Abstract
Percutaneous coronary intervention (PCI) has become an integral step in the management of patients with ischaemic heart disease. Bifurcation lesions are commonly encountered in PCI and are regarded as the most technically challenging lesions to treat. The literature is saturated with studies that explore the best way to treat these lesions, with a variety of techniques introduced to enable stenting of both vessels. However, the optimal method is still undetermined. In this state-of-the-art review, we discuss the various strategies that are currently regarded as ideal, particularly those which involve one or two-stent techniques. Moreover, dedicated bifurcation devices have been developed to facilitate bifurcation stenting and improve outcomes. The role of these devices in this context is also uncertain, therefore we will discuss the current evidence for their use. Finally, a road map on the ‘tips and tricks’ to successfully perform bifurcation PCI is mentioned, which will aid any interventionist performing this procedure in an optimal manner.
INTRODUCTION
Dr Andreas Gruentzig performed the first coronary angioplasty in 1977;1 since then percutaneous coronary intervention (PCI) has become increasingly sophisticated, enabling treatment of complex lesions. PCI has a central role in the management of patients with ischaemic heart disease, as discussed in the recent European Society of Cardiology (ESC) guidelines,2 and nearly 600,000 PCIs are performed each year in the USA,3 compared with 96,143 cases performed in 2014 in the UK.4 Bifurcation lesions are encountered in up to 20% of patients undergoing PCI.5 They are challenging, complex, and often encountered in high-risk surgical candidates, such as the elderly or frail, or those with high SYNTAX scores,6 where bifurcation stenting is the only realistic revascularisation option. In general, a simplified interventional strategy with a ‘less is more’ approach is regarded as best practice. However, with the introduction of dedicated bifurcation devices, questions have arisen around this belief (Figure 1).
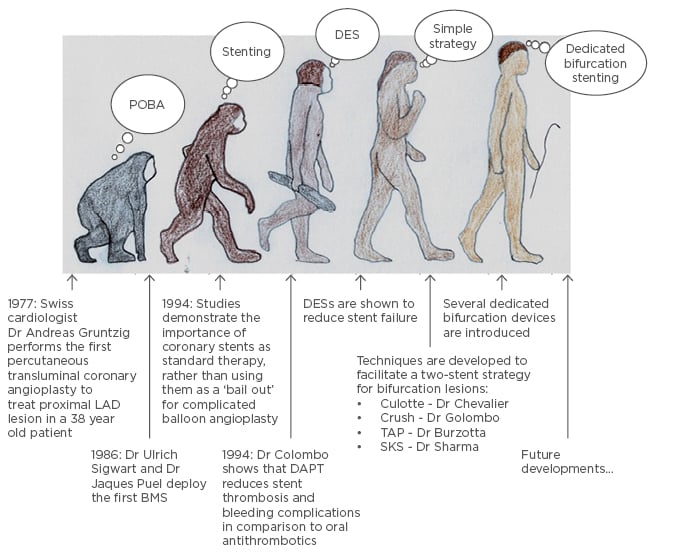
Figure 1: The evolution of percutaneous coronary intervention, from simple angioplasty to complex intervention.
A one (simple) or two-stent strategy can be used to treat bifurcation devices facilitating stenting of both vessels.
BMS: bare-metal stent; DAPT: dual antiplatelet therapy; DES: drug-eluting stent; LAD: left anterior descending artery; POBA: plain old balloon angioplasty; TAP: T and protrusion technique; SKS: simultaneous kissing stent technique.
CLASSIFICATION
The European Bifurcation Club (EBC) has endorsed the Medina classification to describe bifurcation lesions, and the MADS (main, across, distal, side) classification to describe the various ways to start bifurcation PCI.7
The Medina classification consists of three numbers.8 Within this, a score of 1 is used to denote a vessel with a stenosis of ≥50%; otherwise a score of 0 is given. The first number describes the proximal main vessel (MV), the second the distal MV, and the third the side branch (SB). A true bifurcation lesion is regarded as either 1,1,1; 0,1,1; or 1,0,1. It is important to appreciate that the Medina classification only provides an anatomical description of the lesion and not a physiological one. Studies using fractional flow reserve (FFR) have demonstrated a negative correlation between the percentage stenosis of the SB post MV stenting as determined by quantitative coronary angiography (QCA) and FFR assessment (r=-0.41, p<0.001), with FFR demonstrating that only 27% of lesions deemed significant by QCA are functionally significant.9 These data support a single stent strategy in the majority of lesions.
