Abstract
Dengue is a viral infection transmitted by an Aedes aegypti mosquito bite that poses a major threat to public health worldwide. While acute kidney injury secondary to dengue infection is a potentially lethal complication, it remains one of the least studied complications of dengue fever. The underlying mechanism of dengue-associated acute kidney injury is complex because it involves multiple pathways that could independently lead to its occurrence. Therefore, the cornerstone of dengue-associated acute kidney injury management should involve prompt recognition and identification of the at-risk population and administration of appropriate supportive treatment in a timely manner with the aim of preventing both renal and non-renal morbidity and mortality.
INTRODUCTION
Dengue virus infection (DVI) is a vector-borne febrile illness which has a high prevalence in most tropical countries worldwide. DVI can be classified into dengue fever (DF), dengue haemorrhagic fever (DHF), dengue shock syndrome (DSS), and expanded dengue syndrome (EDS). The incidence of DVI has markedly increased throughout recent decades with reported worldwide cases increasing from 2.2 million in 2010 to 3.5 million in 2015.1,2 DVI has the ability to cause a wide spectrum of clinical features from asymptomatic infection to severe systemic organ dysfunctions.3,4 Renal involvements related to DVI have a variety of presentations, including proteinuria, glomerulonephritis, and severe acute kidney injury (AKI).5 AKI is a significant, albeit poorly studied, complication of DVI.6 The incidence of AKI in DVI has shown great discrepancy, with previous studies reporting a range from 0.83–14.40% with a mortality rate of 11.30–60.00%.6-11
PATHOPHYSIOLOGY OF DENGUE-ASSOCIATED ACUTE KIDNEY INJURY
Several mechanisms have been postulated for the pathogenesis of dengue-associated AKI (DAKI), including direct action by the virus, haemodynamic instability, rhabdomyolysis, haemolysis, and acute glomerular injury.12 This is illustrated in Table 1.
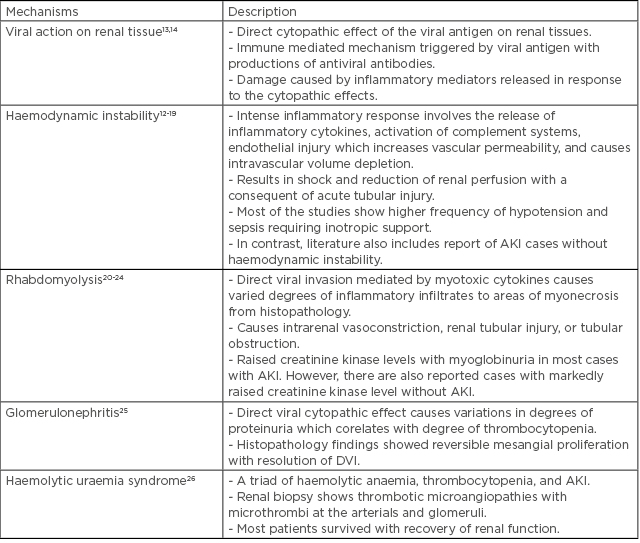
Table 1: Mechanisms of acute kidney injury in dengue fever.
AKI: acute kidney injury; DVI: dengue virus infection.
DIAGNOSIS OF DENGUE-ASSOCIATED ACUTE KIDNEY INJURY
The authors found that most data currently available are derived from heterogenous case series and retrospective case studies. To further complicate the matter, EDS is a new clinical entity added to World Health Organization (WHO) guidelines to highlight the wide spectrum of atypical manifestations of dengue infection affecting various organ systems in DVI.27
The literature demonstrates a significant increase in DAKI, which can lead to a higher mortality rate and prolonged hospital stay.
Based on the Kidney Disease: Improving Global Outcomes (KDIGO) guidelines, AKI can be defined by either an increase in serum creatinine (SCr) by >0.3 mg/dL, an increase in SCr by >1.5-times baseline, or a reduction in urine volume <0.5 mL/kg/hour for 6 hours. It can be further subdivided into Acute Kidney Injury Network (AKIN) stages based on change in SCr level. The Acute Dialysis Quality Initiative’s (ADQI)28 RIFLE criteria are also used in the classification of AKI. The acronym RIFLE defines three grades of increasing severity of acute renal failure (risk, injury, and failure) and two outcome variables (loss and end stage kidney disease).
