Abstract
Currently, the coronavirus disease (COVID-19) pandemic, which is caused by severe acute respiratory syndrome coronavirus 2 (SARS CoV-2), is a major global public health emergency. Cytokine storm is a key factor and plays a major role in disease severity and clinical outcome. Recently, the literature reveals the use of therapeutic plasma exchange to reduce the inflammatory markers. Evidence also exists for the use of convalescent plasma therapy in patients with severe COVID-19. This brief communication explores the advantages on therapeutic plasma exchange with convalescent plasma in patients with moderate-to-severe COVID-19.
INTRODUCTION
Severe acute respiratory syndrome coronavirus 2 (SARS CoV-2) is a positive-sense, single-stranded RNA virus that belongs to the coronavirus family, and it is the seventh in that family known to infect humans. It is responsible for the third pandemic disease in the past two decades. The disease caused by SARS CoV-2 is referred to as coronavirus disease (COVID-19).1 COVID-19 manifests as a mild disease in approximately 80% of infected patients; the remaining would require hospitalisation, sometimes in intensive care with or without respiratory support. Death is often a result of multi-organ failure and is common amongst ‘high-risk’ populations, such as patients aged >60 years; presence of comorbidities including diabetes, hypertension, and associated chronic diseases; and those on immunosuppressants. Globally, scientists and researchers are in the process of discovering drugs, vaccines, and many other modalities to combat this virus.
CYTOKINE STORM
Huang et al.,2 in a study on patients with severe presentations of COVID-19, found that a ‘cytokine storm’ can occur, as evidenced by the presence of high levels of proinflammatory cytokines, such as IL-2, IL-7, IL-10, TNFα, IFNγ-induced protein 10, granulocyte colony-stimulating factor, monocyte chemoattractant protein-1, macrophage inflammatory protein-1 α in sera of these patients.2,3 There are several ongoing trials on targeted therapy to reduce this cytokine storm by inhibiting cytokine response at receptor level and its subsequent pathways.4
THERAPEUTIC PLASMA EXCHANGE
Currently, there are sufficient evidence from the literature to support the role of plasma exchange therapy in various cytokine storm-induced diseases, such as thrombotic thrombocytopenic purpura and cytokine release syndrome, and its effectiveness in reducing the inflammatory markers of a cytokine storm.5-7 In 2015, Liu et al.8 reported the efficacy of therapeutic plasma exchange (TPE) in severe avian influenza disease with a good survival benefit. They also showed that a combination of plasma exchange with continuous venovenous haemofiltration had a better outcome, the latter assisted in maintaining a haemodynamic stability in those with septic shock.
In China, during the present SARS CoV-2 pandemic, Xu et al.9 used artificial liver blood-purification system in severe COVID-19 disease based on their previous experience in managing severe avian influenza with good survival benefit. As a consequence of the present and past experiences, the National Clinical Research Centre for Infectious Diseases in China has placed forthwith expert committee recommendations and guidelines for artificial-liver blood-purification system in the treatment of patients with severe COVID-19. Recent studies by Khamis et al.10 and Shi et al.11 revealed improvements in clinical outcome of severe COVID-19 infections by using TPE.
CONVALESCENT PLASMA THERAPY
One other salvage modality would be to transfuse plasma from patients who have recovered from COVID-19, i.e., convalescent plasma therapy (CPT) that contains high levels of neutralising antibodies to patients with severe presentations of the disease that require either oxygen therapy or those on ventilator support. This form of therapy was used as early as 1918 and 1957 during the ‘flu epidemics’ and more recently for SARS, Middle-East respiratory syndrome (MERS), and Ebola pandemics.12,13 This form of therapy has also been used by the present authors in patients with liver disease who are undergoing liver transplantation and are positive for hepatitis B virus. Plasma rich with anti-hepatitis B virus antibodies, known as hyper immune plasma, was given in a dose of 2,000 IU/L to maintain adequate anti-hepatitis B virus antibody levels to prevent hepatitis B virus recurrence.14
In a press release from February 2020 (unpublished data), China reported that it used plasma of convalescent individuals in 245 patients with COVID-19 and reported good survival benefits. Zhang B et al.15 published their first case series on effectiveness of convalescent plasma in four patients with severe COVID-19, who were on ventilator support with more than one organ failure and who did not show clinical or biochemical improvement after standard recommended therapy. Following CPT, SARS CoV-2 reverse transcription PCR was negative within 3–22 days, and anti-SARS CoV-2 IgG levels were detected 4 days post reverse transcription PCR negativity.
