BACKGROUND AND AIMS
Indoxyl sulfate (IS) and p-Cresyl sulfate (PCS) are uraemic toxins originating from the breakdown of tryptophan and tyrosine/ phenylalanine by microbiota during the intestinal fermentation of proteins.1-3 These amino acids are respectively transformed into indole and p-Cresol and conjugated with sulfate in the liver to form IS and PCS.3 Ninety per cent of circulating IS and CS is bound to albumin, while only 10% of their serum concentration is free and can exert toxic activities.3-4 IS and PCS are progressively accumulated in patients with chronic kidney disease (CKD),5-7 and were shown to promote oxidative stress and inflammation leading to tissue injury as observed in in vitro experiments.8-10 The accumulation of IS and PCS was implicated in bone damage and cardiovascular disease in patients with CKD. Their cardiovascular involvement may be sustained by endothelial dysfunction, vascular calcification, and cardiac hypertrophy.5-7
In keeping with these findings, the authors studied the relationship between serum levels of free IS and PCS with divalent ion metabolism variables and cardiovascular morbidity (stroke, heart failure, angina pectoris, and myocardial infarction) in 139 patients receiving haemodialysis (age: 68±13 years; weight: 65±13 kg; dialysis vintage: 69±71 months).
MATERIALS AND METHODS
Patients were grouped according to tertiles of serum-free IS and PCS to analyse their association with variables of mineral metabolism. Patients in the highest tertile of serum-free IS showed lower dialysis vintage and serum 1,25 hydroxyvitamin D2 (1,25[OH]D2) than patients in the lowest tertile (7.6±4.4 versus 11.7±9.4 pg/mL; p=0.016). They also had lower serum parathyroid hormone (PTH) than patients in the lowest tertile (180±112 versus 260±240 pg/mL; p=0.017). After this baseline determination, PTH was measured every 6 months during a 2-year follow-up. The average of these PTH values was lower in patients in the highest tertile of serum-free IS than patients in the lowest tertile (178±106 versus 254±215 pg/mL; p=0.025; 206±136 pg/ mL in patients in the middle tertile). This finding was confirmed in patients not taking calcitriol during the follow-up (164±59 versus 333±383 pg/mL; p=0.05). No relationships were observed among serum-free PCS concentrations and calcium–phosphate metabolism variables.
RESULTS
During a 4-year follow-up, cardiovascular events occurred in 45 patients: 13 patients were in the lowest tertile of serum-free IS (28.9% of patients in this tertile), 16 were in the middle tertile (33.3%), and 16 were in the highest tertile (34.8%; p=0.8). Conversely, cardiovascular events were associated with tertiles of serum-free PCS as they occurred in 10 patients in the lowest tertile (20.8%), 13 in the middle (29.5%), and 22 in the highest tertile (48.8%; odds ratio: 3.3; 95% confidence interval: 1.3–8.3; p=0.01).
Cox regression analysis confirmed the increased cardiovascular morbidity in patients in the highest tertile of serum-free PCS (represented by the blue line in Figure 1) compared to those in the lowest and middle tertiles taken together (represented by the red line in Figure 1; odds ratio: 2.3; 95% confidence interval: 1.0–5.1; p=0.01). In the used model, body weight and history of cardiovascular events were also associated with cardiovascular events in the follow-up.
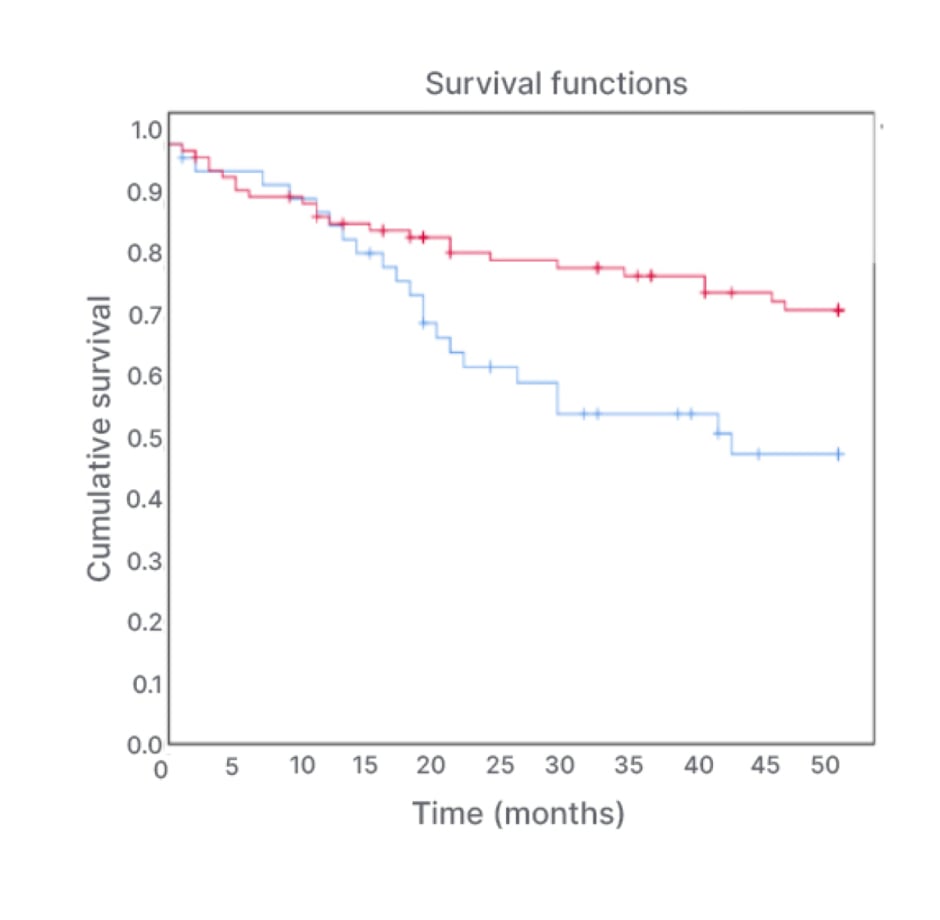
Figure 1: Cox regression analysis evaluating cardiovascular events during a 4-year follow-up in tertiles of serum concentrations of free p-Cresyl sulfate.
CONCLUSION
The authors’ findings confirm the association of serum-free PCS with cardiovascular risk in patients receiving haemodialysis, as previously observed in other studies. These findings also suggest that serum-free IS was negatively related with serum PTH and 1,25(OH)D2, suggesting that IS may have a toxic effect on parathyroid and renal cells, leading to the inhibition of PTH and 1,25(OH)D2 release, with potential implications in the metabolic bone disease developed by patients with CKD. ●