Under the MADS classification (as defined previously), ‘M’ involves stenting of the proximal MV first; ‘A’ is the placement of the stent across the SB first; ‘D’ is the placement of the first stent into both distal branches or at the ostium; and finally ‘S’ indicates when the SB is stented first.7,10 Whichever technique the operator starts with can lead to further stenting, if required.
APPROACH TO BIFURCATION STENTING
The decision to use a one or two-stent strategy ought to be made prior to starting the procedure. With a single stent or provisional technique, the MV is stented first. Then, only if the SB is compromised and judged to be significant is subsequent SB stenting carried out with the T technique. With a two-stent or complex procedure, the operator decides if both MV and SB need stenting from the outset. Following this, we summarise the current literature and evidence for the differing complex approaches (outlined in Figure 2). All techniques begin with the wiring of both vessels, followed by balloon dilation. Procedures should finish with a final kissing balloon inflation (FKBI), which involves ballooning both the MV and SB simultaneously. FKBI has been shown to significantly reduce major adverse cardiovascular events (MACE).11
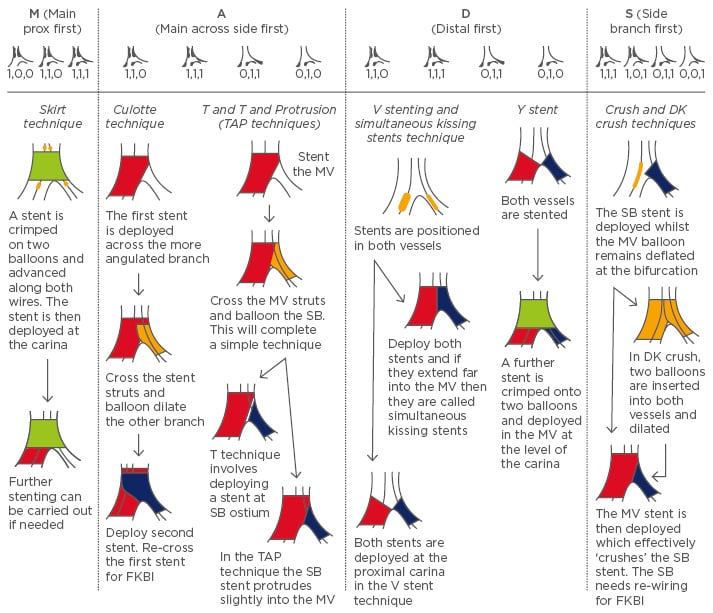
Figure 2: Different complex technique procedures according to MADS (Main, Across, Distal, Side) and how this relates to the Medina classification.
All techniques begin with wiring of both the main vessel (MV) and side branch (SB) followed by balloon dilation. With the complex strategy, the procedures end with a final kissing balloon inflation (FKBI) of both vessels.
Wire on top of a stent means the stent struts are crossed.
Red denotes MV stenting; Blue denotes SB; Green is for proximal MV stenting to the carina; Yellow denotes a balloon.
TAP: T and Protrusion; DK: double kissing.
Skirt Technique
The skirt technique involves crimping a stent onto two balloons and advancing them along the two wires until the carina is reached. The stent is then deployed. The main drawback of this procedure is the need to manually crimp the stent as it can disrupt the polymer in drug-eluting stents (DESs).12
The T Technique, and T and Protrusion Technique
The T technique involves deploying a stent in the MV and then re-wiring the SB through these struts to place a further stent. It treats proximal MV lesions but leads to incomplete coverage of the ostial SB.12 A slight modification, called the ‘T and Protrusion (TAP) technique’ allows the SB stent to protrude slightly into the MV to ensure ostial coverage without significant disturbance to the MV struts.
