Abstract
This article aims to help non-dermatologist medical professionals regarding the current status of photoprotection so that they may be better positioned to advise and respond to their patients. While the effects of solar radiation have long been known to include sunburn and skin cancers derived from ultraviolet B radiation, advances in knowledge now recognise the relevance of ultraviolet A, visible, and infrared light as significant contributors to skin damage. Effects on the skin range from aesthetic signs of photoageing, which accumulate with daily exposure, to skin cancers. Despite some trends towards increased awareness of the dangers of solar radiation and the need for photoprotection, behaviours still put people at risk and sun protection is suboptimal. In addition to the general population, certain population groups require special consideration depending on their work environment, lifestyle, and health status. The efficacy and cosmetic properties of sunscreens have improved greatly and should help to improve compliance with recommended use, but a multifaceted approach focussed on education and enabling uptake of recommendations is essential.
INTRODUCTION
Cumulative or excessive solar exposure is detrimental to skin health.1 Furthermore, certain population groups require extra care. This article is aimed at general medical practitioners who may be called upon to advise their patients on photoprotection. It provides a rationale for photoprotection based on the effects of solar radiation on the skin and the current status of associated skin pathology, highlights aspects of behaviour that may limit adherence to recommendations, looks at some of the key regulations surrounding sunscreens and how they work, and draws attention to special patient groups, including considerations for their management.
EFFECTS OF SOLAR RADIATION ON SKIN
Sunlight is essential for vitamin D synthesis in the human body, which plays a role in bone density and immune function;2 however, uncontrolled exposure, especially over years or decades, leads to photoageing, immunosuppression, skin cancer, and exacerbation of photodermatoses. To understand the precepts of sun protection, it is helpful to understand the components of the solar spectrum and how they affect skin. Solar radiation is generally divided into ultraviolet radiation (UVR), consisting of UVA (at wavelengths of 320–400 nm) and UVB (290–320 nm), as well as UVC (200–290 nm), visible light (400–700 nm), and infrared (IR) (700–1,000 nm). The solar spectrum at the Earth’s surface is limited to wavelengths between 290 and 3,000 nm because UVC is completely absorbed by stratospheric ozone.3 The intensity of UVR reaching the skin depends on factors such as latitude, altitude, season, cloud cover, and time of day.4 UVB exposure is associated with some potent changes in the human body; it is mainly responsible for sunburn (its erythemogenic effect is 1,000-fold greater than short wave UVA),5 and can induce skin cancers (by causing direct DNA damage and covalent bonding between pyrimidine bases), immunosuppression, skin darkening, and ageing.6 Consequently, early sunscreens were designed almost exclusively to protect from UVB. However, it was later demonstrated that both UVA and UVB are causative agents in skin cancers, with UVA acting indirectly mainly by triggering production of reactive oxygen species.7,8 This same mechanism also provides an explanation of UVA as the main contributor to skin photoageing. More recently, the effects of visible light, including erythema, pigmentation, and radical production, have garnered much attention.4,9 Given the multiple detrimental effects of solar radiation, it is easy to understand why photoprotection is an important preventative health strategy and why the approaches to this increasingly include protection beyond the UV range.
