Abstract
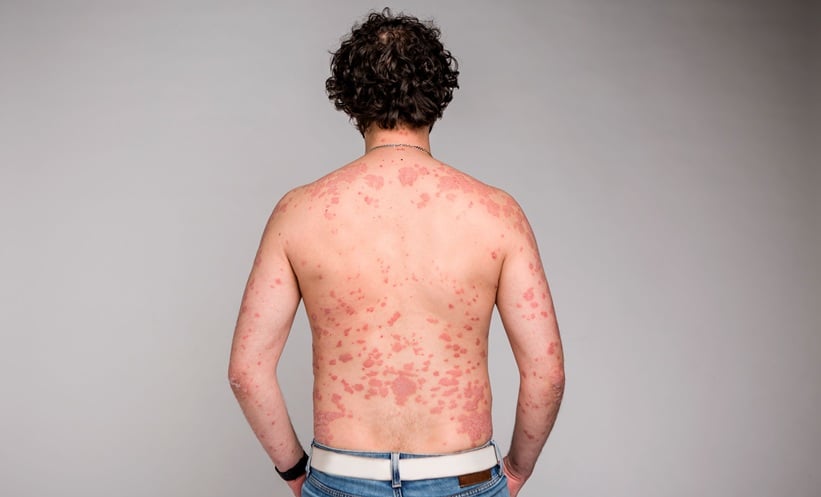
Atopic dermatitis (AD) is a highly pruritic, chronic inflammatory skin disease that affects up to 25% of children and 10% of adults. Approximately 90% of patients with AD are colonised by Staphylococcus aureus , compared with only 5–30% of non-atopic individuals. Th2 cytokines have a permissive effect on microbial invasion, the epidermal barrier, and cell-mediated immunity, which lowers the production of antimicrobial proteins. Superantigen-producing S. aureus colonisation is correlated with serum interleukin (IL)-4 levels. Up to 50–60% of the S. aureus found on patients with AD is toxin-producing.1 S. aureus colonisation, infection, and production of toxins and superantigens is believed to drive, at least in part, the pathogenesis of AD. S. aureus mechanically disrupts epidermal integrity through protease activity, and also has the ability to be internalised by keratinocytes in which it activates the inflammasome and induces apoptosis. Some patients with AD produce specific immunoglobulin E (IgE) antibodies directed against staphylococcal superantigens to an extent that correlates with skin disease severity. IL-4 and IL-13 have also been reported to increase staphylococcal α-toxin-induced keratinocyte death via STAT6 signalling. The S. aureus superantigens staphylococcal enterotoxin B and toxic shock syndrome toxin 1 promote lymphocyte IL-31 production in patients with AD. IL-31 has, in turn, been shown to reduce filaggrin expression and mediate pro-inflammatory cytokine excretion, as well as induce toxin-specific IgE and basophilic activation. The ability of S. aureus to colonise skin affected by AD, and to activate and maintain a Th2 environment allowing, via the destruction of tight junctions, exposure to allergens and thus causing allergic sensitisation, makes it one of the main protagonists of the ‘atopic march’.
Please vie the full content in the pdf above.