Abstract
Background: Aspergillus peritonitis is a rare, potentially fatal complication of continuous ambulatory peritoneal dialysis (CAPD). Several cases of fungal peritonitis (FP) caused by Aspergillus species have been reported worldwide; however, there is a paucity of data from Pakistan. Here, a case of congenital nephrotic syndrome is reported in a patient who progressed to Stage 5 chronic kidney disease when she was 8 years old, which was managed by CAPD and other supportive therapies. The patient developed FP and later succumbed to death despite appropriate management. Early diagnosis and careful interpretation of culture results are, therefore, important for the treatment of these patients.
Case presentation: The patient outlined in this case report had congenital nephrotic syndrome, Stage 5 chronic kidney disease, and was on CAPD. She presented with peritonitis. Initially, broad spectrum antibiotics were started, and peritoneal samples did not yield any growth. Later, consecutive peritoneal samples taken from the patient grew Aspergillus terreus, with septate hyphae seen on a direct smear from the sample. Voriconazole was started immediately, and the patients’ catheter was removed. However, the patient’s condition deteriorated and, after prolonged intensive care and high ventilator support, the patient expired.
Conclusion: A. terreus is an uncommon and deadly pathogen that does not respond to the commonly used antifungal treatments such as amphotericin B. The treatment of CAPD-related FP requires early diagnosis and the use of newer drugs such as voriconazole or caspofungin.
BACKGROUND
Continuous ambulatory peritoneal dialysis (CAPD) is a well-known management technique for paediatric Stage 5 chronic kidney disease (CKD-5) used worldwide.1,2 However, the majority of children in Pakistan are dialysed by haemodialysis (HD), and very few receive successful kidney transplants. CAPD is rarely practiced in Pakistan due to the scarcity of costly dialysis consumables and a lack of education and trained staff.3,4 Peritonitis and catheter-related infections are a major cause of CAPD failure, resulting in switching to HD or death. Although bacterial peritonitis (BP) is the most common complication, fungal peritonitis (FP) is a rare and serious complication of CAPD. The risk factors for FP include recurrent BP, use of broad-spectrum antibiotics, prolonged hospitalisation, underlying proteinuria, immunosuppression, prolonged duration of CAPD, and frequent manipulation of the peritoneal dialysis (PD) catheter. FP is an indication of switching to HD and removing the catheter.
Several cases of FP caused by Aspergillus species (spp.) have been reported worldwide; however, there is a paucity of data from Pakistan.5-8 Here, a case of congenital nephrotic syndrome (CNS) is reported in a patient who progressed to CKD-5 when she was 8 years old, which was managed by CAPD and other supportive therapies. She developed recurrent episodes of BP and ultimately FP and succumbed to death despite use of aggressive systemic and intraperitoneal (IP) antibiotics and antifungal agents, removal of the PD catheter, switching to HD, and receiving intensive ventilator care. Early diagnosis and careful interpretation of culture results are therefore important for the treatment of these patients.
CASE PRESENTATION
The patient in this case report was an 8-year-old female who had CNS since she was 1 month old. Her younger brother also had CNS and had previously expired. She was diagnosed with CNS at another hospital, where she was managed with intravenous albumin, along with diuretics administered periodically. However, genetic or immunological investigations or a kidney biopsy were not carried out. The patient was treated with enalapril and oral diuretics, but not with steroids or any other immunosuppressant. She was asthmatic since early childhood, requiring frequent nebulisation, and had been on and off on montelukast. The patient underwent conservative treatment for CKD at the primary institute for almost 15 months but, due to persistent symptomatic asthma not fulfilling the general fitness required for anaesthesia, long-term vascular access could not be constructed, and she reached CKD-5. Despite use of thyroxine supplements, she exhibited developmental delay with hyperactive behaviour.
The patient was referred to the authors’ institute for CAPD in September 2020. The clinical details of the patient are listed here (Table 1). The patient was diagnosed with generalised oedema, hypoalbuminaemia, severe hypertension, severe anaemia, and symptomatic uraemia, with a serum creatinine level of 11.68 mg/dL and an estimated glomerular filtration rate of 5.26 mL/min/1.73 m2,confirming CKD-5. Her growth parameters (weight: 18 kg; height: 112 cm) were below the third centile for her age group, suggesting long-standing CKD. The patient underwent management of hypertension, anaemia, and uraemia with acute HD. After the laparoscopic placement of a Tenckhoff catheter, CAPD was initiated; the CAPD prescription was 5 exchanges/day with a fill volume of 400 mL of 1.5% dextrose and ultrafiltration of 350–400 mL.
