Peripancreatic fluid collections (PFC) frequently occur in the context of acute and chronic pancreatitis, and trauma. The majority of cases have favourable outcomes, but some patients develop a deterioration in their general condition within the 2nd and 3rd weeks, reaching a high mortality rate of 40–70%. Collection should be properly classified according to the type of PFC, and the best exam to obtain this classification is computed tomography (CT). The current available method is to use the Revised Atlanta Classification (2012); this tries to overcome the existing confusion regarding the reporting of morphological features and complications of acute pancreatitis.
The revised definitions differ between acute PFC associated with acute interstitial oedematous pancreatitis and acute necrotic collections that occur in necrotising pancreatitis within the acute phase. On the other hand, in the late phase after >4 weeks, the classifications describe pancreatic pseudocysts developing from interstitial oedematous pancreatitis or walled-off necrosis, resulting from necrotising pancreatitis (Table 1).1
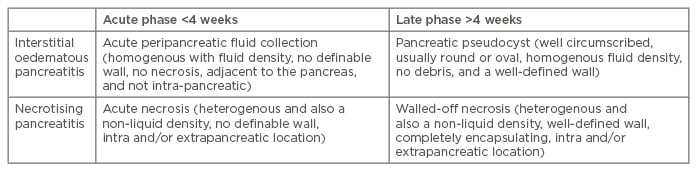
Table 1: Definition of peripancreatic fluid collections based on contrast-enhanced computed tomography criteria.*
*Modified according to the updated Atlanta classification.
In 50% of cases, pseudocysts will resolve spontaneously and, therefore, a conservative expectant approach is often the right clinical decision. Only superinfected or clinically symptomatic pancreatic pseudocysts should be considered for interventional treatment. Infection, pain, malnutrition, or compression of biliary, intestinal, or vascular structures might present an indication for endoscopic, percutaneous, or surgical drainage. The size of the collection alone should not influence the decision for interventional treatment.
Endoscopy is a minimally invasive technique for the drainage of PFC, showing great clinical outcomes, reduced cost compared to alternative methods, and resulting in a shorter length of hospital stay for the patient when compared to surgery results.2 The major problem with endoscopic drainage is the ineffective treatment of the cause. In cases of pancreatitic pseudocysts, this problem frequently leads to infection. In patients with walled-off necrosis, the common problem is the extension of the deep debris into the abdominal flanks or multiloculated cavities. In cases like this, one can use percutaneous or surgical drainages in addition to other measures. Several modifications have been made to endoscopic techniques to improve the outcomes of endoscopic PFC drainage, such as: dual modality treatment that combines endoscopic and percutaneous approaches, the creation of multiple transluminal fistulae (multiple transluminal gateway technique) to improve the drainage of necrotic contents, direct endoscopic necrosectomy, and, recently, the placement of transmural self-expandable metal stents.
In the case of extensive necrosis, it can be necessary to extract the necrotic tissue from the walled-off cyst in order to induce the healing process. This requires the creation of a large calibre transmural hole between the gastric (or duodenal) and cystic lumen to allow the passage of a standard endoscope into the cystic cavity.3 In this scenario, a lot of stents are available (such as metallic, plastic, or more recently, the biflanged metal stent), but up to now they do not seem to have shown any statistical difference in relation to technical success, adverse events, or recurrence if a plastic stent is used to maintain the hole opening.4
Our experience with endoscopic drainage in 56 patients was successful in 87% of cases. Over a period of 21 months, there were 10.5% pancreatitic pseudocyst recurrences, and 5.2% recurrence of newepisodes of pancreatic necrosis; all were managed clinically and/or endoscopically. No mortality was related to the procedure. For our group, we found that endoscopic drainage with daily necrosectomyproved a useful method to remove infected and sterile pancreatic necrosis.5








