Meeting Summary
In the first of two sessions from the 31st European Academy of Dermatology and Venereology (EADV) Congress, Thierry Passeron, University of Nice Sophia Antipolis, France, presented a large international survey that showed that while people are aware of the dangers of sun exposure, many still seek to tan and do not use or may not understand protective measures. In the second half of this session, Susana Puig, University of Barcelona, Spain, discussed data on the use of a high factor, broad-spectrum sunscreen combined with nicotinamide and panthenol. In people with photodamaged skin, the cream was shown to downregulate genes involved in apoptosis, the immune system, and cancer and upregulate collagen genes. In people with actinic keratosis (AK) and its precursor, field cancerisation (FC), treatment led to downregulation of genes involved in a number of signalling pathways, including those implicated in cancer, the cell cycle, and apoptosis. In the second session, Jean-Michel Amici, Université Bordeaux, France, discussed the wound healing process and the role of the immune system and skin microbiome. Delphine Kerob, La Roche-Posay Laboratoires, Paris, France, then presented data from a survey of people with scars that revealed how redness, itching, pain, and burning could persist for at least a year in some people, and skin discomfort can impact quality of life (QoL). Kerob also presented findings from recent studies of effects of the cream Cicaplast Baume B5+® (La Roche-Posay, L’Oréal UK Ltd, London, UK). Cicaplast Baume B5+, which contains a prebiotic complex as well as panthenol, madecassoside, and moisturisers, showed positive effects on skin healing following laser treatment for skin damage.
Introduction
Sun exposure can lead to tanning, but also to skin damage due to the actions of ultraviolet A and B, visible light, and infrared radiation.1,2 One result of sun exposure is field cancerisation (FC), the precursor to actinic keratosis (AK), which in turn is a risk factor for, most notably, squamous cell carcinoma.3 Daily sunscreen use can prevent the development of AK, decrease pre-existing AK, improve FC, and help prevent skin cancer.4,5
Insights From a Large International Study of Sun Exposure and Associated Risks
Thierry Passeron
To gain insight into how people view sun exposure, Passeron presented a survey conducted in 17 countries spanning all but the polar continents. This included 17,001 adults, predominantly Fitzpatrick skin phototypes II (fair skin, blue eyes, tans poorly, burns easily) and III (darker white skin that tans after initially burning), but spanning all phototypes.6
While the majority of respondents agreed with statements saying that sun exposure can cause skin health problems and accelerate skin aging, they also agreed that the sun helps them synthesise vitamin D, gives them energy, and that a tan makes a person look healthy and attractive. Three-quarters also never or very infrequently had moles checked by a dermatologist.
Protection from the sun can include using a suitable sunscreen, covering the body with clothing and headwear, and staying in the shade.7,8 Figure 1 shows that few people systematically use such protection, with some people never doing so.
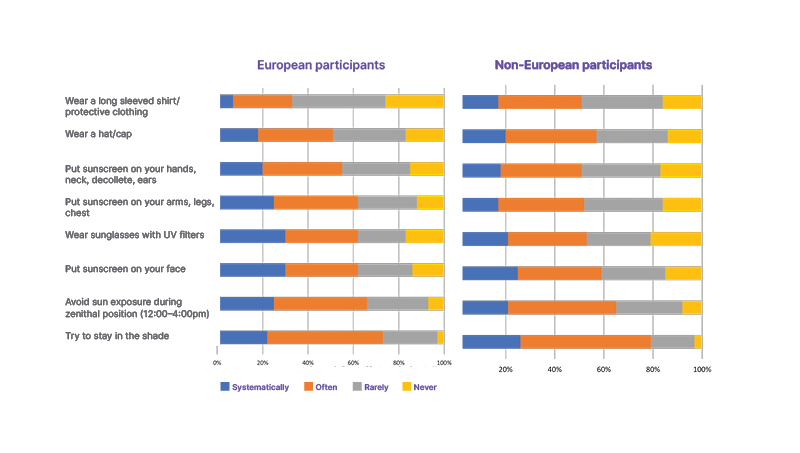
Figure 1: European and non-European habits with regard to sun protection.6
UV: ultraviolet.
