Webinar Summary
During a recent webinar, two leading experts discussed the ongoing impact of COVID-19, including transmission, hospitalizations, and risk to vulnerable populations in high-exposure settings, and highlighted the importance of post-exposure prophylaxis (PEP). They also examined the prevention gap from a primary care workflow perspective, the unmet needs that remain, and how optimal post-exposure care might be implemented.
Successful PEP, they argued, relies on prompt testing and timing. They also stressed the need for simplified prescribing, particularly in vulnerable and/or elderly populations who may already be taking multiple medications. They concluded that future PEP strategies must be simple to implement and prescribe, with clinically robust outcomes in older and/or vulnerable patients, and demonstrably effective at reducing transmission and severe COVID-19 outcomes in the context of real-world care.
Vulnerable People at Direct Risk of SARS-CoV-2 Transmission
In 2026, SARS-CoV-2 transmission and resulting hospitalizations remain a real-world challenge. This is particularly true in high-exposure settings such as households and long-term care facilities (LTCF), including nursing homes, assisted living, and group homes, said Stefan Gravenstein, Professor of Geriatric Medicine and Director of the Division of Geriatrics and Palliative Care, Brown University, Rhode Island, USA; and Lynn McNicoll, Professor of Medicine and Director of Education for the Division of Geriatrics, Brown University, Rhode Island, USA. They highlighted that vulnerable groups, such as older adults, immunocompromised individuals, postoperative patients, patients receiving chemotherapy, and those with underlying comorbidities, are often concentrated in congregate living environments,1 such as assisted living, group homes, and nursing homes. This can create repeated close-contact interactions, thereby multiplying the risk of transmission.
An estimated 75% of adults in the US are considered to be at increased risk of severe COVID-19, having at least one increased-risk condition.2 Those who are at risk and develop COVID-19 are more likely to experience serious outcomes, including hospitalization, ICU admission, intubation or mechanical ventilation, and death.3,4 In addition, SARS-CoV-2 infection in vulnerable patients increases hospitalizations, disrupts chronic care, and worsens overall health outcomes.4,5 Even mild COVID-19 may exacerbate pre-existing chronic conditions, contributing to increased morbidity and mortality.6-13 Key risk factors for severe COVID-19 include age, underlying medical conditions, vaccination status, and sociodemographic factors.2,3 Older age (≥65 years) is a major risk factor, although increased risk is also observed from 50 years onwards.2,3 Clinical risk conditions include cancer, chronic kidney disease, COPD, obesity, serious heart conditions, and Type 2 diabetes.4 Additional high-risk conditions include neurological, respiratory, and cardiovascular disorders, as well as acute kidney injury.6-13 Individuals who are unvaccinated or not up to date on vaccinations are also at increased risk of severe outcomes.3
Sociodemographic factors, including race, ethnicity, and socioeconomic status, further contribute to disparities in COVID-19 severity.3 Not only are care settings prone to higher risk of transmission, but vulnerable patients in these environments are often also at greater risk of severe outcomes after developing COVID-19.3
SARS-CoV-2 Transmission: Indirect Risk
McNicoll emphasized that the transmission risk of SARS-CoV-2 extends beyond vulnerable patients. Intimate, prolonged contact between patients/residents and caregivers, such as around-the-clock care, bathing, and feeding, increases the likelihood of caregivers acquiring and spreading SARS-CoV-2, including to other LTCF residents and their own families. Gravenstein added that in nursing homes, staff members providing care to multiple residents can further amplify the transmission risk. Moreover, up to four in 10 household contacts experience a SARS-CoV-2 infection, with household secondary attack rates at 43–47%.14,15 This in turn fuels transmission to the wider community, explained the speakers. Early intervention is, therefore, critical to protect high-risk populations as well as those around them. Recognizing both those at direct risk and those likely to transmit infection in the primary care setting is essential to identifying appropriate candidates for PEP.
