Abstract
Background: Heart failure (HF) is underdiagnosed among patients with diabetes. Awareness is required to improve its management and to reduce its impact.
Objectives: To suggest a risk assessment tool that could facilitate the early diagnosis of HF and even reduce its incidence by facilitating individualised management plans.
Methods: Assess current medical literature, searching for parameters that indicate a higher risk of HF among the diabetic population.
Results: Twenty-four parameters were found that could be the potential basis for a risk stratification tool.
Conclusion: The concept of a risk stratification tool is presented. Work on validating will be required. It has the potential to affect the future management of patients with diabetes and to reduce the incidence and prevalence of HF in this population.
BACKGROUND
Patients with diabetes have a two-fold increase in the risk of heart failure (HF).1 Unfortunately, many patients with diabetes remain undiagnosed for HF, even when screening tools like the brain natriuretic peptide (BNP) blood test have been around for some time.2,3 This test remains underutilised.4 Furthermore, it is not very specific; in consequence, one has to consider how to increase its predictive value and how the screening for HF could be improved. Looking at the literature for other parameters, combining their predictive value, and creating a potential tool to assess more specifically those at risk of developing HF could be possible before the need for a referral for tests like an echocardiogram to confirm the presence of HF; therefore, it could improve screening. Thinking proactively, it could also be used to determine a pre-HF status in the same way that patients are monitored when presenting with pre-diabetes to change its natural progression to established disease. Assessing a patient in more detail, assessing the risk of developing HF, and managing those factors could be a way to reduce the prevalence of this condition. The authors, therefore, aim to provide a proof-of-concept for a risk stratification tool to be used in primary care.
METHODS
A non-systematic review of the literature was conducted, looking for “heart failure risk” AND “diabetes” in PubMed with a filter to limit results to the last 5 years. A total of 48 manuscripts were found and, among them, 16 articles were considered for further analysis. Additional papers from references were searched to sustain statements of risk factors already known and were also included in this review.
RESULTS
It can be summarised that a family physician, while reviewing a patient with diabetes, could calculate the risk this patient has of developing HF by taking into consideration particular parameters.
General factors:
- Age of the patient, as the risk of developing HF increases with increasing age5-7
- Sex, as the risk is significantly greater among women8
- The length of time the patient has had diabetes, which is associated with a higher risk of developing HF5,6
Lifestyle factors:
- Smoking is associated with an increased risk of HF7
Symptoms suggestive of heart failure:2
- Ankle swelling
- Dyspnoea
- Fatigue
Physical examination findings:
- Obesity, or elevated BMI and waist to hip ratio (WHR) increase the risk of HF6,9
Blood tests:
- Higher levels of HbA1c are associated with the development of HF6,10
- Raised BNP levels3,6,9
- Note that raised uric acid levels have been associated with the development of HF11
Medication:
- Some oral antidiabetics have a negative effect on HF, such as insulin, sulfonylureas (i.e., glibenclamide), dipeptidyl peptidase-4 inhibitors (i.e., sitagliptin), and thiazolidinediones (i.e., pioglitazone)
- Other oral antidiabetics have a positive effect on HF, such as sodium-glucose co-transporter-2 inhibitors (i.e., dapagliflozin) and metformin
- Some pharmaceutical agents seem to be more neutral, such as glucagon-like peptide-1 (i.e., liraglutide)3,12
Comorbidities:
- Cardiovascular disease increases the risk of HF, whether atrial fibrillation, hypertension, ischaemic heart disease, or peripheral vascular disease2,5-7,9
- Increased risk of HF linked to chronic kidney disease, dyslipidaemia, and obstructive sleep apnoea5,6,13
DISCUSSION
Several parameters are linked to developing HF or additional risk to be admitted with HF (Table 1).2,3,5-12,14-16 Up to now, the focus on screening has been on the use of BNP and not on these other factors. It has to be argued that it should be possible to validate their combined risk as part of a new scoring tool that could provide a more valuable and accurate risk assessment of patients with HF.