CURRENT STATUS: SKIN CANCER AND SUN BEHAVIOUR
Skin Cancer in Europe
Global incidence rates of melanoma and nonmelanoma skin cancer (NMSC) continue to increase. Worldwide, the highest rates are reported in Australia and New Zealand; within Europe, northern European countries see the highest incidence at 23.9 per 100,000 in Sweden (2012) versus 13.0 per 100,000 for Europe.10 NMSC, which may be excluded from or incompletely recorded in registries, is more challenging to quantify, but incidence rates for basal cell carcinoma (BCC) of 90–129 per 100,000 person years, European standard, are described.10 The majority (99%) of NMSC is BCC and squamous cell carcinoma (SCC), BCC being approximately 3–4 times more common than SCC.10
While history of episodic sunburn is associated with increased risk of melanoma, it is thought that cumulative solar exposure is key in the pathogenesis of NMSC.10 NMSC occurs in up to one-third of outdoor workers compared with only 5% of office workers.11 NMSC has a low metastatic potential and mortality rate, but still has a high burden of morbidity and cost.10,12 Because increased age is a risk factor for NMSC, it seems likely that increased rates of the disease will accompany the ageing population. Up to 90% of skin cancers are related to UVR; consequently, UV exposure remains the most important modifiable risk factor in preventing skin cancer.6,10,13
Sun Protection Awareness and Behaviour
Despite overall increasing skin cancer rates (in 2018, approximately 300,000 new cases of melanoma and over 1 million cases of NMSC were diagnosed worldwide),14,15 some countries lead the way in preventative health education, demonstrating that such education can indeed prove effective. Going against the trend, Australia has successfully managed a recent decrease in melanoma incidence, and a similar downtrend is anticipated for New Zealand.10,16 This follows multiple initiatives from their cancer councils over the past 35 years, including early childhood programmes to increase public awareness and improve sun safety (use of protective clothing and hats, adequate sunscreen use, and avoidance of excessive exposure).17,18
General recommendations from the World Health Organization (WHO)1 regarding sun protection are to limit midday sun exposure (from 10 am to 4 pm), to seek shade (particularly during midday hours), to consider the UV index when planning activities, to use protective clothing, to wear a wide-brimmed hat and sunglasses (with 99–100% UVA and UVB protection), and to use broad-spectrum minimum sun protection factor (SPF) 15+ sunscreen liberally applied at 2-hour intervals, or after swimming or exercising. Artificial sun lamp and sun bed use should be absolutely discouraged and patients should be informed of the risk: these are categorised as Group 1 carcinogens.1,19 Interestingly, sunbed use, while highest in northern European countries with low levels of sun, is also paradoxically high in Spain and Italy; practitioners in these countries should not therefore assume that such advice does not apply to their population.20 The European Skin Cancer Foundation’s (ESCF)21 recommendations do not differ greatly from those of the WHO: the ECSF suggests an SPF of at least 25, plus high UVA protection, applied 20–30 minutes
before exposure.
Thus, multiple behavioural modifications are recommended, and modifying long-term behaviour is a complex task. Sunscreens form just one component of sun protection; they should not be considered sufficient protection on their own but used as one of multiple methods to protect the skin. However, they are often the main, or only, form of protection used. Modern sunscreens can provide high protection levels and are available in many formats, but despite these advances, data on the actual use of sunscreens also paints a rather unsatisfactory picture. While some studies show improvements over time, most of them demonstrate persistent misunderstandings and inadequate behaviours. A report by Cancer Research UK®22 found several significant positive trends in sun protection behaviours between 2003/2008 and 2013. The most notable improvements were in covering up (an estimated 30% of the population reported this behaviour) and using factor 15+ sunscreen (an estimated 50% of the population reported this behaviour). A study comparing university students in 2000 versus 1990 in 13 European countries found that the proportion using sun protection increased over the decade studied, with men showing a greater increase (but starting from a lower level), while women remained more likely than men to use sunscreen.23 In a Welsh study by Jackson et al.,24 despite increased knowledge, subjects with a past history or family history of melanoma did not have safer sun behaviour. Even in a private dermatology clinic in the USA a significant proportion of patients, including patients with skin cancer, did not understand the risk associated with sunlight and stated they had not received counselling on the subject.25
A Spanish study of beachgoers found that, despite reported use of high-factor sunscreens, 70% of individuals interviewed reported a history of sunburn.26 Importantly, they reported false beliefs about sunscreen safety leading to longer sun exposure behaviours. It should also be borne in mind that when sunscreens are used, the rules of applying 2 mg/cm2, the quantity needed to achieve a homogeneous film at the surface of the skin, and reapplying every 2 hours, are often forgotten by users. This was found in a Danish study in which only around one-quarter of the recommended amount of sunscreen was applied to the whole body.27
Motivations for sun-seeking or unsafe behaviour range from aesthetic reasons such as ‘looking better with a tan,’26 to inconvenience,28 which can relate to the cosmetic properties of sunscreens: they are often sticky, greasy, or leave residues, particularly at high SPF. Such persistent misconceptions and insufficient protective behaviour indicate that public educational strategies and physician education of patients are essential. Photoprotection must be multifaceted; it should be emphasised to patients that sunscreen use does not justify otherwise unsafe behaviour and that avoidance of midday sun or prolonged exposure and wearing protective clothing, including wide-brimmed hats and glasses, should be viewed as highly important actions.
