Abstract
Objective: Obesity and thyroid nodules are very common. Many studies have reported that weight gain is a new risk factor for goiters and thyroid nodules. In this study, the authors aimed to evaluate thyroid morphology in obese women and tried to link thyroid morphology with anthropometric parameters.
Design: A cross-sectional study was conducted between January 2017 and January 2018. Overweight and obese Syrian females, aged >35 years, without past or recent thyroid complaints, and living in the southern region of Syria were included in the study. Weight, height, and hip and waist circumference were measured to calculate anthropometric parameters. Thyroid volume and prevalence of thyroid nodules were compared among BMI groups as defined by World Health Organization classifications. Body Surface Area (BSA), waist circumference (WC), waist:hip ratio, and waist-to-height ratio (WtHR) were compared between nodule groups.
Results: A total of 140 overweight and obese females, with a mean age of aged 53±7 years were studied. Mean thyroid volume was 12.4±7.4 mL, with the highest volume identified in the Obesity Class III group (14.9±10.9 mL). Significant positive correlations were found between thyroid volume and weight, BMI, WC, BSA, and WtHR (p<0.05). Thyroid nodule prevalence was 84.3%, and 22.0% of the nodules were fine needle aspiration indicated. Weight and BSA were positively correlated with thyroid nodules (p<0.05), while BMI was not.
Conclusion: Thyroid volume was positively correlated with increased weight, BMI, WC, BSA, and WtHR in Syrian females. Positive correlations were found between weight, BSA, and thyroid nodules after age and thyroid stimulating hormone were excluded.
INTRODUCTION
A goiter is a nodular or diffuse enlargement of the thyroid gland. It is defined as an adaptive response of the thyroid follicular cells to any factor that impairs thyroid hormone synthesis.1,2 Genetics, iodine deficiency, and thyroid stimulating hormone (TSH) are the most important factors that contribute to elevated thyroid volume and nodules formation,2-4 with women and the elderly more likely to have thyroid nodules.5-7 Some studies have suggested other risk factors, such as smoking, obesity, and insulin resistance, contribute to nodule formation.3,8-10 Recently, the association between metabolic obesity and thyroid morphology has received attention. While some studies demonstrated a significant positive correlation between obesity and altered thyroid morphology,6,11-13 others did not.14-16 BMI, body surface area (BSA), waist circumference (WC), waist:hip ratio (WHR), and waist-to-height ratio (WtHR) are clinical parameters of body fat and visceral obesity.17,18
In this study, the authors evaluate thyroid gland morphology, via ultrasonography, in a sample of overweight and obese females, and examine its relation to BMI and other anthropometric parameters in a mild iodine deficiency area.19
METHODS
Population Features
A cross-sectional study was conducted between January 2017 and January 2018. Participants were females aged >35 years and had lived in southern Syria (an area of mild iodine deficiency)20 for the last 10 years. The participants were selected from outpatient clinics, patients in the hospital wards, and their relatives at Al-Mouwasat University Hospital, Damascus, Syria. Subjects with previous or recent thyroid complaints were excluded. All individuals who gave informed consent were interviewed using a questionnaire that collected information regarding patient age, demographic characteristics, medical history, family history of thyroid disease, smoking, and dietary iodised salt.
Anthropometric Parameters
Weight (with light clothes) and barefoot height were measured using the Seca Scale Model 713 device (Boian Surgical, Padstow, Australia). Waist circumference (the midpoint between the top of the iliac crest and the lower margin of the last palpable rib) and hip circumference (around the widest portion of the buttocks)17 were measured by the same physician with a flexible tape. BMI, BSA, WHR, and WtHR were calculated as follows:
- BMI = weight (kg)/height (m)2.
- BSA (Dubois formula) = 0.007184×(height [m]0.725)×(weight [kg]0.425).21
- WHR = waist circumference (cm)/hip circumference (cm).17
- WtHR = waist circumference (cm)/height (cm).18
Depending on BMI, participants were classified into four groups (overweight [BMI: 25.0–29.9 kg/m2], Obesity Class I [BMI: 30.0–34.9 kg/m2], Obesity Class II [BMI: 35.0–39.9 kg/m2], and Obesity Class III [BMI: ≥40 kg/m2]) according to the World Health Organization classification.22 All subjects with BMI <25 kg/m2 were excluded.