The Culotte Technique
The culotte stenting technique achieves full coverage of the bifurcation. It involves wiring both vessels and placing a stent across the more angulated branch. The other vessel is then re-wired through these struts and a second stent is deployed. FKBI is performed after re-wiring of the first vessel, again through the struts of the second stent.13 A culotte can be used in all bifurcation angles but is time consuming, and limited by the creation of a two-stent layer.12 Additionally, the culotte as opposed to the crush technique requires vessels of similar sizes.
V and Simultaneous Kissing Stent Technique
The V technique involves advancing stents into the SB and distal MV and then inflating them both to create a proximal carina.14 It can only be used if the proximal MV is free from significant disease. If the carina extends a significant amount into the proximal MV, then it is called the simultaneous kissing stent technique.15 It is useful in bifurcations with a tight proximal stenosis in the MV. The major advantage of these techniques is that wire access to either branch is maintained; however, treating a proximal lesion after using the V technique can be very difficult and requires re-wiring of the SB.12
Y Technique
The Y technique involves deploying a stent in both the distal MV and SB. A further stent is then placed in the proximal MV over two balloons and deployed at the carina.12 This ensures all three stents are covered.
Crush Technique
The crush technique involves placing un-inflated stents in both vessels and then deploying the SB stent followed by removal of its wire. The MV stent is then deployed and the balloon splays the struts of the SB, effectively ’crushing’ them.16 This enables good coverage of the ostium of the SB; however, re-wiring the SB for FKBI can be difficult and time consuming.12
The Reverse Crush Technique
The reverse crush technique is often used when a suboptimal result is achieved with provisional SB stenting; in cases of inadequate ostial SB appearance, another stent is advanced into the SB and retracted 2–3 mm into the MV, with another balloon advanced into the MV at the level of the bifurcation. The SB stent is deployed first followed by inflation of the MV balloon.
Double Kissing Crush Technique
The double kissing (DK) crush stenting technique involves stenting the SB first and then inflation of a balloon in the MV to crush the first stent. A first kissing balloon inflation is performed by placing balloons in both vessels and inflating them. Then the MV stent is deployed followed by an FKBI.17
Keep It Open Strategy
Unlike the other strategies described, the keep it open (KIO) strategy does not involve balloon dilation of the SB or FKBI. With this strategy, the MV is treated whilst the jailed wire is left in the SB to KIO. It is used if the SB is too small to stent, or is clinically irrelevant but has ostial disease.5
Whichever technique is used, the patency of vessels needs to be as optimal as possible and they should not be covered with multiple layers of stents.
PROVISIONAL OR COMPLEX STRATEGY
Several studies have assessed which strategy is superior, and these are discussed in Table 1.17-25 We focus primarily on the use of DESs, as it is well established that a complex approach using bare- metal stents (BMSs) yields much higher rates of MACE and restenosis of the MV and SB than with a DES, hence contemporary practice is to use DESs for bifurcations.26 A meta-analysis pooled studies that used DESs in order to compare these two approaches. There was a similar risk of cardiac death (odds ratio [OR]: 0.99; 95% confidence interval [CI]: 0.4–2.41, p=0.98), target-lesion revascularisation (TLR; OR: 1.72; 95% CI: 0.95–3.12, p=0.07), and target-vessel revascularisation (TVR; OR: 1.59, 95% CI 0.94–2.69, p=0.07) between the simple and complex approaches.27 However, there was a significantly lower incidence of myocardial infarction (MI) at follow-up (OR: 0.6; 95% CI: 0.43–0.86, p=0.005) after simple strategy surgery.27 However, the DK-CRUSH-II trial showed that rates of MI were similar between the DK crush and simple strategies (3.2% versus 2.2%, p=0.751).17 This trial only included true bifurcation lesions and a SB of ≥2.5 mm, which may account for these differences.17 Given these disparities, we need to be careful when concluding that complex approaches truly have a higher risk of MI.
