Meeting Summary
The symposium explored new approaches to optimising the value of psoriasis management from the perspective of physicians, patients, and healthcare systems, drawing on research and evidence from real world clinical practice. The value in improving the management of psoriasis means boosting the efficacy of patient management, improving the power of outcomes measurement, raising the quality of care, and working more effectively with payers.
Many patients with moderate-to-severe psoriasis do not currently receive high quality care and are often undertreated, with many not receiving systemic therapy despite this being recommended by evidence-based guidelines. Reasons for not initiating or maintaining systemic therapy included long-term safety concerns, convenience of use, and cost, even though psoriasis can result in irreversible cumulative life impairment.
The growing recognition that psoriasis is a systemic inflammatory disorder that is associated with a wide range of comorbidities, including obesity, cardiovascular disease, diabetes, hypertension, and depression, underlines the need for systematic evaluation and treatment of comorbidities and the use of systemic treatment. Setting and implementing treatment goals is considered essential for driving up the value of psoriasis care. These should include measures that matter most to patients, taking into account the impact of psoriasis on their quality of life, including involvement of visible areas and nails, pruritus, and recalcitrant plaques, in addition to objective measures such as their Psoriasis Area Severity Index (PASI) score. Comprehensive management of psoriasis should provide treatment or referral to relevant specialists working in an integrated way across a networked service.
Improving Value in Psoriasis Management
Professor Matthias Augustin
“Improving the value of psoriasis management means optimising the efficacy of patient management, improving the power of outcomes measurement, increasing the quality of care, and working more effectively with payers,” suggested Prof Augustin as he opened the symposium. He suggested that measuring values of psoriasis management is often limited to a narrow focus on cost containment. “But healthcare organisations are starting to see improvement in value for patients as a key goal in shaping their strategies,” he said.
Important issues in improving psoriasis management include improving communication with patients, optimising long-term management of psoriasis, keeping up-to-date with new treatment strategies, best practice and guidelines, managing comorbidities, and demonstrating value in healthcare, Prof Augustin proposed. He pointed to the Global Report on Psoriasis1 published recently by the World Health Organization (WHO), which sets out the need for improved psoriasis care based on the epidemiology and burden of the disease.
The WHO report1 warned that psoriasis causes great physical, emotional, and social burdens, significantly impairing patient’s quality of life. It noted that too many people suffer needlessly from psoriasis due to an incorrect or delayed diagnosis, inadequate treatment options, and insufficient access to care. Recommendations included ensuring that patients with psoriasis have access to comprehensive, individually adapted treatment, including treatment of comorbidities, based on a model of people-centred and integrated health services.
Prof Augustin reported on the first European Dermatology Health Care Survey2 which is collecting data on dermatological care in 38 European countries. Results so far reveal wide variations in how psoriasis is currently managed in different countries, such as the percentage of dermatologists prescribing systemic drugs ranging from <5% in Cyprus to 100% in several countries, including Austria and Norway. Of the countries taking part in the survey, 18 had national psoriasis guidelines yet 14 did not.