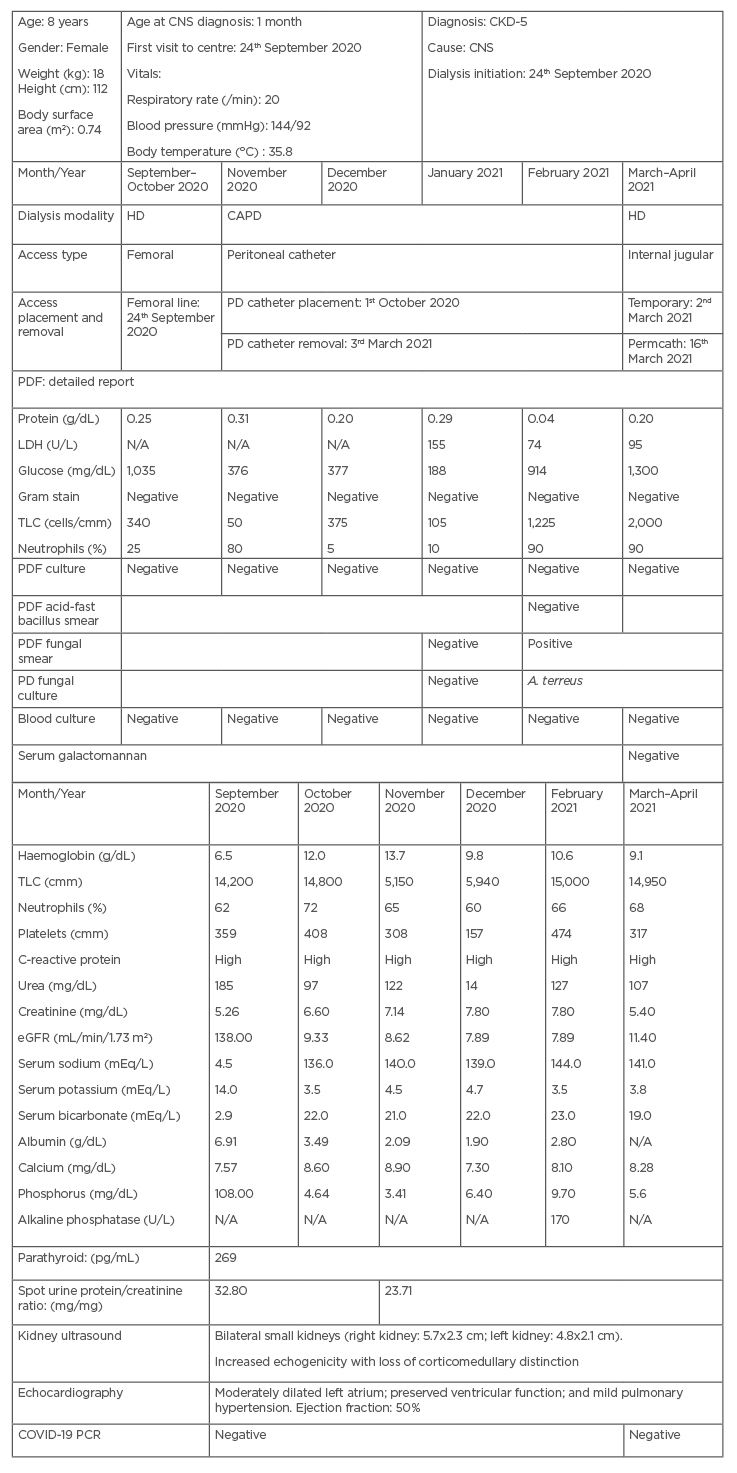
Table 1: The characteristics of the patient outlined in this case report with Aspergillus terreus peritonitis on continuous ambulatory peritoneal dialysis for Stage 5 chronic kidney disease.
A. terreus: Aspergillus terreus; CAPD: continuous ambulatory peritoneal dialysis; CKD-5: Stage 5 chronic kidney disease; CNS: congenital nephrotic syndrome; eGFR: estimated glomerular filtration rate; HD: haemodialysis; LDH: lactate dehydrogenase; N/A: not applicable; PD: peritoneal dialysis; PDF: peritoneal dialysis fluid; TLC: total leukocyte count.
The patient had recurrent episodes of BP during CAPD, as suggested by the high cell count in the PD fluid analysis, though the cultures remained negative (Table 1). Each episode was treated with IP antimicrobials, with clinical and PD fluid count improvement. Lastly, she presented with high-grade fever with associated chills and rigors for 5 days. Initially, IP antibiotics were started after sending PD-effluent samples for analysis and culture, but the patient’s condition did not improve after 7–10 days. She remained sick, with continuous dull pain, fever, and occasional vomiting, and her levels of C-reactive protein increased from 24 to 48 mg/L. PD fluid analysis showed a very high cell count (1,225 /mm3) with more than 90% polymorphs. Bacterial cultures and the acid-fast bacilli smear were negative.
Subsequent dialysate effluent samples revealed septate hyphae on microscopy with 3% potassium hydroxide. After 3–5 days of incubation at 37 °C, Sabouraud dextrose agar yielded buff tan, cinnamon brown, velvety, powdery colonies with a yellow tan reverse (Figure 1). Microscopic examination with lactophenol cotton blue solution revealed septate hyphae; smooth, relatively short conidiophores; and vesicles with smooth, round conidia on compactly columnar, biseriate phialides, equal in length to the metulae and covering the upper half of the vesicle. It was identified phenotypically as Aspergillus terreus (Figure 2).9

Figure 1: Macroscopic colony morphology of Aspergillus terreus culture on Sabouraud dextrose agar.