Evolving Strains and Increased Transmissibility
Both experts noted that evolving variants of SARS-CoV-2, particularly Omicron, have higher transmissibility and reinfection rates.14,15 Omicron has a reported higher household secondary attack rate (42.7%), compared to the earlier alpha variant (36.4%).14,15 Omicron is also associated with 1.8 times higher hospitalization rates compared with the earlier delta strain, even while causing milder illness, due to the higher number of infections caused.16 The variable incubation period of Omicron, which is commonly 3–5 days depending on the strain, creates a narrow critical window for optimal intervention,17,18 Gravenstein emphasized. He explained that this makes prompt assessment and diagnosis critical for PEP to be effective, with optimal timing varying depending on the antiviral agent used. He highlighted the rapid evolution of SARS-CoV-2, particularly within the Omicron lineage, noting the emergence of the Omicron sublineage BA.3.2 in late 2024. By February 2026, BA.3.2 had been detected in wastewater samples from 25 US states and had been reported in at least 23 countries.19 Experts concluded that the overarching message should be to remain vigilant. Early identification and post-exposure intervention are crucial to prevent spread and reduce transmission risk and disease burden, they said.
The Importance of Early Intervention: Post-exposure Prophylaxis
Both experts emphasized the importance of early PEP intervention in those exposed to SARS-CoV-2, with the aim of protecting vulnerable populations and reducing transmission in high-risk settings.20-22 Utilization of oral antiviral PEP in the early stages of COVID-19 may reduce the viral load, duration of symptoms experienced, and risk of developing severe illness and long COVID, offering meaningful clinical benefit and reduced risk of transmission.23-34
Both experts agreed they had observed a shift in the attitudes of patients contracting SARS-CoV-2 from the start of the pandemic to today, including a reduced urgency around COVID-19 results and delays to testing and seeking treatment, with individuals not considering reaching out to their primary care doctors. As a result, people with suspected infection may delay testing or not test at all when symptoms develop. They noted that this prevention gap is a practical, real-world challenge, particularly important for those in high-contact settings and households.
After exposure to SARS-CoV-2, as with other respiratory diseases such as influenza, there is a critical period early in the viral replication cycle in which interventions such as antivirals are most effective and may reduce transmission (Figure 1).35-37 The experts noted that this is of particular importance to prevent outbreaks in high-exposure settings, which are associated with high-risk populations.38 Through early intervention in pre-symptomatic patients, primary care physicians may be able to protect vulnerable patients.
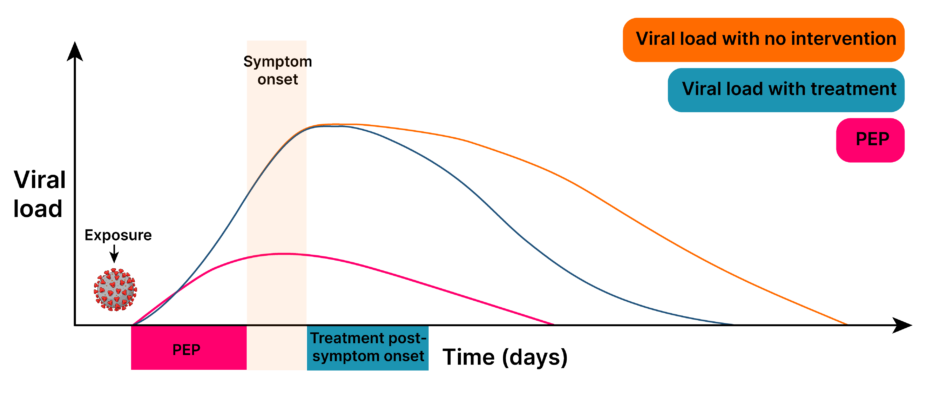
Figure 1: Antiviral intervention is most effective early in the viral replication cycle.35-37
PEP: post-exposure prophylaxis.
In a similar context of influenza, PEP administered immediately after exposure may suppress viral load,39 reduce secondary household transmission, and slow community spread.40,41
Overcoming Barriers to Prompt Treatment in Primary Care
While primary care practitioners may not necessarily come into contact with high-risk patients on a regular basis, they are more likely to encounter members of households or caregivers who are likely to be in contact with those individuals. Therefore, experts noted it is imperative that they focus on identifying candidates for PEP to help manage the risk of further exposure and transmission.