Another benefit forthcoming with use of CPT was the reduction in severity of entry of SARS CoV-2 by cross-neutralisation, further suggesting that convalescent sera of SARS CoV-2 had neutralising antibodies.4 These virus-specific antibodies usually peak at 4 months and gradually taper over the next 24 months.16 Studies in animal models have shown that anti-SARS CoV-2 IgG is likely to protect an individual from subsequent exposures too.17 Possible mechanisms of convalescent plasma in COVID-19 includes direct neutralisation of virus; control of overactive immune system, such as cytokine storm; and immunomodulation of hypercoagulable state.18
COMBINED EFFECT OF THERAPEUTIC PLASMA EXCHANGE WITH CONVALESCENT PLASMA THERAPY
Based on the above literature evidence on the role of CPT as well as TPE in managing the cytokine storm, there is a case series of novel therapeutic approaches, such as TPE using SARS CoV-2 CP from recovered individuals in patients with severe COVID-19.19 This will not only reduce the cytokine storm, but also provide the patients with virus-specific neutralising antibodies and thereby improve the overall survival of patients who have organ failure in one or more organ. This study emphasises the importance of the timing of immunomodulatory treatments. As IL-6 level will be peak in between 7 and 14 days after the onset of symptoms, early initiation of TPE with CP would be beneficial to patients with COVID-19 who are symptomatic. Interestingly, the literature review by Kesici et al.19 revealed that no major adverse events have been reported by using TPE or CP or the combination of both in patients infected with SARS CoV-2, although it might increase procoagulant state of the patients because it involves 5% albumin or fresh frozen plasma. Moreover, Jaiswal et al.’s20 study on patients with severe COVID-19 who required mechanical ventilator support showed significant clinical improvement with use of TPE with CP. Table 1 summarises the differences in the transfusion of CPT with and without TPE. There are case reports of TPE with intravenous Ig in a patient critically ill with COVID-19.11
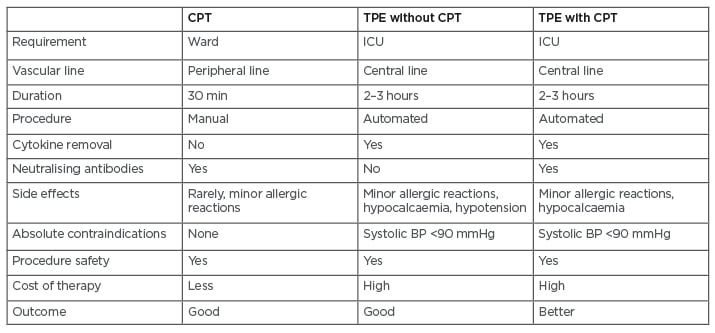
Table 1: Differences between convalescent plasma therapy, therapeutic plasma exchange, and the combination of convalescent plasma therapy with therapeutic plasma exchange in patients with severe coronavirus disease (COVID-19).
BP: blood pressure; CPT: convalescent plasma therapy; ICU: intensive care unit; TPE: therapeutic plasma exchange.
CONCLUSION
Early intervention of CP as well as TPE has showed benefit to patients symptomatic with COVID-19. Combination of CPT with CP novel modality is likely to circumvent the imminent mortality in patients with severe COVID-19, while scientists and researchers are working on vaccines and other pharmacotherapeutic agents for preventing mass transmission in the community.







