INTRODUCTION
In the early 1990s, clinicians in Central America and Sri Lanka noticed an upsurge of chronic kidney disease (CKD) in agricultural communities, predominantly amongst sugarcane workers and paddy farmers. Clinically, these patients did not present traditional causes of CKD such as glomerulonephritis, polycystic kidney disease, and hypertension; the latter only as a late feature. Proteinuria was rare, but when present was of the tubular type. Ultrasound showed bilateral shrunken kidneys and renal biopsies revealed a chronic tubulointerstitial nephropathy. This condition is known under many names, including CKD of unknown aetiology/cause (CKDu) and Mesoamerican nephropathy (only in Central America); recently, chronic interstitial nephritis in agricultural communities (CINAC) has been suggested as an overarching name.1 Because of recent increasing awareness, multiple similar regional epidemics of CKDu have been suspected around the globe (i.e., the Uddanam region of Andhra Pradesh in India, Egypt, Tunisia, Senegal, and Peru, probably with more to follow).2 Up to now, there is no consensus on its aetiology. Heat stress or dehydration and toxic agrochemical exposure are the two major hypotheses. Also, there are no diagnostic criteria that can directly identify patients with CINAC.
METHODS
To address these issues, the authors analysed renal biopsies by light and electron microscopy in CINAC patients (Sri Lanka: 18; El Salvador: 10; India: 1; France: 3), comparing patients to those with normal kidney function at implantation and after 6 and 12 months of calcineurin inhibitor (CNI) therapy, transplant patients on CNI with indication biopsies (n=24), proteinuric nephropathies (n=15), light-chain disease (n=4), several cases on nephrotoxic drugs (lomustine, clomiphene, lithium, tenofovir, cisplatinum), and patients with reduced renal function of various causes (n=20).3 In addition, a rat study was conducted comparing the histopathology of heat stress or dehydration with cyclosporine nephrotoxicity.
RESULTS
In addition to previously described histopathological changes, there was a unique constellation of proximal tubular cell (PTC) lesions including cellular/tubular atrophy, cell fragment shedding, weak-to-nonproliferative capacity of the PTC, increased proliferation/hypertrophy of the distal tubules, and the presence of enlarged dysmorphic lysosomes with a light–medium electron-dense matrix containing dispersed dark electron-dense nonmembrane-bound round/irregular ‘aggregates’ (Figure 1).3 Identical renal lesions were observed in 55–80% of renal transplant protocol biopsies taken after 6 and 12 months of CNI therapy as well as in indication biopsies, whereas in implantation biopsies the prevalence of the lesion was 10-fold lower. Moreover, several cases on nephrotoxic drugs (lomustine, clomiphene, lithium) and a subset of patients with light-chain disease, all conditions that can either directly or indirectly be linked to CNI, presented the same lesion. Control biopsies (n=66) of normal kidney, toxic nephropathies (tenofovir, cisplatinum), and overt proteinuric patients of different aetiology to some extent could demonstrate the tubular cell changes observed by light microscopy, but not or very rarely those that were observed by electron microscopy. Rats treated with cyclosporine for 4 weeks developed similar PTC alterations, which were absent in a dehydration group.
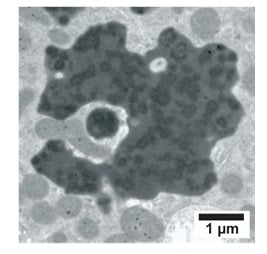
Figure 1: Enlarged dysmorphic lysosome containing dispersed electron-dense round to irregular aggregates as observed in a patient with chronic interstitial nephritis in an agricultural community.
Image reproduced from Vervaet et al.,3 published as an open-access article under the CC BY- NC-ND license (http://creativecommons.org/licenses/by-nc-nd/4.0/).
CONCLUSION
In conclusion, a sensitive constellation of renal PTC lesions was detected associated with CINAC and some toxin-induced nephropathies, amongst which was CNI nephrotoxicity. This indicates a toxin-induced aetiology for CINAC and suggests a pathomechanistic involvement of the calcineurin pathway, although involvement of other pathways cannot be excluded.