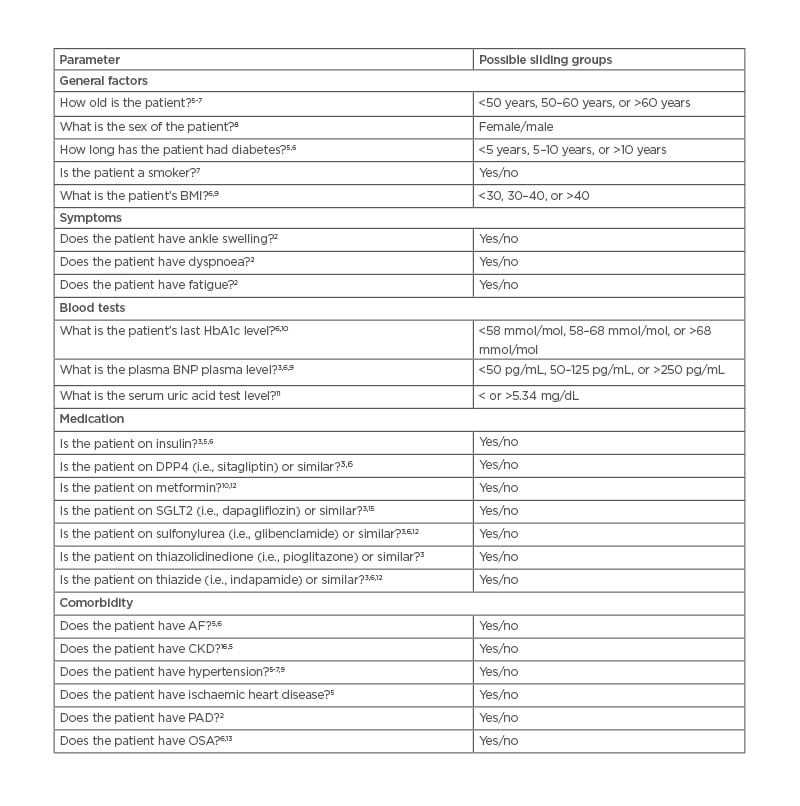
Table 1: Suggested parameters for a risk assessment tool.
AF: atrial fibrillation; BNP: brain natriuretic peptide; CKD: chronic kidney disease; DPP4: dipeptidyl peptidase-4; OSA: obstructive sleep apnoea; PAD: peripheral artery disease; SFLT2: sodium-glucose co-transporter-2.
In primary care, family physicians are probably screening patients with diabetes with BNP to diagnose early HF, but it is not enough. BNP alone is sensitive enough, but its use is probably not as widespread as required to make an impact on reducing the burden of undiagnosed HF. BNP is utilised too little to consider it as part of a wider assessment of the management of the patient that could, theoretically, reduce the incidence of HF by acting on those parameters that could be amenable to change (such as lifestyle changes, medication, or management of comorbidities).
A more structured way to assess patients is needed to change the goal from early HF detection to reducing incidence by managing the risks better, allowing an informed patient to understand how to better their chances, and to engage them in the relevance of those changes that can significantly alter their future health.
HF risk stratification tools to understand the risk of patient hospitalisation and death after discharge from hospital have been developed.5,17 Berg’s tool was based on five independent parameters (prior HF, history of atrial fibrillation, coronary artery disease, estimated glomerular filtration rate, and urine albumin-to-creatinine ratio), while the tool developed by Meta-Analysis Global Group in Chronic Heart Failure (MAGGIC) was based on 13 patient factors (age, gender, diabetes, chronic obstructive pulmonary disorder, HF diagnosed within the last 18 months, current smoker, New York Health Association [NYHA] classification, use of β-blockers, angiotensin-converting enzyme inhibitors or angiotensin II receptor blockers, BMI, systolic blood pressure, creatinine, and left ventricular ejection fraction).5,17 These tools were intended for different use in secondary care. Although Berg’s tool could be the basis of a wider tool to assess the risk of developing HF, not just the risk for hospital admission, MAGGIC’s tool is for use in patients already diagnosed with HF.5,17 General practitioners are in need of a new instrument to be able to proactively manage patients with diabetes differently, similar to the concept of QRISK3, thus reducing the prevalence of HF.18 Preventative treatment could be initiated before the potential diagnosis of HF is made; patients, understanding their risk of developing HF, could put more interest on lifestyle changes as improving diabetes alone does not seem to be enough to trigger these changes. Furthermore, the management of diabetes could be transformed by selecting different hypoglycaemic agents and with additional cardiovascular support in the way of renin–angiotensin–aldosterone inhibition therapy, for example.9
Finally, starting to use a risk calculator will have additional effects on improving and updating this type of tool, looking for more ways to understand the risk of developing HF as well as preventing it, promoting research in this subject.
CONCLUSION
Family physicians could reduce the burden of HF among patients with diabetes by using a risk calculator like the one suggested here. It is time to be more proactive and, thus, a tool to assess the risk of HF and, potentially, to reduce its prevalence in the population should be validated.