Overall, only 10% of Europeans and 14% of non-Europeans were found to systematically or regularly undertake all the protective measures shown in Figure 1. This rose to only 21% in a cohort of 655 Europeans considered at risk to sun exposure as a result of skin conditions such as skin cancer and photodermatoses, or who were using a sun-limiting medication.
Sunscreen should be used regularly, year round, every 2−3 hours, even on cloudy days.8 In this survey, most respondents only used sun protection when on vacation, two-thirds said they sought to tan with sun protection, a quarter believed that sun exposure without sunscreen is safe if they are already tanned, and many thought that sunscreen with sun protection factor (SPF) ≥50 is only for those who are at risk from sun exposure.
Additionally, it was found that two-thirds of Europeans and three-quarters of non-Europeans only applied sunscreen once or twice a day, and many did not use sun protection on cloudy days. It was also found that between one-half and three-quarters of respondents did not understand the differences in the various wavelengths of electromagnetic radiation that can affect the skin, or in SPF values.1,2
Passeron concluded by stating: “We have a lot of work to do […] as most individuals are not protecting themselves and don’t care most of the time.”
A Sunscreen Formula for Photodamage and Actinic Keratosis
Susana Puig
Puig presented a study investigating whether a high factor, broad-spectrum sunscreen combined with nicotinamide and panthenol could repair skin photodamage. Fourteen adults (phototypes II–III) applied the sunscreen daily to photodamaged skin on the external forearm and ‘healthy’ non-photodamaged skin on the internal forearm for 4 weeks. Biopsies were taken at baseline and study end.
After 4 weeks of use, small, non-significant decreases were found in measures of stratum corneum integrity, epidermal thickness (plus or minus the stratum corneum), dermal–epidermal junction undulation, and keratinocyte quantification. Analysis of cell damage markers in the biopsies also showed small, non-significant decreases.
Pre-treatment RNA sequencing revealed that 1,552 genes were significantly differentially expressed between healthy and photodamaged skin. Genes significantly upregulated in photodamaged skin compared with healthy skin are shown in Table 1, and include those involved in collagen degradation and apoptosis.9,10 Comparing pre- to post-sunscreen exposure in photodamaged skin showed that 5,429 genes were significantly differently expressed. Pathways that were significantly downregulated are shown in Table 1 and include those involved in the cell cycle, apoptosis, the immune system, cell communication, cellular energy, and cancer.11-13 Significant upregulation was shown in a number of collagen genes.
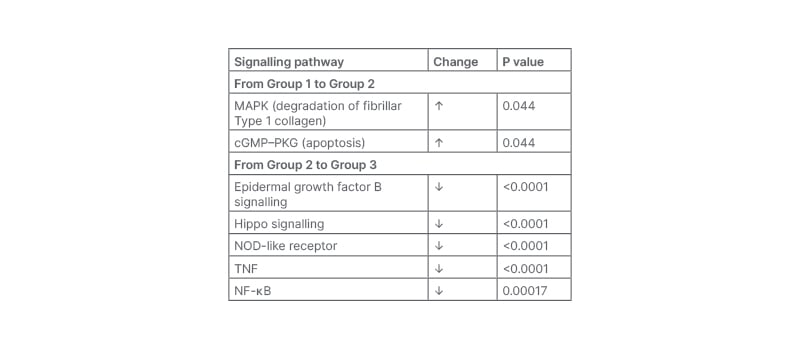
Table 1: RNA sequencing analysis of gene expression10
Group 1: healthy samples; Group 2: lesion (photodamage) pre-treatment samples; Group 3: lesion (photodamage) post-treatment samples.
cGMP-PKG: cyclic guanosine monophosphate–protein kinase G; MAPK: mitogen-activated protein kinase; NF-κB: nuclear factor κ-light-chain-enhancer of activated B cells; NOD: nucleotide-binding oligomerisation domain.