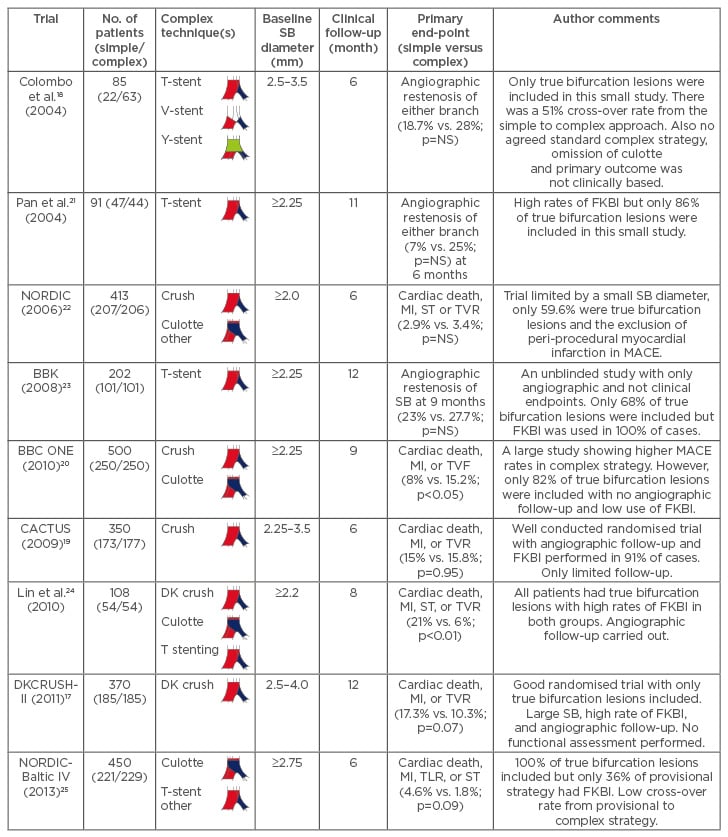
Table 1: The current studies that compare a simple to a complex strategy using drug-eluting stents.
It is important to note that FKBI is associated with better outcomes and a complex strategy should only be considered in a true bifurcation lesion (1,1,1; 1,0,1; or 0,1,1).
FKBI: final kissing balloon inflation; MI: myocardial infarction; MV: main vessel; SB: side branch; ST: stent thrombosis; TVF: target-vessel failure; TVR: target-vessel revascularisation; TLR: target-lesion revascularisation; MACE: major adverse cardiovascular events; NS: not significant.
The EBC consensus states that the provisional approach should be the preferred technique for most bifurcations.7 However, if there is significant SB ostial disease or if the vessel is particularly large and supplies a large area of myocardium, then a complex strategy should be used.7
TWO-STENT TECHNIQUES: ARE THERE DIFFERENCES BETWEEN VARYING STRATEGIES?
On the whole, the majority of interventionists prefer the provisional approach; however, with ‘true’ bifurcation lesions (Medina 1,1,1; 1,0,1; 0,1,1) in large vessels a complex two-stent technique is sometimes necessary. In particular, if a provisional approach results in injury, recoil, or dissection in a large SB, one has to commit to two stents. Below we describe the current literature comparing the techniques described in Figure 2, including the clinical and angiographic outcomes.
The Culotte Technique Versus Crush Techniques
The first randomised study comparing culotte and crush techniques was from the NORDIC PCI group.15 The majority of patients included had true bifurcation lesions with SB angulations <70°.28 At 6 months the number of MACE was comparable between culotte and crush (3.7% versus 4.3%, p=0.87), but at the 8-month angiographic follow- up, culotte was associated with significantly less in-stent restenosis (4.5% versus 10.5%, p=0.046).28
Further studies over 36 months have confirmed these findings with similar MACE rates in both culotte and crush (16.7% versus 20.6%, p=0.32).29 It should be noted, however, that the majority of the increased MACE is due to SB TVR, and whether this is of clinical relevance or just an angiographic anomaly related to the procedure remains to be seen. There was a trend towards increased restenosis rates in the crush group, but this did not translate into increased TLR29 as cardiologists may have thought the restenosis did not need revascularisation, hence this may be related to the aggressiveness of the interventionists’ treatment style. Studies focussing on ischaemic-driven TLR are therefore more clinically relevant. There were lower rates of FKBI in the crush group (crush 84.3% versus culotte 91.6%, p=0.02),29 probably owing to the difficulty of crossing multiple stent layers; however, this did not translate into higher rates of stent thrombosis.