Both experts highlighted the need for rapid, on-site point-of-care testing in primary practice. Using multiplex PCR, which can distinguish COVID-19 from respiratory syncytial virus or flu, clinicians can act during the visit and administer PEP rather than waiting for delayed lab results from external providers, Gravenstein and McNicoll noted. They emphasized the importance of a prompt response, as delays can increase hospitalizations and mortality.34 Study results suggest that early antiviral treatment, as soon as possible after infection onset, is needed to mitigate transmission, preventing 50% more infections compared to late treatment (started 3–5 days after symptom onset).34
With optimal PEP delivery shown to occur immediately after exposure in influenza studies, McNicoll and Gravenstein emphasized the overarching importance of acting quickly in similar contexts, including for SARS-CoV-2.39-41 They also stressed that ensuring it is simple to prescribe would enable early intervention when it is most effective. Patients in LTCF settings and those interacting with vulnerable individuals should be prioritized, they added.
Real-World Implementation into Clinical Workflow
Rapidity of PEP delivery is reliant on confirmed exposure to SARS-CoV-2, for which the experts recommended a clinic-based nucleic acid amplification test or multiplex testing, with repeated testing if results are negative but suspicion remains. Both experts reiterated that rapid in-clinic testing is preferable to waiting for results from an external center.
Considering real-world use of PEP in geriatric patients, they noted the importance of oral antiviral medications that are suitable for use in frail, older individuals and with minimal drug–drug interactions. They also noted that clinical trials often assess efficacy in younger, healthy individuals, meaning it can be difficult to extrapolate to older patients presenting in clinical practice who may be on a range of drugs. This can sometimes result in prescriber reluctance.
In addition, both experts maintained that the usefulness of non-pharmacologic measures should not be underestimated. Masking, handwashing, general hygiene, and isolation, where feasible, should always be implemented if COVID-19 is suspected, even if tests are negative.
Clinical Profiles of Emerging Post-exposure Prophylaxis
Both experts recognized the promise of emerging oral antivirals to provide protection for at-risk groups, particularly in those where vaccine efficacy may be waning or limited.42 Clinical trials have shown potential, but certain agents are not yet approved in the US for PEP, they noted. McNicoll emphasized that, when evaluating clinical outcomes for COVID-19 PEP, clinicians are interested not only in whether a patient develops COVID-19, but also in disease severity, symptom duration, and the need for hospitalization.18,21,22
SCORPIO-PEP Study
Ensitrelvir is currently under investigation for the prevention of symptomatic SARS-CoV-2 infection in household contacts of an individual with symptomatic COVID-19.18,43 In the SCORPIO-PEP trial, when treatment was started within 72 hours of symptom onset in the index patient, ensitrelvir was effective in significantly protecting household contacts from COVID-19, including those at high risk. The investigational drug was well tolerated, with no new safety concerns, suggesting potential for other settings with high-risk populations.18,43
Oral Nirmatrelvir-Ritonavir PEP
Nirmatrelvir/ritonavir has been assessed for efficacy and safety as PEP in asymptomatic household contacts with COVID-19 when administered within 96 hours for 5 or 10 days. In a Phase II-III double-blind trial (n=2,736), PEP with nirmatrelvir/ritonavir for either 5 or 10 days did not significantly reduce confirmed symptomatic COVID-19 compared with placebo.18,21
Oral Molnupiravir (MOVe-AHEAD)
The Phase III household prevention trial with molnupiravir (MOVe-AHEAD) was a randomized, controlled, double-blind Phase III trial comparing molnupiravir with placebo (n=763). The primary efficacy endpoint was the incidence of COVID-19 through Day 14 in modified intent-to-treat participants without detectable COVID-19 at baseline. It was found that molnupiravir was well tolerated, but did not meet the prespecified superiority criterion as a PEP.44
Conclusion and Future Outlook
COVID-19 continues to pose a significant risk to vulnerable populations, particularly those in congregate living facilities and those with at-risk characteristics. Paired with evolving, more transmissible disease variants and a waning awareness within the general population, it is crucial for primary care physicians to remain vigilant in their practice: vaccination, rapid testing, and masking remain essential to ensure robust management and prevention of the spread of SARS-CoV-2. Recognizing both indirect and direct risk, particularly for those in high-contact care settings, is key to reducing transmission and protecting those most at risk from hospitalization or severe outcomes. Looking ahead, emerging PEP strategies may help address persistent prevention gaps in high-risk settings, subject to further evidence generation and regulatory review.