The AKIN and RIFLE classifications were used to define AKI or DAKI in most documented case studies and retrospective case series, as was the conventional AKI criteria (SCr >2 mg/dL). A systematic review conducted by Mallhi et al.27 found that the AKIN criteria was more sensitive as it picked up a higher incidence of DAKI (3.3–13.3%). Mallhi et al.27 compared five studies which used the conventional definition (SCr >2 mg/dL) and AKIN criteria. They found that when using AKIN criteria, the incidence of AKI was higher. This can be explained because AKIN criteria classify AKI as a 1.5-times increase in SCr from baseline within 7 days (compared to conventional SCr >2 mg/dL), or as increased SCr >26.2 µMol/L from baseline within 48 hours.
In contrast, Kuo et al.11 and Basu et al.9 reported a very high incidence of DAKI when using RIFLE criteria (27.1% and 35.7%, respectively). The high incidence reported by Basu et al.9 was because of the low number of dengue patients (28 patients) and the fact that the RIFLE criteria was only validated in half of the studied participants.
Most authors did not outline the reason why a specific classification was chosen. Nevertheless, many agreed that there is an urgent need to develop a consensus on the definition of AKI. This would help to demonstrate the true incidence and burden of disease, and it would also enable a comparison of studies in the field. Table 2 illustrates different studies that used AKIN, RIFLE, or conventional AKI criteria.
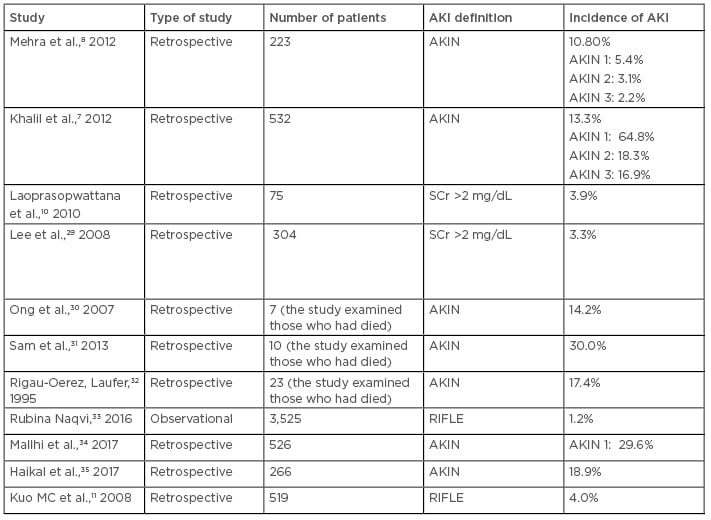
Table 2: Incidence of dengue-associated acute kidney injury in studies using various acute kidney injury classifications.
AKI: acute kidney injury; AKIN: Acute Kidney Injury Network; DHF: dengue haemorrhagic fever; DVI: dengue virus infection; RIFLE: risk, injury, failure, loss, and end-stage kidney disease.
CLINICAL FEATURES OF DENGUE-ASSOCIATED ACUTE KIDNEY INJURY
A retrospective chart review by Naqvi et al.33 demonstrated that the presence of fever, jaundice, oligo-anuria, and vomiting are strongly associated with development of DAKI. Based on a review of the literature, the authors did not find any validated specific constellation of symptoms at presentation of illness that may render a higher probability of developing AKI during the course of DVI.
However, in 2017, Mallhi et al.34 proposed a predictive model consisting of different variables (including sex, type of DF, and transaminitis) which cause AKI in dengue patients based on a large retrospective cohort subjects of 667 dengue cases. By using this predictive model, the authors found excellent accuracy in predicting the possible development of AKI in dengue patients.34
MANAGEMENT OF DENGUE-ASSOCIATED ACUTE KIDNEY INJURY
General Management
Over the decades, there has been difficulty in understanding the exact mechanism of DAKI because of the complexity of its pathogenesis. Having a clearly defined approach to managing DAKI is important to prevent further complications that can cause an increase in morbidity and mortality rates. The main principle of management for DAKI is to restore the haemodynamic status with either crystalloid or colloids. Early detection of DAKI and prompt management of any subsequent complications is needed to reduce the chance of mortality as well as to prevent the development of chronic kidney disease (CKD) after recovery from the acute event. Clinical assessment of patients’ haemodynamic and fluid status plays a major role in adequate volume replacement.36
Nevertheless, there is limited literature on the use of haemodialysis as a supportive treatment in DAKI while awaiting disease and renal recovery. There are currently no guidelines in terms of when to initiate renal support, the optimal dialysis dose, or the preferred modality of renal replacement therapy in treating AKI during DVI.37 However, this should not become a barrier to initiating haemodialysis in the event of AKI, and most nephrologists or physicians will initiate dialysis based on conventional indications. One observational study investigated the use of haemodialysis as a supportive treatment in DAKI in Pakistan and studied 43 patients with DAKI who were administered fluids and haemodialysis.33 A total of 37 patients had a complete recovery, and 6 died during the acute phase of illness. Of the 37 patients, 31 had received haemodialysis.33
Management According to Pathogenesis
Beside general management, DAKI patients should be managed as per the underlying AKI causes. Table 3 summarises the literature reported for the proposed mechanism of DAKI, management, and outcome of patients with DAKI.