The Culotte Technique Versus the Double Kissing Crush Technique
The culotte and double kissing crush techniques were compared in a randomised trial involving unprotected distal left main bifurcation lesions. The culotte technique had significantly higher 1-year MACE rates compared to DK crush (16.3% versus 6.2%, p<0.05).30 This was mainly driven by the need for TVR. These improved MACE rates were also apparent in lesions with a bifurcation angle ≥70°. This would suggest that refinement of a full-crush technique into either a mini-crush or DK crush has more favourable outcomes and it is currently the preferred approach.
Culotte Stenting Versus T Stenting and T and Protrusion Stenting
A retrospective study compared culotte with T stenting, and showed improved MACE rates at 9 months with the former technique (13.3% versus 27.3%, p=0.051).31 Indeed, there was significantly lower residual stenosis at the SB ostium with culotte compared with T stenting (3.44±7.39% versus 12.55±11.47%, p<0.0001).31
Complex techniques were compared in a study investigating strut apposition and stent coverage of the bifurcation lesion. It is recognised that stent malapposition leads to an increased risk of late stent thrombosis.32 This small study compared culotte, crush, and T/TAP techniques using micro-computed tomography.32 They found higher rates of malapposition within the bifurcation with crush compared to culotte or T/TAP techniques (41.5±8.2%, 31.4±5.2%, and 36.7±8.0%, respectively).32 The percentage of residual stenosis at the SB ostium was similar with all techniques (32.7%, 29.2%, and 25.9%, respectively).32 However, there was a significantly higher rate of strut malapposition in the proximal vessel with the crush and culotte techniques compared with T/TAP (39.1±10.7%, 26.1±7.7%, and 4.2±7.2%, respectively, p<0.01).32 The study highlighted that even after FKBI, strut apposition remained poor in all complex techniques.
Crush Versus T Stenting
A retrospective study compared crush to T stenting of bifurcation lesions. At 1-year follow- up culotte, as opposed to T stenting, had lower rates of TLR (14% versus 31.1%, p=0.01) and TVR (16.5% versus 32.8%, p=0.02).33 Indeed, there was significantly less restenosis of the SB with culotte compared with T stenting when FKBI was used (8.6% versus 26.5%, p=0.04).33
Bioresorbable Vascular Scaffold in Bifurcations
The future use of bioresorbable vascular scaffolds (BVS) is of interest as it is possible that struts across the SB in large vessels will eventually dissipate, and so as long as patients remain on prolonged dual antiplatelet therapy, no adverse outcome can ensue. This hypothesis may also hold for patients with complex layers of scaffold. Some case reports with reasonable outcomes have been described so far; however, randomised trials are still required and the current recommendation is against the use of BVS for bifurcation lesions.34,35
DEDICATED BIFURCATION DEVICES
Dedicated bifurcation devices have been used in an attempt to overcome the problems associated with complex strategies by enabling both vessels to be more easily stented using a single delivery device. They are broadly divided into three categories:36
Devices that are deployed in the main vessel and facilitate side branch stenting by maintaining access to it, usually via side-ports
These devices facilitate provisional T or V stent techniques, with examples including:
- Antares® (Trireme Medical, USA): This BMS has an opening aperture in the middle of its shaft to facilitate T stenting. No adverse events were reported at 30 days in a small first-in-man (FIM) study.37
- BioSS Expert stent® (Balton, Poland): This DES is advanced into the MV until a marker is positioned at the carina, which identifies a zone with reduced struts to facilitate SB stenting using the T technique. It had an 11.1% TLR rate at 1 year.38
- Drug-Eluting Balloons (DEBs):39 There is evidence in small studies that support the use of DEB for the treatment of in-stent restenosis. There has been some use of DEB in the SB of a provisional T approach and it has also been utilised before or after the use of dedicated BMS bifurcation devices such as Tryton™ (see following). In patients that cannot have dual antiplatelet therapy or are frail, and in whom plain old balloon angioplasty is considered for the bifurcation lesion alone, FKBI DEB can be used. However, the precise role of DEB in bifurcation lesions requires further investigation.
