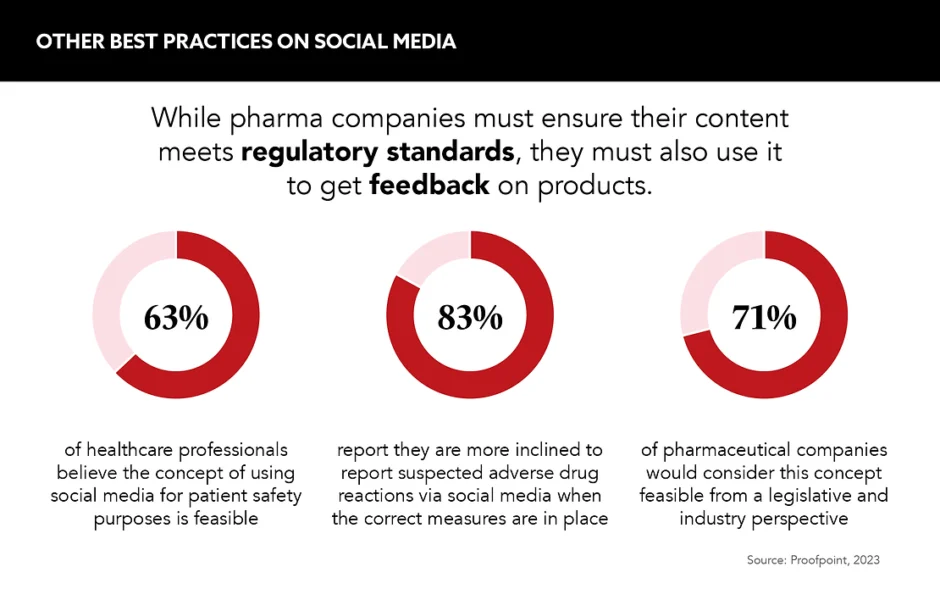
As the UK PMCPA publishes its first social media guidance for pharmaceutical companies, what can they do to avoid traps when it comes to posting online?
Words by Saša Janković
It feels like we live in an age where everyone and their dog is sharing the minutiae of their lives on social media. Almost everyone has heard of someone who posted what they thought was an innocuous photo, reel or comment on their online channels only to be deluged with responses pointing out where they’ve failed to ‘read the room’, or worse.
For individuals the fallout can range from minor embarrassment to, in some cases, legal action – but in the world of the pharmaceutical industry these consequences can be reputationally, financially and regulatorily even more severe.
Indeed, this year the Prescription Medicines Code of Practice Authority (PMCPA) – the self-regulatory body which administers the Association of the British Pharmaceutical Industry (ABPI) Code of Practice for the Pharmaceutical Industry – has ruled on 28 alleged social media breaches made by pharma companies and their employees in 2022.
The cases make for interesting reading and while some appear to be obvious breaches, others are less so. For example, a podcast video on a patient organisation’s YouTube channel only revealed the sponsoring pharma company’s involvement at the end of the 33-minute video. This meant that viewers were not initially aware of the sponsorship.
Another example saw a pharma employee reported to the PMCPA for posting a link to a national news story using the word ‘safe’ in relation to the company’s product. Any material associated with a social media post, including a link within a post, is regarded as part of that post.
There was also a violation involving a personal LinkedIn profile of a pharma employee who had a brand name and indication mentioned in their job title. This was considered as promoting a prescription only medicine (POM) to the public.
Legal pitfalls
In the UK, pharma companies are prohibited from advertising POMs to the public and from advertising a medicine before it has been granted a marketing authorisation allowing its sale or supply.
In the USA, while federal law does not bar drug companies from advertising any kind of prescription drugs, the FDA outlines that “companies cannot use reminder ads for drugs with certain serious risks (drugs with ‘boxed warnings’)” and requires “all risks be communicated”.
For UK-based pharma marketers using social media, Dr Amit Aggarwal, Executive Director of Medical Affairs and Strategic Partnerships, ABPI, says it is important to remember the basic principle of the ban on advertising medicines to the public before posting online.
He explains that the laws against advertising an unlicensed medicine or unlicensed indication “applies not only to pharmaceutical companies themselves, but other organisations, healthcare providers, as well as individuals”, so “even mentioning the name of a medicine on social media may constitute promotion”.
Even mentioning the name of a medicine on social media may constitute promotion
In addition, Andrew Austin, Partner, Freshfields Bruckhaus Deringer, points out: “Social media advertising by the pharmaceutical industry must also comply with generally applicable and sector-specific rules covering the advertising of medicinal products.”
Other traps to avoid
Another less obvious breach for pharma companies in the UK is the use of tagging and hashtags.
For example, Dr Aggarwal explains that if a pharma company tags a healthcare professional on social media, thereby linking them to an account containing promotional material for the company’s drug, this would “likely breach the ABPI Code if there was evidence that the promotional content appeared on the linked account at the time the link was made”.
Similarly, he warns: “Choosing a hashtag that contained a claim for a prescription only medicine would likely constitute promotion”, and “an indication or therapy area – e.g. obesity – might constitute promotion of a product if it was used in combination with other language which could identify a specific product.” In addition, the PMCPA dictates that even corporate news and announcements should not directly or indirectly mention products.
If in doubt, speak to the PMCP
Check the guidance
Handily, the new PMCPA Social Media Guidance 2023 helps companies use social media in a compliant way, including by sharing key questions to consider before carrying out any social media activity such as:
- What is the objective of the activity?
- What content will be made available?
- Who is the audience (for example, public, health professionals, media or investors) and is the content suitable and appropriately signposted for them?
- Is the content in line with company guidance that is clear and consistent with all applicable codes, laws and regulations?
Dr Aggarwal advises training for the whole organisation on the ABPI Code and reviewing recently published cases. If pharma companies really want to stay out of the social media doghouse, he adds: “If in doubt, speak to the PMCPA [who] are always happy to provide advice to companies to help them maintain high standards, support high-quality healthcare and stay within the Code.”