SUNSCREEN PRODUCTS: COMPOSITION AND PROPERTIES
Traditionally, sunscreens aimed simply to prevent sunburn at isolated exposures. Nowadays, in light of knowledge regarding the effects of chronic sun exposure, including UVA and its role in skin ageing, much more comprehensive protection is recommended. Consistent, everyday use is prudent, and sun filters, their vehicles, and additional ingredients are constantly being innovated to go beyond ‘just’ preventing sunburn, with the inclusion of antioxidants, DNA repair enzymes,29 or skin hydrating agents. In Europe, most sunscreens are classified as cosmetics under regulation (EC) No 1223/2009.30 For cases in which clinical conditions are targeted, they may be classified as medical devices and undergo clinical trials as such.29
Sunscreens contain sun filters: molecules that absorb, reflect, or scatter solar radiation, limiting the quantity of radiation that reaches the skin. As a result of no single agent providing full solar protection, products are usually composed of a mixture of sun filters to cover the largest part of the UV spectrum. Filters can be broadly divided into organic/chemical and inorganic/physical filters (Table 1).
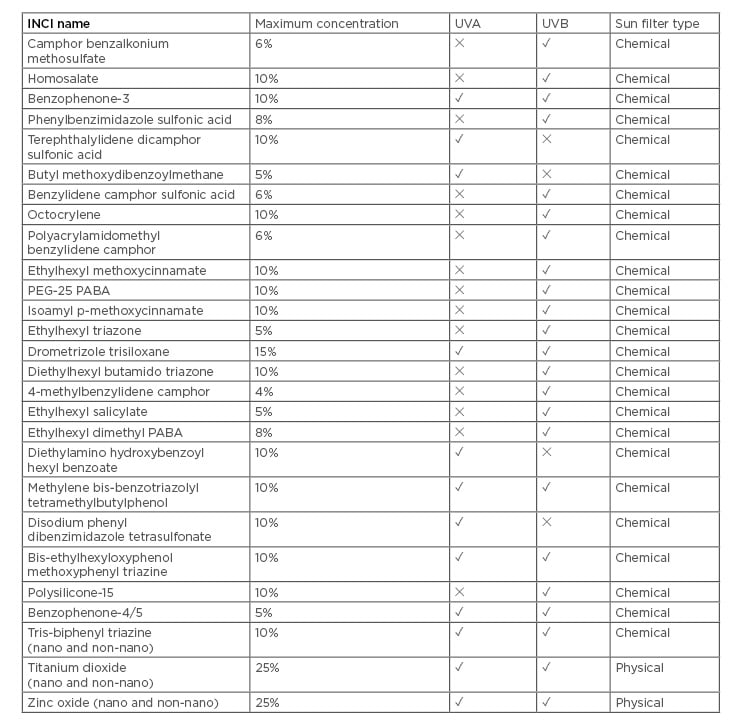
Table 1: Details of sun filters approved for use in Europe.
INCI: international nomenclature of cosmetic ingredients; PABA: para-aminobenzoic acid; PEG: polyethylene glycol; UV: ultraviolet.