Barriers to Guideline-Based Treatment
Professor Matthias Augustin
Considering the factors that limit provision of psoriasis care according to guidelines, Prof Augustin suggested three main barriers (Figure 1):3
- Physician factors: Lack of knowledge by physicians is a common problem in psoriasis care, particularly regarding the complexity of comorbidities, in addition to lack of awareness of guidelines, treatment goals, and limited interdisciplinary co-operation
- Patient factors: Many people have limited knowledge about psoriasis or treatment options and adherence to treatment is often poor, primarily due to concerns about treatment risks and side effects
- External factors: There are wide variations in psoriasis healthcare and implementation of guidelines and training, in addition to poor health infrastructure and the failure of health systems to fund good quality care
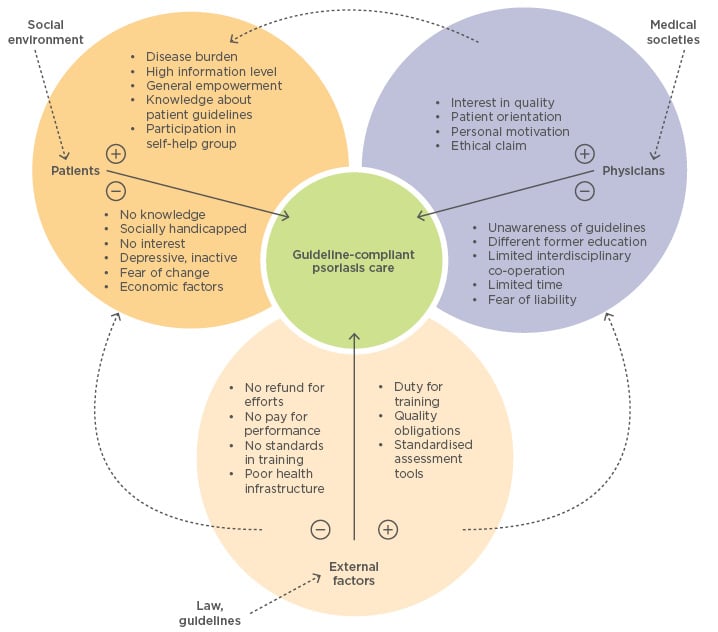
Figure 1: Barriers to guideline-compliant psoriasis care.
Adapted from Eissing et al. 2015.3
How to Improve the Value of Psoriasis Care
Professor Matthias Augustin
A key step in improving the value of psoriasis care is to set treatment goals and support physicians, patients, and healthcare systems in achieving these. The recent European consensus on treatment goals for moderate-to-severe psoriasis4 recommended using two measures to assess the impact of treatment: the change of PASI score from baseline until evaluation and the absolute Dermatology Life Quality Index (DLQI). The consensus advised that after induction and during maintenance therapy, treatment can be continued if the reduction in PASI score is ≥75%. Treatment should be modified if the improvement in PASI score is <50%. Where the therapeutic response improves by ≥50% but <75%, therapy should be modified if DLQI is >5 but continued if DLQI is ≤5. “Implementation of treatment goals in the management of psoriasis will improve patient care and reduce the problem of undertreatment,” suggested Prof Augustin. He added: “It is essential that the patient’s perspective, as measured by their quality of life, drives treatment decisions.” The annual national conference on healthcare for psoriasis in Germany agreed the following goals for 2010–2015:5
- Patients with psoriasis have a good quality of life
- Psoriatic arthritis will be detected and treated early
- Comorbidities in patients with psoriasis will be detected and treated early
- Children with psoriasis are treated early and have a good quality of life
Treatment goals should include measures that matter most to patients, he suggested, taking account the impact on their lives with involvement of visible areas and nails, pruritus, and recalcitrant plaques, in addition to objective measures such as their PASI score.
Monitoring has shown that quality indicators for psoriasis care have improved markedly over the last 10 years and regional variations have been reduced.6 “A co-ordinated nationwide psoriasis programme based on goal orientation can contribute to better quality of care and optimised outcomes,” concluded Prof Augustin.7
Measuring Value in Psoriasis Care: Physician and Patient Perspectives
Professor Marc Radtke
Physicians’ and patients’ perspectives on psoriasis and its management have changed dramatically over the past decade and it is essential that the value of care for both groups is measured as part of optimising quality, argued Prof Radtke. He noted that inpatient treatment was common in 2004, with patients typically remaining in hospital for 1–2 months each year. Treatments were generally topical, with different treatments being used on an intermittent basis, rotating different options. Comorbidities were considered coincidental rather than part of a systemic disease.