Thyroid Morphology
Thyroid ultrasound, using a L14-6N (6-14 Mhz) Mindray (Providian Medical Equipment, Highland Heights, Ohio, USA) ultrasound device, was performed with the patients in the supine position. Total thyroid volume was determined by the addition of the volumes of both lobes. The lobe volume was calculated using the formula: (length x width x thickness x π/6).23 Isthmus volume was ignored unless it had nodules, whereupon their volumes were calculated and added to the total volume. A goiter was defined as thyroid volume ≥18 mL in females.24,25 Each detected nodule (≥2 mm) in the gland was evaluated for fine needle aspiration (FNA) indications according to the American Thyroid Association (ATA) criteria.26
Serum TSH was measured in the central laboratory of the hospital, using either IcromaTM (Boditech Med Inc., Chungcheon-si, South Korea), IMMULITE® (Siemens Healthineers, Erlangen, Germany), or Liaison® XL (DiaSorin, Saluggia, Italy) kits, with a 0.3–5.0 μIU/mL reference range.
STATISTICAL ANALYSES
Data analysis was performed using the SPSS software version 23.0 (IBM, Armonk, New York, USA), where p values <0.05 were considered statistically significant. Competitive data were compared among different groups based on variable distribution using the Kolmogorov–Smirnov test. T test and analysis of variance (ANOVA) were used for normal distribution, while Mann–Whitney and Kruskal–Wallis analyses were used as nonparametric tests. The chi square test was used for nominal data and Spearman’s rho for correlation.
RESULTS
General Characteristics
A total of 140 overweight or obese females were studied. Their mean age was 53±7 years (Table 1). Mean BMI was 34.2±5.1 kg/m2 (24.2% overweight, 32.9% Obesity Class I, 27.9% Obesity Class II, and 15.0% Obesity Class III). In total, 87.9% of subjects had a WC of >88 cm and 85.9% had a WHR of ≥0.85.
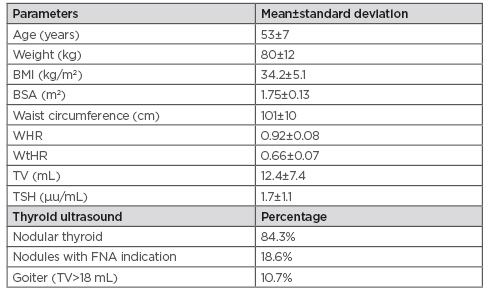
Table 1: Characteristics of the 140 female patients included in the study.
BSA: body surface area; TSH: thyroid stimulating hormone; TV: thyroid volume; WHR: waist:hip ratio; WtHR: waist-to-height ratio.
Thyroid Volume and Anthropometric Parameters
Goiter (thyroid volume >18 mL) prevalence was 10.7%. Significant differences were found in thyroid volume among BMI groups (p<0.05). The Obesity Class III group had the highest thyroid volume (14.9±10.9 mL) (Table 2). Spearman’s rho showed a statistically significant positive correlation between thyroid volume and BMI before and after excluding age and TSH as confounding factors in partial correlation (r=0.24; p=0.004 and r=0.23; p=0.008, respectively). Thyroid volume was also positively correlated with weight, WC, BSA, and WtHR (p<0.05) (Table 3).
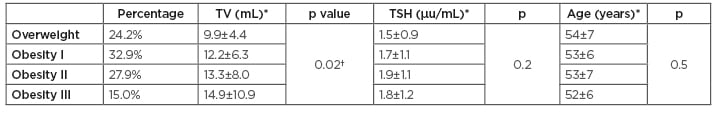
Table 2: Comparison of thyroid volume and thyroid stimulating hormone among BMI groups.
*Mean±standard deviation
†Statistically significant
TSH: thyroid stimulating hormone; TV: thyroid volume.
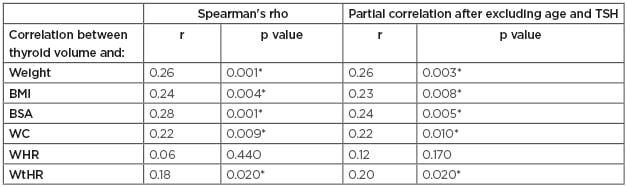
Table 3: Correlations between thyroid volume and anthropometric parameters.
*Statistically significant
BSA: body surface area; TSH: thyroid stimulating hormone; TV: thyroid volume; WC: waist circumference; WHR: waist:hip ratio; WtHR: waist-to-height ratio.
Thyroid Nodules and Anthropometric Parameters
Thyroid nodules were detected in 84.6% of the participants, 22% of which had FNA indication. The patients were divided into three groups: no nodules (15.7%), nodules without FNA indication (65.7%), and nodules with FNA indication (18.6%) (). Weight, WC, and BSA were higher in nodule groups with significant difference (p<0.05). There were significant positive correlations between weight, BSA, and thyroid nodules after age and TSH were excluded (r=0.17, p=0.049).