- Invatec Twin-Rail (Invatec S.R.L, Italy): This BMS is pre-mounted on double balloons and the stent is deployed by simultaneous kissing inflation using a single indeflator. It aids a provisional T approach and had a 14.3% MACE rate at 7 months in a small FIM study.37
- Multi-Link Frontier™ (Abbott Vascular Devices, USA): This BMS is pre-mounted on double balloons, which are joined by a mandrel. The mandrel is retracted when the device is at the carina, releasing the SB balloon to enable T stenting.40 It has now been discontinued.
- Nile Croco® (Minvasys, France): A BMS is crimped on two balloons pre-mounted onto two catheters. The MV balloon has a marker on it indicating the position of the SB orifice, thus when the stent is deployed, the SB balloon can be advanced into the SB. It had a 14% MACE rate at 6 months.41
- Petal™ (Boston Scientific, USA): This device contains an aperture in the mid section of the DES, with deployable struts to cover the ostium of the SB. It had a 7% TLR rate at 1 year in a FIM study.42
- SLK-view™ (Advanced Stent Technologies, USA): This BMS contains side apertures which enable SB access with a provisional T technique, although it is no longer available.37
- Stentys® (Stentys, USA): This is not a true dedicated bifurcation stent but the links of the DES can be removed by balloon angioplasty. It had a 3.7% MACE rate at 1 year.43
- Xience SBA (Abbott vascular devices, USA): This had an opening port in the body of the DES for SB access44 but it never came to market.
- Y-Med Side-Kick™ (Y-Med, USA): This device contains exit ports that enable the SB to be wired when the BMS is in the MV. It facilitates the T approach and had a 5.8% MACE rate at 3 months.37
Devices that are used to treat the side branch first with subsequent main vessel stenting
Examples include:
- Sideguard® (ArraVasc, Ireland): This is a self-expanding BMS, mounted on a balloon delivery system enabling its precise delivery to the SB. Further MV stenting is carried out using the crush or T techniques.45 It is no longer available.
- Biguard™ (Lepu Medical Ltd., China): This device uses a DES to stent the SB with facilitated access to the MV through its wider struts to enable a provisional T, culotte, or DK crush approach. It had a 10.6% MACE at 12 months.46
- Tryton™ (Tryton, USA): This is similar to the culotte approach due to the use of a BMS made of three zones divided into a SB, MV, and transitional zones. The stent is deployed in the SB with the transition zone facilitating MV stenting.47
Conical stents that cover the whole bifurcation
One example of a conical stent is the Axxess stent™ (Devax, USA). This self-expanding device is advanced along the wire that has the steepest bend to the proximal MV, until markers on the DES are in the distal vessel. The sheath is gently retracted enabling markers to flare into the opposite vessel. The device is gently advanced whilst the sheath is fully retracted to allow full deployment.48
A characteristic of many of these new devices is the high rate of procedural success. The Axxess and Tryton stents will be discussed further given their common use in clinical practice and larger study populations. The DIVERGE study investigated clinical outcomes with the Axxess stent with a follow-up of 3 years.48 Further treatment with a CYPHER® stent was advised if there was residual stenosis in any vessel of >30% or if there was evidence of dissection.48 The cumulative MACE rate was 9.3% at 1 year and 16.1% at 3 years.48 These outcomes were comparable to other studies investigating complex approaches. In CACTUS, the 6-month MACE rate was similar to the 3-year rates found in DIVERGE.19 3-year MACE rates after PCI in bifurcation lesions in patients with multivessel disease were similar to those seen in DIVERGE.49 Although the initial results are promising, further randomised trials are needed.