Nowadays the inflammatory nature of psoriasis is clearly understood, systemic treatment has become a standard in most countries, and healthcare provision is comprehensive. “The time to achieve control of the disease is much shorter than in the past and treatment encompasses a holistic approach in an interdisciplinary setting,” Prof Radtke suggested. Recent surveys have revealed the gap between physicians’ perceptions of factors contributing to the severity of psoriasis and what most bothers patients. Patients reported that the most bothersome skin symptom was pruritus (itching) (38%)8 while a survey of 391 dermatologists working in Europe and North America found that only 7% selected itching as the most important factor contributing to patients’ quality of life.9
From the physicians’ perspective the expectations for treatment have changed as new treatments have become available, with increased expectation for higher rates of skin clearance than in the past. The survey9 found their top attributes for ideal therapy focussed on safety, with 36.6% wanting therapy with no increased risk of serious infection or cancer and 17.4% desiring therapy with a manageable tolerability profile. The top unmet needs were improved efficacy (35.5%) and improved long-term safety (33.5%).
In terms of treatment expectations Prof Radtke suggested that efficacy, as assessed by clinical improvement measured by the percentage reduction of skin lesions on the PASI score, is of central importance to physicians. He noted that a network meta-analysis of randomised controlled trials10 showed that the efficacy of different therapies in moderate-to-severe psoriasis differed considerably. He noted that maintenance of efficacy with no loss over time was also important to physicians, together with efficacy on retreatment, if this is required. “Long-term efficacy is increasingly important. We expect long-term disease control with psoriasis treatment,” he noted, adding that there are significant differences between different therapies in sustained efficacy over time.11
What Matters to Patients?
Professor Marc Radtke
“It is also essential to assess the outcomes of psoriasis care according to patients’ needs,” Prof Radtke told the symposium. He noted that some of the currently used measures for assessing psoriasis, including the DLQI and PASI, are often insufficient to accurately monitor disease severity in individual patients. “Optimising treatment in practice includes the patient perspective,” he argued, recommending that subjective assessment at the patient level should be included in assessment of treatment effects.12
The patient benefit index assesses a patient’s needs with a questionnaire before psoriasis therapy and measures the patient’s benefits after treatment. Patients are asked questions about their expectations of treatment that include those related to PASI scores, such as recovery from skin lesions, but also questions that go far beyond this, including how important they rank being free of itching and being able to sleep better. Prof Radtke said it was also important to consider the time needed for psoriasis treatment as this has been shown to be the major predictor of quality of life in patients with psoriasis.13 Prof Radtke concluded that greater use of patient-reported outcomes is essential in improving the value of psoriasis care. He suggested that these should measure the impact of the disease and its treatment at the individual patient level, using condition-specific measures that take into account the considerable psychosocial and emotional impact of living with psoriasis.
Recognising That Psoriasis is a Systemic Disease
Professor Diamant Thaci
“Psoriasis is a complex systemic disease,” said Prof Thaci. “In the past we perceived psoriasis as only a skin disease. But it is not just about keratinocytes. We now recognise it is a T cell-driven disease involving multiple cytokines,” he told the symposium. The underlying systemic inflammation occurring in patients with psoriasis is associated with a wide range of comorbidities, including diabetes and cardiovascular disease. “Systemic biologics demonstrate the importance of tumour necrosis factor-a (TNF-a) and other cytokines in psoriasis,” he pointed out, adding that inflammation is the underlying process.
Hyperinsulinaemia (an increase in insulin levels)14 is linked with insulin resistance and the Boehncke et al. study15 has demonstrated that insulin resistance was correlated with the severity of psoriasis. A more recent study teased out the inter-relationships between obesity, psoriasis, and psoriatic arthritis, demonstrating that obesity increases the risk of both psoriasis and psoriatic arthritis as well as increasing insulin resistance, diabetes, dyslipidaemia, and metabolic syndrome, which are in turn also increased by psoriasis.16
The systemic abnormalities associated with psoriasis start in childhood. A recent study17 showed that obese children with psoriasis had higher total serum cholesterol, low-density lipoprotein cholesterol and triglycerides than children of similar BMI without psoriasis. They also had higher levels of alanine aminotransferase.