The colonies observed were buff tan, cinnamon brown, velvety, and powdery, with a yellow tan reverse.
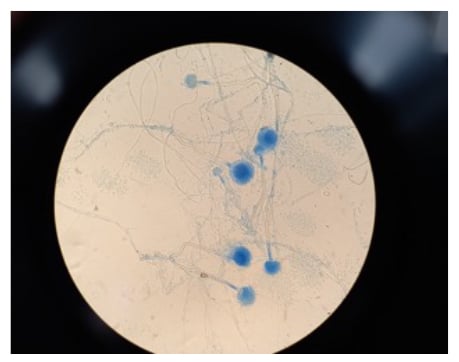
Figure 2: Microscopic appearance of Aspergillus terreus, stained using lactophenol cotton blue solution, on Sabouraud dextrose agar (magnification x40).
This image shows compactly columnar, biseriate phialides covering the upper half of the vesicle.
Voriconazole was started and continued in the recommended systemic and intraperitoneal dose, the CAPD catheter was removed, and the patient was switched to HD through a temporary double-lumen catheter, which was later replaced by a permcath. However, after 2–3 weeks, she presented with hypertensive encephalopathy and respiratory distress. Her serum galactomannan test was negative, so invasive aspergillosis was ruled out. The patient’s condition deteriorated, requiring mechanical ventilator support, and her Glasgow Coma Scale (GCS) score declined. After a prolonged stay under intensive care and high ventilator support, the patient developed ventilator-associated pneumothorax and subsequently expired.
DISCUSSION
The authors describe a case of FP with A. terreus in an 8-year-old child from Pakistan with CKD-5 secondary to CNS on CAPD. From the authors’ experience, this is the first reported case of a CNS patient who survived and underwent CAPD, since most children die during the first 2–3 years of their diagnoses, often as a result of intercurrent infections rather than uraemia. Though CNS was not confirmed genotypically but was onset during early infancy, due to the death of the patient’s younger brother from CNS and the persistence of oedema, nephrotic range proteinuria, and hypoalbuminaemia in the patient, her progression to CKD-5 at this age strongly favours the diagnosis of CNS.
FP accounts for 3–10% of cases of CAPD-related peritonitis.6,10,11 In a study from Saudi Arabia, patients with CNS and those younger than 5 years old had a higher incidence of FP, with a higher rate of catheter removal than in patients diagnosed with BP.12 FP is mostly caused by Candida spp. In a study from India,among 142 episodes of peritonitis, 20 (14%) cases were due to FP.13 Prior exposure to antibiotics was seen in nine cases, 18 cases were due to Candida spp., 12 cases recovered and CAPD was re-initiated, the modality was changed to HD in four cases, and the patients expired in four cases.13 Filamentous fungi less frequently cause FP; however, different types of filamentous fungi have been reported to cause peritonitis, including Aspergillus, Paecilomyces, Penicillium, and Acremonium spp.5,7,14 Although Aspergillus spp. rarely cause FP (2–5% of cases), it has a high morbidity and mortality rate and its diagnosis and treatment is challenging.5,8,14 Multiple Aspergillus spp. have been reported in FP, including A. terreus, Aspergillus fumigatus, Aspergillus niger, Aspergillus flavus, Aspergillus sydowii, and Aspergillus oryzae.6,14Aspergillus spp. is an environmental organism and may enter the peritoneal cavity via dialysis catheters.
Factors like malnutrition, hypoalbuminaemia, left ventricular dysfunction, and anaemia are well-established risk factors for high mortality in children on dialysis.5 Persistent nephrotic range proteinuria and hypoalbuminaemia, poor dietary intake, asthma, CKD-5 requiring dialysis, malnutrition, recurrent hospitalisation, and systemic and IP antibiotic use for recurrent peritonitis and invasive procedures, may have contributed to the patient’s fatality. Prolonged duration of PD (more than 36 months) may be associated with Aspergillus peritonitis.15 Although the patient received PD for only a few months, a delayed diagnosis and the persistence of catheter infection may have led to death in this case.
Several cases of FP caused by Aspergillus spp. have been reported worldwide; however, there is a paucity of data from Pakistan.5-8 Data from a study of 42 patients on acute and chronic PD showed 35 episodes of peritonitis (83.34%), with 11.00% diagnosed as FP and one of these FP cases due to Aspergillus spp.16 Due to limitations of diagnostic modalities, the authors’ diagnosis of CNS and FP could not be confirmed by molecular methods.
CONCLUSION
FP due to Aspergillus spp. is associated with high morbidity and mortality. A. terreus is an uncommon and deadly pathogen that does not respond to the common antifungals, including amphotericin B. Despite its difficult diagnosis and rare occurrence, an early diagnosis by culture using multiple samples of PD effluent, as was the case in the authors’ setup, may help in the decision to remove the PD catheter and to use newer drugs such as voriconazole or caspofungin. Early diagnosis and careful interpretation of culture results are, therefore, important for the treatment of these patients.