A second study evaluated the effects of the sunscreen for repairing AK and FC in 20 patients who applied the cream for 8 weeks. From the data obtained of differences between FC and AK at pre- and post-treatment, a model was developed whereby AK progression from FC includes increases in stratum corneum and living epidermal thickness; keratinocyte layer number, volume, cytoplasm, and atypia; and dermal–epidermal junction undulation. Treatment reduced living epidermis thickness and number of keratinocyte layers in the AK areas and average volume and variability of keratinocyte nuclei in the FC areas.
AK and FC biopsy analysis showed significant decreases in expression of the cell cycle protein p21 and increases in thymine dimers following treatment. Skin damage that was previously present was also reduced. RNA sequencing data showed 1,555 differently expressed genes in the AK samples and 40 in the FC cases. Pathways downregulated in AK samples post-treatment included those involved in p53 signalling, cell communication, proteoglycans associated with cancer, apoptosis, muscle contraction, and sphingolipids. Pathways downregulated in both AK and FC included those involved in cancer, cell cycle, and glucose ho meostasis.14,15
Puig concluded that “high broad spectrum photoprotection is able to reverse some photodamage and modulate DNA repair and carcinogenesis genes.”
The Process of Wound Healing and the Role of the Microbiome
Jean-Michel Amici
Amici discussed the four main stages of wound healing. Initially, haemostasis involves vasoconstriction and the formation of a fibrin clot to control bleeding and provide a scaffold for immune cells.16,17 Next is inflammation, involving recruitment and activation of phagocytic cells to sterilise the wound and clear the site,18 and macrophages to engulf apoptotic and necrotic cells and pathogens.16 The degree of response at this stage can impact the following stages.18
The next stage is proliferation of keratinocytes, endothelial cells, and fibroblasts. These cells help in formation of granulation tissue,19 wound re-epithelialisation,18,19 and myofibroblast accumulation, which contracts the wound to decrease surface area.18 At 2−3 weeks post-injury, granulation tissue has formed, covered with new epithelial cells.20 The final stage, remodelling, can last for over a year.21 Here, collagen is realigned to form an organised network and there is extensive extracellular matrix remodelling.18 Also taking place is apoptosis of endothelial cells, fibroblasts, and macrophages.22 The result of this process is collagenous scar formation that increases the overall strength of the repaired skin.18
During wound healing, the co-ordinated response involves adaptive immunity, which can help fight infection and is essential for proper wound resolution and innate immunity, involving pathogen response and the production of antimicrobial peptides (AMPs). Two major AMPs, cathelicidins and defensins, are produced by keratinocytes and aid in modulating the innate immune response and activating adaptive immunity.23
The skin microbiome includes commensal micro-organisms in the form of bacteria (including Staphylococcus, Corynebacterium, and Cutibacterium species), mites, and fungi. It helps maintain skin acidification, create a hostile environment for pathogens, and in induction of host AMPs and antimicrobial molecules.23-26 For example, Staphylococcus epidermidis can activate toll-like receptor-2 signalling to induce keratinocyte AMP expression.23,27,28 In wound healing, S. epidermidis can induce CD8+ T-cells, triggering the upregulation of immune regulation and tissue repair-related genes, improving re-epithelisation of affected skin and accelerating wound repair.29-31
Many factors can influence wound healing, including the wound’s cause and location and a person’s age, phototype, nutrition status, and comorbidities. There can also be complications, such as atrophic, hypertrophic, and keloid scars; chronic wound occurrence; and superinfection.32
In Amici’s view, what is needed to improve wound healing is a balanced skin microbiome to limit pathogen colonisation and increase AMP production; control of inflammation to avoid post-inflammatory hyperpigmentation and abnormal scarring; and a synergistic relationship between the immune system and the skin microbiome to help accelerate wound repair and improve re-epithelialisation.