A systemic review and meta-analysis18 demonstrated that the risk of having diabetes mellitus increased with the severity of psoriasis. Patients with mild psoriasis had a 53% higher risk of diabetes than people without psoriasis (odds ratio [OR]: 1.53) and those with moderate-to-severe psoriasis had a 97% higher risk of diabetes (OR: 1.97). “The more severe the psoriasis, the more inflammation and this increased the risk of diabetes,” Prof Thaci suggested.
Potential Impact of Current Therapies on Comorbidity
Professor Diamant Thaci
It is important to consider the potential impact of psoriasis therapy on comorbidities, advised Prof Thaci. He noted that the Medical Board of the National Psoriasis Foundation (NPF) found the following after reviewing the literature:19
- Phototherapy: has no major cardiovascular impact and may reduce levels of pro-inflammatory cytokines
- Acitretin: can increase serum lipids and triglycerides but has not been shown to increase cardiovascular risk
- Cyclosporine A: can increase blood pressure, serum triglycerides, and total cholesterol
- Methotrexate: is generally associated with a decreased risk of cardiovascular disease morbidity and mortality
- Among the biologics, data for TNF-a inhibitors suggest an overall reduction in cardiovascular events; most data on short-term ustekinumab use suggest no effect on major adverse cardiovascular events, although some authorities remain concerned
“Currently there is not enough evidence to recommend therapies for psoriasis based solely on cardiovascular impact,” said Prof Thaci. “Longer-term studies are needed on this.”
Looking to the future, Prof Thaci predicted that moderate-to-severe psoriasis will be treated systemically to ensure optimal impact, recognising the systemic nature of the underlying disease process. “Psoriasis is a systemic disease, which means systemic treatment is needed,” he told the symposium, adding that treatment should be tailored much more to meet patients’ needs. He suggested that optimised management of psoriasis clinics, providing an integrated approach to the evaluation and management of patients’ comorbidities, is important to improve the value to patients and health systems.
Therapies: Optimising Value to Patients
Professor Kristian Reich
“There is no excuse for not treating psoriasis effectively with the growing range of options now available,” suggested Prof Reich. He noted that options in systemic therapy for psoriasis are available or in development for a growing range of targets including: anti-TNF-a agents (etanercept, infliximab, adalimumab, plus golimumab and certolizumab, currently used for psoriatic arthritis); anti-interleukin (IL)-12/23p40 (ustekinumab); anti-IL-17A (secukinumab plus ixekizumab in Phase III trials); anti-IL-23p19 (tildrakizumab, guselkumab, and BI 655066 in Phase III development) and PDE4 inhibitors (apremilast).
In terms of selecting psoriasis therapies Reich proposed two options:
1. Strive for the Best Efficacy
Research shows that patients’ expectations of therapy are met more completely by psoriasis therapies achieving higher PASI scores.20 Real-world data from the German Registry showed little difference in patients’ quality of life between treatments achieving PASI scores of 90 and 100, but there was a major difference between those scoring 75 compared to those scoring 90, Prof Reich reported. “So you could say, ‘the higher the PASI score the better’,” he suggested. “We have enormously effective drugs achieving high PASI scores. But how are these drugs being used by dermatologists to make patients’ lives better?” he asked.
A real world survey of patient perspectives in the management of psoriasis, the Multinational Assessment of Psoriasis and Psoriatic Arthritis (MAPP) Survey, called nearly 140,000 households in Western countries selected at random and identified 3,426 patients to ask about their treatment. Over one-third (37%) of the 168 patients with psoriasis affecting >10 palm-sized areas of their body were currently on no treatment for their condition. Just over half (52%) were on topical treatment only, while 5% were on oral plus topical therapy, and 5% were on a biologic plus topical therapy.8 “There is a massive problem with undertreatment. Although we have effective therapies many patients with psoriasis are not receiving them,” said Prof Reich. “Striving for the best is not what is happening as far as many patients are concerned.” Itching was the most important factor contributing to the severity of their psoriasis, given by 38% of patients taking part in the survey. This was followed by the location and size of skin lesions (17%), scales (11%), and flaking (10%).8 “We have tended to overlook the importance of itching to patients in the past,” said Prof Reich.
