature in areas of the body that defy Dessau’s typical topographic criteria. Herein, the authors described an unusual case of a 69-year-old female who presented with hidradenitis suppurativa in her calf post-thermal injury. This case will add to the sparse literature on hidradenitis suppurativa in apocrine-devoid sites and lends credence to the argument that the pathogenesis of hidradenitis suppurativa is multifactorial and complex in nature.
INTRODUCTION
Hidradenitis suppurativa, or acne inversa, is a chronic, recurrent inflammatory disease of the skin and subcutaneous tissue characterised by painful, recurrent nodules that results in abscesses, scarring, and the formation of chronic draining sinus tracts. It was historically described as a disease of the apocrine gland affecting the intertriginous skin of the axillary, inguinal, inframammary, and anogenital region. Present evidence, however, suggests that it is a disease of the follicular epithelium and the pathogenesis is complex and multifactorial, implicating genetic, microbiome, physiological, and environmental factors. Hidradenitis suppurativa is currently diagnosed using the modified Dessau definition that was adopted by the 2nd International Conference on Hidradenitis Suppurativa in San Francisco, California, USA.1 It comprises three criteria:
- Typical lesions: deep-seated painful nodules or blind boils in early stages that may develop into abscesses with draining sinuses, bridging scars, and ‘tombstone’ double-ended pseudo-comedones as disease progresses.
- Typical topography: axillae, groins, perineal and perianal region, buttocks, and infra and intermammary folds.
- Chronicity and recurrences.
All three criteria must be met for establishing the diagnosis, as such the diagnosis of hidradenitis suppurativa is made predominantly on the basis of its typical clinical presentation and there is no pathognomonic test, it still remains a diagnostic challenge for physicians with a reported mean delay to reach diagnosis of 7 years.2 In recent years, several cases of apparent hidradenitis suppurativa have been reported in the literature in areas of the body that defy Dessau’s typical topographic criteria suggesting that rarer variants may exist outside of diagnostic criteria. Presented herein is a report on a case of a 69-year-old female who presented with hidradenitis suppurativa-like lesions in the lower extremity many months post-thermal insult, on a background of limb swelling from chronic venous insufficiency.
CASE REPORT
The patient is a 69-year-old Indian female with no past medical history of note who had previously received treatment elsewhere after scalding her left calf. Her recovery was complicated by recurrent skin and soft tissue infections, requiring 2 months of antibiotic therapy. She was subsequently referred 8 months post-injury to the authors’ service for calf swelling and a presumed chronic venous insufficiency ulcer. The ulcer was 2×3 cm in size, located over the distal third of the left medial calf, moderately deep and sloughy, and had been present since the initial insult. Her ipsilateral dorsalis pedis and posterior tibial pulses were strong, and toe pressures were excellent at 124 mmHg on the right and 133 mmHg on the left. Venous duplex confirmed venous insufficiency of the left long saphenous vein. She was initially scheduled for elective VenaSealTM (Medtronic plc, Minnesota, USA) ablation of her long saphenous vein and wound debridement but returned to the emergency department after a vein in the ulcer edge ruptured with copious bleeding requiring oversewing and admission for more expedient surgery.
During debridement, the ulcer was noted to be unusually deep, nearly 2 cm, for its size (2×1 cm). She was discharged with daily povidone-iodine gauze packing of the wound. Follow-up duplex ultrasound confirmed complete ablation of the long saphenous vein.
The ulcer remained static over the following month; furthermore, at follow-up she had developed a bullous lesion high on the proximal contralateral aspect of her calf which ruptured and discharged clear fluid when expressed. This was deroofed in clinic revealing an underlying 2.5 cm diameter superficial cavity tunnelling caudally for some 15.0 cm. An MRI was performed to exclude osteomyelitis, which revealed an unusual horseshoe-shaped tract within the subcutaneous fat starting at the superolateral wound above, tunnelling caudally then making a turn to tunnel transversely across the posterior calf to the medial aspect of the calf, then ascending superiorly to exit at the original wound. There was no evidence of osteomyelitis (Figure 1). She underwent formal operative deroofing and debridement of the entire tract. Intraoperatively, a long, 1.5cm deep, subcutaneous tract that was approximately 0.7–0.8 cm wide was found connecting both wounds in continuity (Figure 2), which filled with pink amorphous gelatinous tissue highly reminiscent of the invasive proliferative gelatinous mass (IPGM) found in hidradenitis suppurativa wounds.
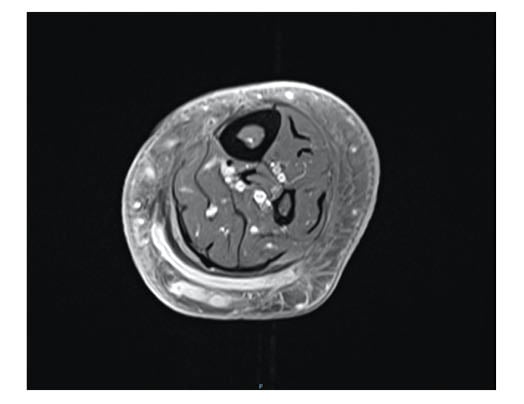
Figure 1: Axial T1-weighted MR image showing a sinus tract that crosses the midline to the lateral aspect of the left calf.