Historically, organic filters were thought to absorb UVR, and inorganic to reflect and scatter it. However, recently it was confirmed that titanium dioxide and zinc oxide protect primarily via absorption of UVR and not through significant reflection or scattering. For these two inorganic sunscreens, the scattering and reflection increases in the visible part of the spectrum, which is why sunscreens formulated with these ingredients may leave a white appearance on the skin.31 Another difference between the two classes is that inorganic filters, even in nanoparticles, have not been shown to permeate the skin, whereas organic filters can cross the skin barrier and have been found at low levels in the systemic circulation.32 It should however be stressed that, after decades of use, sunscreens have not been demonstrated to adversely impact human health.6,33
Sunscreen efficacy is assessed in validated standardised tests and is usually described as the SPF value. The concept of SPF is used worldwide as an in vivo measure of the ability of a sunscreen to prevent sunburn (erythema, mainly caused by UVB): the SPF is defined as the ratio of [least amount of UVR required to produce minimal erythema on sunscreen-protected skin] to [amount of UVR required to produce the same minimal erythema on unprotected skin] (International Organization Standardization [ISO] 24444: 2019).34 Other non-erythema-based sun protection factors have been proposed,35-7 and some may form new ISO-approved testing methods in the near future.38,39 In vitro methods of SPF testing may also come into use,40 which would avoid the inherent disadvantages of human volunteers, including the ethical aspects of inducing sunburn and practical aspects such as time required. Currently, SPF value and the corresponding sun protection level (very high: SPF 50+; high: SPF 30–50; medium: SPF 15–25; or low: <SPF 15) remains the best index to communicate the protection level of a sunscreen to consumers, even if the conditions under which it is measured cannot fully reflect actual use. In Europe, the UVA protection factor can be measured in vivo by ISO 2444241 or in vitro by ISO 24443.42 This protection factor should be at least one-third of the SPF value; if this threshold is met, ‘UVA’ is written inside a circle on the packaging.43,44
While discussing ways to increase the uptake of sun protective behaviours, it is pertinent to mention the concerns of some users regarding the impact of sunscreen use on their vitamin D status. Since endogenous vitamin D synthesis requires skin being exposed to UVB, the logical question raised is whether vitamin D levels are affected by sunscreen use. However, latest research published indicates that there is no evidence of this in practice. An expert review of the literature concluded that “sunscreen use for daily and recreational photoprotection does not compromise vitamin D synthesis, even when applied under optimal conditions.”1
SPECIAL SITUATIONS AND POPULATION GROUPS TO CONSIDER
In addition to the general recommendations, it is important to tailor these to the individual, in terms of both behavioural modifications and appropriate sunscreen products. Depending on their specific situation and health status, certain factors should be taken into consideration because health beliefs and behaviours are complex and may relate to perceived risks and benefits.26 These are, in turn, influenced by factors such as the individual’s medical history, family history, exposure to educational materials, or educational level.24,28 For the purposes of this review, special population groups can broadly be categorised into five groups: 1) those undertaking recreational, acute, and intermittent high-to-extreme UV exposure (e.g., at the beach or skiing) where the user principally wants to be protected from sunburn; 2) daily photoprotection in a skincare routine in which chronic damage and photoageing prevention is the main driver; 3) skin diseases aggravated by sunlight where the patient wants to prevent flares; 4) immunocompromised patients; and 5) occupational exposure in outdoor workers with the objective of preventing skin cancers.
Prevention of Sunburn
Limitation of exposure is key, which may include adapting planned activities according to time of day and UV index. When advising or deciding on a sunscreen, taking into consideration that UVB is the main causative agent for solar erythema, SPF is the most relevant indicator because it is directly indicative of sunburn protection. The importance of high SPF has been observed in recent studies in outdoor extreme conditions, which found that SPF 100 sunscreens provided more protection than SPF 50 products under these extreme conditions.45,46 Importantly, for individuals with fair skin, even very high SPF (50+) may offer insufficient protection in conditions of very-high-to-extreme UVR, confirming that sunscreens should not be the only photoprotection strategy used.47
Prevention of Skin Ageing
Photoageing results from repeated UVR with subsequent reactive oxygen species production and activation of matrix metalloproteinases. Signs include skin roughness and dryness, wrinkles, and uneven pigmentation and telangiectasia, usually on the face, neck, chest, and dorsal hands. For such daily use, an SPF of 30 may be considered sufficient. Good UVA protection is needed to prevent photoageing, and finding a formulation that is pleasant to use may take priority over high SPF values if it is likely to result in regular use. The combination of sun filters and antioxidants such as vitamins (C and E, niacinamide), polyphenols, or flavonoids, have additive effects in reducing the concentration of free radicals in the skin.48,49 Such details relate to sunscreen use, and daily habits should also be addressed.