Considering potential reasons for the undertreatment of psoriasis, Prof Reich suggested that factors include: the perception of psoriasis by both physicians and patients; lack of experience in managing psoriasis among some physicians; fear of side effects with treatment by physicians and patients; reimbursement issues, particularly for more expensive therapies; and convenience of use, with some treatments requiring blood tests.
2. Broad use of Appropriate Systemic Therapies
Broad use of systemic therapies for psoriasis requires drugs that achieve a good balance between efficacy and safety. Large molecule biologic therapies, such as those acting on IL-6 or TNF-a, disrupt extracellular communication between a variety of cells including macrophages and keratinocytes.21 “However, more recently developed drugs such as apremilast act at the level of intracellular signal transduction,” Prof Reich noted, adding that this may potentially improve the balance between therapeutic benefits and side effects.
Summing up, he suggested: “Different patients require different treatment options depending on their individual risk factors, disease profile, and personal preferences.” In his personal view he recommended that phototherapy be used as induction therapy, followed by conventional systemic therapies such as methotrexate where appropriate. He proposed that the phosphodiesterase-4 inhibitor apremilast is considered as a second-line therapy in patients with moderate stable disease with limited disease burden, with or without psoriatic arthritis, and in those with particular safety concerns. The use of biologics, including adalimumab, ustekinumab, and secukinumab should be used based on individual patient profile.
“Treatment goals must include measures that matter most to patients,” Prof Reich proposed. These should take account of the impact on their lives of involvement of visible areas and nails, pruritus, and recalcitrant plaques, in addition to objective measures such as their PASI score. “To understand patient benefit, we have to ask the patient,” he concluded.
Managing Comorbidity: Providing Integrated Care
Professor Ulrich Mrowietz
Psoriasis is associated with a range of other non-dermatological conditions, generally referred to as psoriasis comorbidity (Figure 2), explained Prof Mrowietz. “This is not just a disease of the skin. It is a complex condition,” he told the meeting. “The major implication of psoriasis being associated with other conditions is the now widely accepted concept of psoriasis as a systemic inflammatory disease.”22
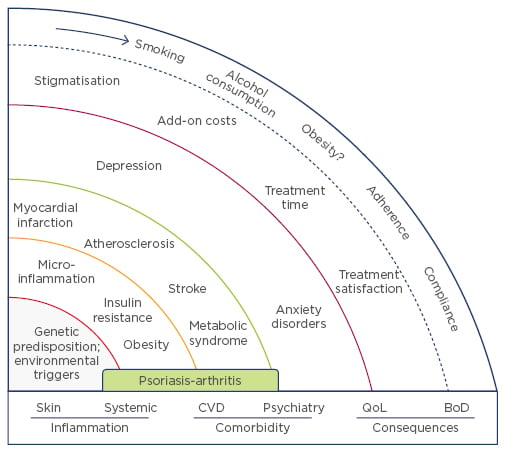
Figure 2: The spheres of psoriasis disease.
CVD: cardiovascular disease; QoL: quality of life; BoD: burden of disease.
Adapted from Mrowietz et al. 2014.23
Obesity is the most important comorbidity in psoriasis.23 Obesity is also a risk factor for the disease. Studies have shown that having a BMI >30 kg/m2 doubles the risk of developing psoriasis and is an independent risk factor for the disease across different ethnic groups.24 Prof Mrowietz noted that obesity also reduces the response to psoriasis treatment, with a large Italian study showing that obese patients did not respond as well to systemic treatment as lean patients did.25 In addition, obesity is associated with psoriatic arthritis; patients with a BMI <25kg/m2 in young adulthood have a 75% chance of not having psoriatic arthritis compared with only a 35% chance of being free from psoriatic arthritis in those with a BMI >30.26
Why is obesity so important in psoriasis? Prof Mrowietz explained that fat cells increase in number and size with weight gain, which leads to increased production of the chemokine MCP-1. This recruits inflammatory macrophages which produce TNF-a and IL-6. The change in metabolic profile with obesity produces hormones such as leptin, resulting in insulin resistance. “The result is constant micro-inflammation throughout the whole body,” Prof Mrowietz told delegates. He reported intriguing research suggesting that increased levels of inflammatory cytokines such as TNF-a and IL-6 in the central nervous system can cause depression,27 noting that increased rates of anxiety and depression occur in both children and adults with psoriasis.