The efficacy of the Tryton device was analysed in the TRYTON study, which compared it to SB balloon angioplasty.47 At 9 months, the primary endpoint of TVF occurred in 17.4% of the bifurcation stent group, but also in 12.8% of the provisional group (p=0.11), meaning it did not meet the non-inferiority endpoint.47 However, there was reduced stenosis in the SB with the Tryton device, although this did not relate to differences in binary restenosis rates.47 Only 41% of the SB diameter inclusion criteria of ≥2.25 mm was met, which may have adversely affected these results.
TIPS AND TRICKS
With a provisional approach, we suggest leaving a jailed wire in the SB to KIO and help facilitate location of the vessel if SB stenting is required at a later point. It is often useful to wire the more difficult branch first to ensure the wire does not twist around an already placed wire as a result of excessive manipulation.
Difficulty with Side Branch Access
Initially try different wire tip curves or guidewires. A longer and wider curve can improve access to an occluded SB.50 A hydrophilic guidewire allows easier recrossing into the SB but carries a risk of dissection, whereas a stiffer wire enables better precision using torque but involves difficulties with manipulation.50 Alternatively, the ’reverse wire technique’ can be used which involves advancing the curved wire into the distal MV and slowly retracting it towards the SB until it is engaged.7 It can then be advanced into the SB. If these techniques fail, then MV balloon predilation can help to change the angle with the SB but this does pose a risk of plaque rupture into the SB.50 Additionally, rotational atherectomy can debulk ostial SB lesions that have a high plaque burden.50 Finally, if these measures fail than placing a microcatheter at the level of the carina and exchanging various stiffer or steerable wires through it can help entry into the SB; the Pilot family of wires are particularly useful in this regard.
Difficulty Re-wiring the Side Branch Through the Stented Main Vessel Struts
If the aforementioned tips fail, then try the proximal optimisation technique. This involves using a short, oversized, non-compliant balloon to expand the proximal stent to just before the carina such that the angle between the MV and SB are changed to facilitate SB re-wiring.7 Another technique involves using the SB jailed wire to inflate a small balloon at the SB ostium to help SB re-wiring.50 Once access has been achieved, the MV stent will require balloon dilation and FKBI. Finally, the placement of a proximal guide extension device such as a guideliner or a mother- in-child technique will facilitate the crossing of balloons and stents into the SB. In general, noncompliant balloons are recommended.
Final Kissing Balloon Inflation and Two-Step Kissing Balloon Inflation
It is important to use noncompliant balloons to enable high pressure inflations and more predictable expansion. This is evidenced from intravascular ultrasound studies and bench testing.51 FKBI is recommended for all complex strategies and can be useful in provisional stenting if a significant SB stenosis remains after MV stenting.7 The COBIS registry showed that routine use of FKBI after simple stenting can be harmful due to increased rates of TLR.52 Lastly, a refinement to KBI has recently been demonstrated to be more effective at reducing SB ostial stenosis; by performing a two-step KBI, which involves placing a noncompliant balloon in the SB and performing a prolonged inflation at high pressures (>20 atm), ostium can be opened before FKBI.53
CONCLUSION
Coronary intervention has evolved through the years to produce more complex PCI. Although several studies support the ‘less is more’ notion for bifurcation lesions, there are circumstances when a complex approach is needed. The introduction of dedicated devices has simplified this strategy but it remains to be determined if they improve outcomes. The use of FFR and intracoronary imaging (optical coherence tomography or intravascular ultrasound) also facilitates choice of strategy and reduces unnecessary lesion treatment, ensuring adequate final stent optimisation in both the MV and the SB. With further device and procedural refinement, we envisage that dedicated devices will eventually play a central role in the treatment of true bifurcation lesions.