BACKGROUND
Lack of affordable renal replacement therapy (RRT), especially in limited resource settings, puts patients with kidney failure at the risk of imminent death. Globally, between 2.2 and 7.1 million patients may have died prematurely in 2010 because they did not have access to RRT.1 The authors of the recently published Global Kidney Health Atlas projected that in 2030, 14.5 million people will have end stage kidney disease and need RRT, yet only 5.4 million will receive it because of economic, social, and political factors.2
METHOD
The authors propose allo-haemodialysis (alloHD) as a novel dialytic RRT modality. With alloHD, the patient’s blood flows in the dialyser counter-current to the blood of a healthy subject (‘buddy’). Fluid and toxins transferred from the patient to the buddy are excreted by the buddy’s healthy kidneys (Figure 1A).
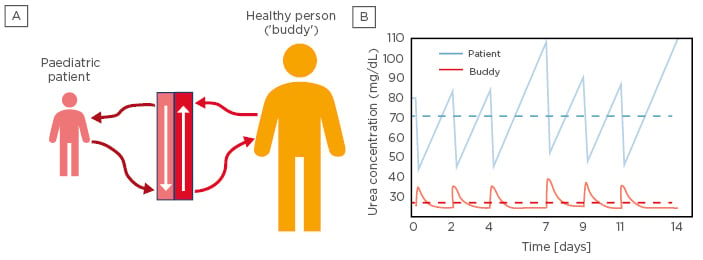
Figure 1: Allo-Haemodialysis in a paediatric patient and adult ‘buddy’.
A) Allo-haemodialysis setup in which a paediatric patient (left) is dialysed against a healthy subject (buddy; right). B) Urea kinetics in a paediatric patient (blue) and healthy adult buddy (red). Solid lines correspond to intra and inter-dialytic urea levels; the dashed lines denote time-averaged urea concentrations for both patient and buddy, respectively.
To quantify the alloHD efficacy, the authors used a two-compartmental model of urea for the patient and buddy. The buddy model also accounts for increased diuresis due to the transferred fluid and urea. The fluid and urea transfer across the hollow fibre is represented by a spatiotemporal model,3 in which the urea concentration in the patient’s blood decreases along the hollow fibre while it increases in buddy’s blood, which is flowing in the counter-current direction in the space that is the classical dialysate compartment.
In the analysis, a scenario in which a 20 kg paediatric patient is dialysed against a 70 kg buddy was simulated. The authors use the developed model to assess the long-term urea kinetics in both subjects using a paediatric dialyser (surface area 0.9 m2), blood flow rates of 150 mL/min, an ultrafiltration volume of 1 L/session, a dialysis time of 4 hours, and a thrice weekly alloHD schedule.
RESULTS
Urea kinetics for an initial urea concentration of 80 mg/dL (patient) and 25 mg/dL (buddy), respectively, were computed. The results suggest that three alloHD sessions per week are enough to keep the patient’s time-averaged urea concentration below the initial urea concentration and achieve a weekly standard Kt/V between 1.57 and 1.70 (Figure 1B).
CONCLUSION
While this analysis is limited to urea, other toxins will also be transferred from the patient to the buddy. Further ex vivo and animal studies are required to gain further insights into this novel RRT concept.