Metabolic syndrome is another important condition commonly associated with psoriasis and with the subsequent or parallel development of atherosclerosis implicated in cardiovascular complications including myocardial infarction (MI) and stroke.28 Patients with psoriasis have an increased risk of MI compared to people without psoriasis, and those who have had a MI have an increased risk of a second event.
Effective management of psoriasis includes screening patients for comorbidities and providing treatment or referral to relevant specialists working in an integrated way across a networked service. Prof Mrowietz suggested: “It is essential to identify associated conditions such as obesity, and lifestyle factors including smoking and alcohol consumption, which can exacerbate psoriasis. Patients should be encouraged to join weight loss and smoking cessation programmes.” A retrospective analysis of psoriasis patients undergoing bariatric surgery for obesity showed that 70% achieved remission within 6 months.29 “Weight loss is a very meaningful intervention,” he said.
Systemic anti-inflammatory drugs have been shown to reduce the risk of cardiovascular events in patients with psoriasis. One study has shown that the risk of MI was reduced with TNF-a inhibitors compared to treatment with topical agents.30 Five-year follow-up of a Danish nationwide cohort showed that methotrexate was associated with significantly lower rates of cardiovascular events (hazard ratio [HR]: 0.53) and there was a comparable protective effect with biological drugs (HR: 0.58) compared to other anti-psoriatic therapies including cyclosporine (HR: 1.06) and retinoids (HR: 1.80).31
Prof Mroweitz noted that effective treatment of cardiovascular risk factors including diabetes, dyslipidaemia, and hypertension are also important. “This underlines the need for a multidisciplinary approach to managing patients with psoriasis,” he pointed out. He recommended that all patients with moderate-to-severe psoriasis be screened regularly for risk factors including blood pressure, lipids, BMI, glucose metabolism, and depression.32 “Risk screening must be holistic, using a comprehensive screening tool,” he said.
Summing up, Prof Mrowietz said that comorbidity is an important facet of psoriasis, with a wide range of commonly occurring comorbidities, including cardiovascular disease, depression, and anxiety. “Screening for comorbidities and effective management is essential for improving the care of patients with psoriasis,” he concluded. An integrated approach (Figure 3), involving co-operation between different disciplines and centres of excellence working effectively with networks of psoriasis centres, is essential to improving the management of psoriasis.
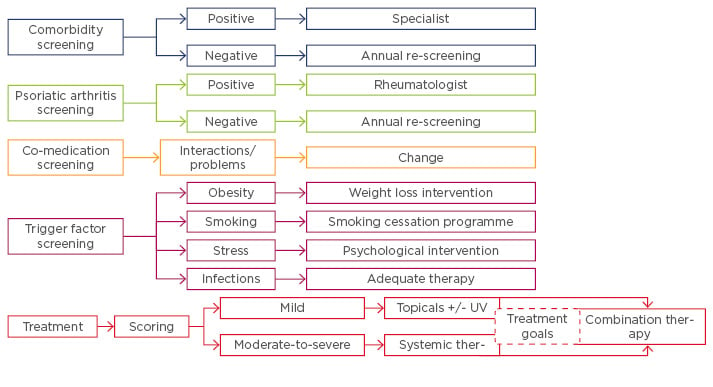
Figure 3: Integrated concept of psoriasis management.
UV: ultraviolet.
Adapted from Mrowietz et al. 2014.23