Photoprotection in Photodermatoses
Photodermatoses represent a heterogeneous group of diseases with an abnormal cutaneous reaction to sunlight. Photoprotection is a key element of their management and selection of the most appropriate sunscreen usually depends on the identification of the wavelengths responsible for inducing the disease. Polymorphic light eruption, the most common photodermatosis with a prevalence of 10–20% in the general population,50,51 and lupus erythematosus, the most common photoaggravated dermatosis,51,52 are triggered by UVA and UVB; thus, in addition to protection with clothing, exposed areas require a broad-spectrum sunscreen with high SPF and high UVA protection.
In subjects with pigmentary disorders such as melasma, the deleterious role of visible light and particularly its blue component has been confirmed:53 daily use of a broad-spectrum sunscreen including visible light protection is essential. Iron-oxide-containing sunscreens (tinted sunscreens) have been shown to absorb high energy visible (HEV), the short wavelengths of the visible light spectrum, and help prevent the pigmentary effect of this part of visible light.38,54
Immunocompromised Patients
Organ transplant recipients represent a high-risk group for skin cancers as a result of their post-transplant immunosuppressive therapy.55 Other diseases requiring immunosuppressive therapies, such as inflammatory bowel disease, have also been identified as having increased risk of skin cancer.56,57 The risk of SCC may be increased several hundred-fold in transplant recipients,58 and tumours may behave more aggressively.58 Type and level of immunosuppression play a role in the incidence of skin cancer.58 Strict sun avoidance and use of very high SPF products is essential; consequently, vitamin D supplementation may be required.
Children represent a population group with a physiologically immature immune system, and also generally spend a greater amount of time outdoors, therefore requiring a careful approach to photoprotection and reliance on adults to enforce it.59,60 A pleasant-to-use, water-resistant, and rub-proof sunscreen formulation may provide a practical improvement to photoprotection in real-life use.
Outdoor Workers
Despite the fact that a high number of outdoor workers worldwide are exposed to UVR for the majority of their working life, as well as the existing literature on NMSC risk factors, solar exposure risk remains undervalued as an occupational risk factor61 and skin cancers are scarcely reported as occupational disease.11,62 Peters et al.63 estimated that in 2011 in Canada, 6.3% of NMSC cases were attributable to occupational exposure to UVR. Yet, awareness of prevention strategies recommended by health authorities remains low among these high-risk groups.3,64
Education on protective clothing, whether something does or does not constitute protection, and addressing common misconceptions (for example, a so-called ‘protective’ tan) are fundamental.
The SPF recommendation should be as high as possible: at least SPF 50+. Perceived barriers to occupational sunscreen use include taking too long to apply, causing discomfort or irritation, and financial expense.28 Addressing barriers such as cost, messy application, unpleasant after-feel, and stinging of the eyes may therefore improve compliance.65 Another key difficulty is achieving 2-hourly reapplication, as this is not always feasible.
In the near future, preventative interventions should include collective measures such as specific legislation, workers’ education, and training; individual measures such as personal protective equipment including sunscreens; and health surveillance of outdoor workers.66 The benefits of such formal and informal workplace initiatives are likely to outweigh the costs and should be borne in mind for workplace policy decision-makers.
Conclusions and Future Areas of Study
Effective photoprotection is best achieved by exposure avoidance: seeking shade, use of protective clothes and glasses, avoiding midday sun, and appropriate use of sunscreens. Photoprotection is a key preventative health strategy as most skin cancers are a result of UVR exposure. Yet, uptake of photoprotective measures remains insufficient and inconsistent. Current limitations do not stem from a lack of efficacy of existing methods or products, but rather from adherence to use and misconceptions or inconveniences leading to unsafe behaviours.67
Everyday sun protection is advisable to the general public, but some groups are at increased risk. Tailoring advice to individual situations and addressing barriers to compliance are likely to contribute to better usage. Widespread cultural changes appear to still be somewhat in their beginnings, with some apparent improvements in knowledge that are not yet accompanied by the corresponding behaviours. Initiatives to normalise sun-safe behaviour are required to protect against skin ageing, chronic dermatoses, and, most importantly, skin cancers. Clear examples of successful educational and nationwide campaigns are available, and medical practitioners have a significant role to play in protecting vulnerable groups and empowering and educating their patient population.