INTRODUCTION AND AIMS
Current first-line oral nucleos(t)ide analogues, entecavir (ETV) and tenofovir, have had a major impact on chronic hepatitis B virus (HBV) (CHB) treatment. Nevertheless, the risk of developing hepatocellular carcinoma (HCC) cannot be completely eliminated regardless of long-term virological remission,1 thus, risk stratification and early detection of HCC are of great importance for patients with CHB. Several scores for predicting the risk of HCC have been developed.2 Recently, a risk score was developed for Caucasians receiving ETV or tenofovir called the PAGE-B score. This score estimates HCC risk within the first 5 years of treatment.3 Our study aimed to identify predictors of HCC in Tunisian CHB patients treated with ETV, to assess predictive performance of the PAGE-B risk score, and to compare its accuracy with other HCC risk prediction models, such as the REACH-B score, before and during treatment.
PATIENTS AND METHODS
This is a retrospective study covering 5 years (2011–2016) and including all patients with CHB with or without cirrhosis undergoing ETV for at least 12 months. The date of enrolment was the first day of treatment. Excluded patients were those with hepatitis C virus (HCV), HIV, or hepatitis D virus (HDV) coinfection; co-existing primary liver disease; HCC at enrolment; and HCC development within 12 months after enrolment. Clinical and biological examinations and abdominal ultrasound were performed every 6 months. Diagnosis of HCC was based on findings from sectional imagery or on histological evidence.4 PAGE-B and REACH-B scores were calculated at enrolment and 1 year after treatment. The PAGE-B score is based on age, sex, and platelet count at the start of therapy.3 The REACH-B model includes age, sex, alanine aminotransferase level, hepatitis B e antigen (HbeAg) status, and HBV DNA level as variables.5 Predefined thresholds (10 and 17 for PAGE-B and 8 for REACH-B) allowed us to classify patients according to the probability of developing HCC.
RESULTS
Overall, 67 patients were enrolled with a mean age of 47 years (24–73 years). Twenty-eight patients (43%) had been previously treated with pegylated IFN in 25 cases and lamivudine in 3 cases. Thirty-six patients (55%) had cirrhosis. Six patients (9%) were HbeAg-positive. HCC occurred in 14 patients (20%) within 5 years, with a mean duration of 45 months (12–58 years). Thirteen (92%) of these patients were cirrhotic. At 1 year of ETV, virological remission was obtained in 51 patients (75%), increasing to 90% at 2 years without reducing the risk of HCC (p=0.53). Univariate and subsequent multivariate Cox proportional hazard regression analyses showed that older age, male sex, and HBeAg positivity were independent predictors of HCC development (Table 1). The area under the receiver operating characteristic curve of PAGE-B was significantly higher than that of the REACH-B (0.91 versus 0.80 at 5 years, p=0.03). When calculated at 1 year after ETV, the PAGE-B showed higher predictability than that of the REACH-B, with an area under the receiver operating characteristic curve value of 0.88 versus 0.67 and 95% confidence interval of 0.769–0.992 and 0.566–0.972, respectively, p=0.001. Five-year HCC-free survival in patients with a PAGE-B score ≤10 was 94% versus 62% with a PAGE-B score >10 and 30% with a PAGE-B score ≥17 (p<0.010). Furthermore, the 5-year HCC-free survival in patients without cirrhosis with a PAGE-B score <10 was 100%.
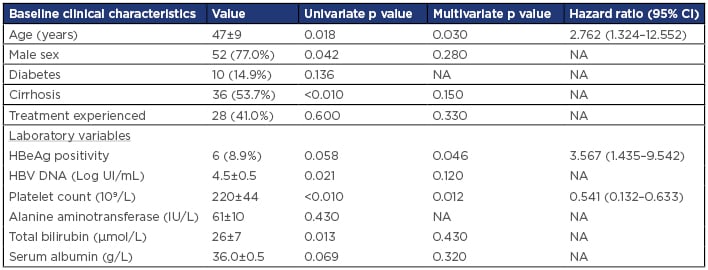
Table 1: Independent predictors of hepatocellular carcinoma development.
CI: confidence interval; HBeAg: hepatitis B e antigen; HBV: hepatitis B virus; NA: not applicable.
DISCUSSION AND CONCLUSION
Our study confirmed that the risk of HCC persists in CHB patients treated with ETV regardless of virological remission. Older age, lower platelet count, and positive HbeAg were independent predictors of HCC occurrence. These factors have been widely described in the literature as being predictive of HCC development: age reflects accumulation of liver damage;6 platelet count represents a marker of liver disease severity and may be a more reliable factor than cirrhosis itself;7 and HbeAg positivity is associated with a recurrence of cycles of necrosis and regeneration, which increases the probability of malignant transformation. Moreover, our study showed that PAGE-B had high accuracy for the prediction of HCC development in this sample of CHB Tunisian patients treated with ETV. This score is, therefore, reliable, easy to calculate, reproducible, and dynamic. In addition, we identified a particular group of patients (CHB without cirrhosis with a PAGE-B score <10) who had an almost zero risk of developing HCC within 5 years of treatment and, therefore, do not require close supervision.