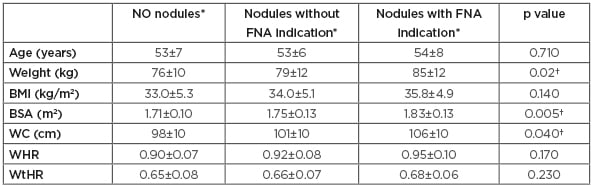
Table 4: Comparison of anthropometric parameters among nodule groups.
*Mean±standard deviation
†Statistically significant
BSA: body surface area; FNA: fine needle aspiration; TSH: thyroid stimulating hormone; TV: thyroid volume; WC: waist circumference; WHR: waist:hip ratio; WtHR: waist-to-height ratio.
DISCUSSION
Obesity is a complex, multifactorial, chronic disease. Its prevalence has been increasing dramatically worldwide, almost tripling between 1975 and 2016.27 Obesity is considered a major risk factor for diabetes, hypertension, and cardiovascular diseases. Recently, many studies have tried to find other comorbidities associated with obesity. Thyroid disorders are the most common endocrine diseases. High prevalence of goiter and incidental thyroid nodules has led researchers to investigate their relationship with obesity.
In this study, the authors evaluated thyroid gland morphology in a sample of middle-aged, obese females with no thyroid complaints. The Obesity Class III group had the highest goiter prevalence and the largest thyroid volume. Positive correlations were found between thyroid volume and other anthropometric parameters (weight, BSA, WtHR, and WC), but not WHR.
These results were aligned with those of many other studies.28-32 Dauksiene et al.29 showed that BMI was an independent predictor of goiter development. Eray et al.32 showed that thyroid volume was positively correlated with BMI, with an important reduction in weight loss after 6 months. Turcios et al.28 found that BMI and BSA were positively linked to thyroid volume but WHR was not. WtHR, as a metabolic parameter, has not been studied with thyroid volume before. The positive results presented here indicate its importance as an independent predictor for thyroid volume.
Regarding thyroid nodules, a high prevalence of thyroid nodules (84.6%) was documented in the mildly iodine deficient southern region of Syria; this was higher than the mean prevalence in other studies, including 44.9%, 34.2%, and 35.4% in Yemen,33 South Korea6 and China,11 respectively. It is hypothesised that the high prevalence maybe due to the age group and female sex. Values of weight, BSA, and WC were the highest in the FNA-indicated nodules group in a statically significant pattern, while BMI and WHR were insignificant. Panagiotou et al.16 reported that WC was the only anthropometric parametric value that was significantly higher in the nodules groups compared to the no nodules group. Song et al.13 found a positive significant correlation between thyroid nodules and BMI and BSA, which became insignificant with BMI after being modulated by age and TSH as confounding factors. In a Korean survey, on apparently healthy people, Moon et al.6 documented that WC and BMI were highest in the thyroid nodules group.
The mechanism of the association between obesity and thyroid morphology is not entirely understood. Obesity is characterised by many complicated mechanisms, of which insulin resistance is the most important.34 Hyperinsulinaemia, in the case of insulin resistance, can up-regulate hepatic growth hormone receptors and suppress insulin-like growth factors binding proteins, leading to increased bioactive insulin-like growth factor 1 (IGF1) levels.35 Insulin and IGF1 act via their highly homologous receptors,36,37 which are expressed in thyrocytes by TSH.38 They synergise with TSH, enhancing proliferation and differentiation of thyroid cells,38 and having anti-apoptotic effects (Igf1).39 This hypothesis is supported by the findings of Anil et al.,40 who demonstrated that metformin (an insulin sensitiser agent) therapy significantly decreased thyroid volume in subjects with insulin resistance. Insulin resistance may explain the goitrogenic effects of obesity, but more studies are still needed.
LIMITATIONS
In this study, the authors faced many limitations. The focus was placed on thyroid dimensions to calculate the volume, but this did not evaluate thyroid echogenicity. The study did not include grey-scale analysis41 and could not detect the possible accumulation of lipids in the thyroid in cases of high BMI. In addition, there was a lack of description of autoimmune thyroiditis echographic features which may be present in patients with obesis.41
CONCLUSION
This study indicates an association between obesity and thyroid morphology in middle-aged Syrian females. High prevalence of goiter and thyroid nodules in obese females should highlight the importance of evaluating the thyroid gland using thyroid ultrasound as a screening test. Early detection of thyroid nodules will result in a better prognosis.