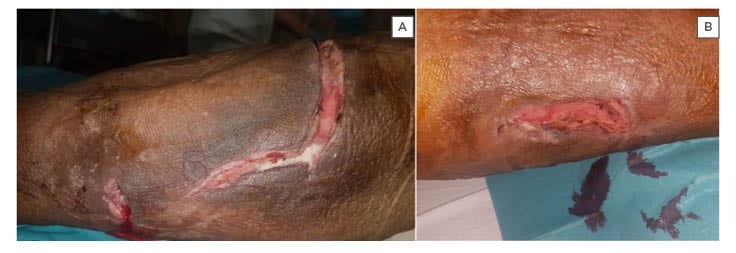
Figure 2: A) lateral calf wound in continuity with B) the medial calf wound via a posterior tract ascending superiorly up the calf towards the knee and then transversely across the shin where it had ruptured superficially.
Aerobic tissue culture grew group B beta-haemolytic strep (Streptococcus agalactiae) and Staphylococcus aureus. Acid-fast bacilli smear was negative. Histopathology of the tissue reported ulceration with granulation tissue formation, mixed inflammatory infiltrates, and reactive nuclear changes with hyperkeratosis of the epithelium, in keeping with previous descriptions of hidradenitis suppurativa.3 There was no evidence of dysplasia or invasive malignancy. The wounds were treated with Vacuum Assisted Closure® (VAC) therapy in combination with Grade 2 graduated compression stockings. A month later, the wounds had granulated flesh and VAC therapy had been ceased (Figure 3).
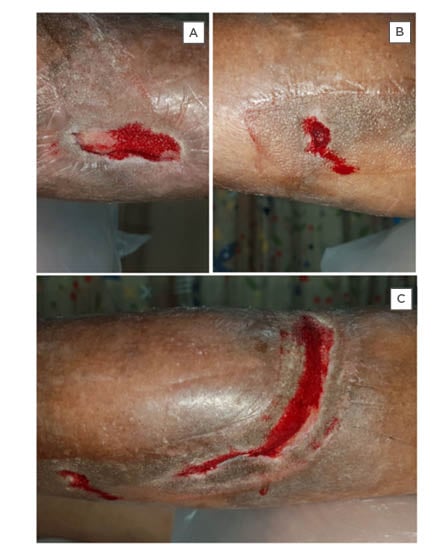
Figure 3: A) medial shin wound; B) lower lateral shin wound; C) and ascending calf wound across the shin, granulated nearly flush with sealing of the posterior tunnel.
DISCUSSION
Hidradenitis suppurativa was historically assumed to be attributable to entrapment of secretions from apocrine glands with secondary bacterial infection and the intertriginous areas are commonly affected. It is postulated that these sites predispose to friction, shearing forces, and pressure or other physical irritation from clothing and undergarments.4 In recent decades, contemporary opinion has shifted and it is now believed that hidradenitis suppurativa is caused by inflammatory obstruction of the follicular portion of the folliculopilosebaceous unit (FPSU) in patients with a predisposition to rupture of the FPSU.5,6 To date, only three cases of hidradenitis suppurativa of the lower extremity have been reported in the literature, making this an unusual presentation in a site devoid of apocrine glands.3,4,7 One of the cases described was hidradenitis suppurativa of a lower limb amputation stump, which the authors postulated was attributable to recurrent mechanical friction against the ambulatory prosthesis resulting in follicular trauma and secondary bacterial infection.7 None of the previous cases detailed scalding as the precipitant. In the authors’ opinion, the initial thermal injury directly damaged the FPSU at the site of contact of her medial calf causing follicular occlusion which in turn triggered the chronic inflammatory process leading to the spreading IPGM-filled sinus tract across her posterior calf and ascending up the contralateral side of her calf where it subsequently ruptured through the relatively thin skin of her shin. This hypothesis is consistent with the three-stage sequence of events in the pathogenesis of hidradenitis suppurativa proposed by Vossen et al.,8 who suggested that the initial event is triggered by follicular occlusion and dilation, which is driven by endogenous and exogenous factors. This is followed by rupture of the dilated follicle, triggering a cascade of inflammatory pathways. The third event is chronic inflammation with development of sinus tract resulting from the presence of epithelial strands, imbalance between matrix metalloproteinases and tissue inhibitors of metalloproteinase, and elevated activity of tissue growth factor.8
CONCLUSION
This case adds to the sparse literature of hidradenitis suppurativa in apocrine-devoid sites and lends credence to the argument that the pathogenesis of hidradenitis suppurativa is multifactorial and complex in nature. In this current case, the lesion was successfully treated using VAC therapy in conjunction with therapies, VenaSeal, and graduated compression stockings, to manage tissue swelling from chronic venous insufficiency.