The Impact of Scars
Delphine Kerob
Symptoms associated with wound healing, discussed Kerob, include pain, itching, bruising, inflammation, erythema, desquamation, and scab formation. Supportive measures for managing scars for an optical outcome include basic wound cleaning, healing optimisation, photoprotection, and corrective makeup to help protect healing skin and improve QoL during scar formation.33
A large international epidemiological survey, involving 11,100 adults (18−74 years) from China, Brazil, the USA, Russia, and France, was conducted to assess the impact of scars on a patient.34 Respondents included an almost equal number of males and females, predominantly phototypes III−IV, but with the inclusion of all phototypes. About half of respondents had a scar due to an accident, illness, or surgery, with 78% having them for >1 year (n=4,186).34
Overall, 44% of respondents with newer scars (<1 year) reported redness, 35% itching, 29% pain, and 24% burning. While at significantly lower levels, these sensations were still reported by people with scars <1 year old, with respective values of 19%, 15%, 12%, and 10%. However, few people used any treatment for scar management, with only 11−13% prescribed a healing cream, antibiotic ointment, or antiseptic solution and 5−11% using a repairing cream, moisturising cream, massage, or scar camouflage product. Additionally, only 31% of respondents said they protected the scar from the sun, with only 47% using an SPF sunscreen. Of these, most used one with a high (29%) or very high (47%) SPF.34
This survey also found that having a scar can impact a person’s QoL, with scores on a measure of such higher in the first few months, then decreasing after 1 year, though, for some, still showing some QoL impacts at this time. In respondents that reported skin discomfort, their symptoms significantly impacted several aspects of life, including leisure activities, clothing choice, and intimacy, compared with those without skin discomfort.
Kerob concluded that “understanding the process of scarring is key to recommending proper skin care and improving outcomes.”
Wound Healing Management
Kerob also presented several studies regarding the effects of the topical cream Cicaplast Baume B5+, which contains Tribioma, a prebiotic complex including inactivated bacteria, yacoon root extract, and sugars. Other ingredients are shea butter, glycerine, panthenol, madecassoside, zinc, manganese, and spring water.
A randomised controlled trial conducted in China included 84 predominately female participants aged 34−50 years with skin damage in the form of facial pores, acne scars, photoaging, etc. Participants underwent a non-ablative fraction laser procedure, and were then treated with a collagen dressing applied once every 2 days for 14 days. Half of the participants also applied Cicaplast Baume B5+ twice a day. Significant differences in favour of Cicaplast Baume B5+ included increased hydration and sebum content as well as decreased water loss, erythema index, and melanin index. There were also fewer adverse events in the Cicaplast Baume B5+ group.35
The second study, also in China, was conducted with 45 females (25−50 years; phototypes III–IV) with atrophic acne scars or pores. They underwent ablative fractional CO2 laser resurfacing on both sides of the face. On one half of the face, Cicaplast Baume B5+ was applied, on the other, the regular hospital-issued emollient was used. Significant differences were shown in favour of Cicaplast Baume B5+ for scab shedding time, erythema index, and water loss.36
The next study, conducted in Thailand, included 12 males and eight females (16−46 years; phototypes III−V) with atrophic acne scars. Following fractional CO2 laser resurfacing, Cicaplast Baume B5+ was applied to half the face and a 0.02% triamcinolone acetonide cream to the other half, twice daily for 7 days. At all timepoints, to Day 60, there were no significant differences between the treatments in terms of redness, crusting and scaling, or post-inflammatory hyperpigmentation.There were no serious side effects.37
An open-label study in Poland and Mauritius, presented as part of this session, involved 52 participants (3−70 years old; phototypes I−V) with skin irritation conditions, including a superficial burn, dry patch, or cheilitis. Participants applied Cicaplast Baume B5+ at least twice a day for 14−24 days. There was progressive complete recovery over the time period, with 70% completely healed by Day 21. Significant improvements, favouring Cicaplast Baume B5+, were shown for erythema, cracks, and oedema. Pain and pruritus also significantly approved immediately after first application of Cicaplast Baume B5+. Clinical scores showed significant improvements from the start to the end of the trial.
In conclusion, Kerob said that “Cicaplast Baume B5+® is a suitable skin care (product) for superficial wounds and post-procedures.”