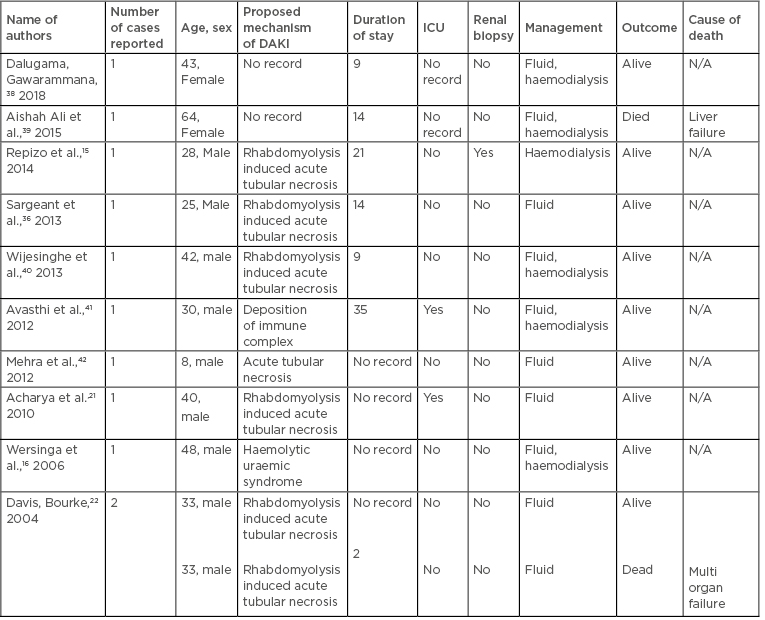
Table 3: Mechanisms of acute kidney injury in dengue fever.
DAKI: dengue associated acute kidney injury; ICU: intensive care unit; N/A: not applicable.
Management of the Complications
Besides management of DAKI, there is a need to identify and treat the potential complications which almost invariably increase the rate of mortality or morbidity in patients. Complications that are associated with DVI are rhabdomyolysis, glomerulonephritis, hypotension, CKD progression, and prolonged hospital stay.
Rhabdomyolysis is a complication that is rarely taken seriously in DVI. It can increase the risk of AKI, which will eventually increase the patient’s risk of mortality and morbidity. Patients with rhabdomyolysis usually present with dark-coloured urine or a non-specific type of generalised myalgia with correspondingly raised SCr kinase. Adequate fluid replacement and urine alkalinisation with sodium bicarbonate has been shown to improve the condition of rhabdomyolysis and improve the outcome of AKI.43 Monitoring changes in creatinine kinase is necessary to guide management of the treatment and assess response. There are three case reports, from 1996, 2007, and 2015, that showed good renal outcome for patients who had rhabdomyolysis with DAKI and had responded well to fluid replacement.20,23,24
Apart from acute tubular necrosis, the renal cortex, which is rich in glomeruli, can also be affected in DAKI. Patients with glomeruli involvement usually present with nephritic syndromes, which are proteinuria, haematuria, oedema, or hypertension. Immune complex deposition on the glomerulus has been proposed as one of the mechanisms of glomerular injury in DAKI.13 There were a few case reports regarding renal biopsy proven glomerulonephritis as complications of DVI. Upadyaha et al.17 reported a 15-year-old male with DAKI in whom renal biopsy showed IgA deposition. The patient was managed with fluid replacement and haemodialysis. There was evidence of resolution of the IgA deposition 6 weeks later, which was confirmed with repeated renal biopsy. Lizarraga et al.25 also reported a 66-year-old woman who was admitted for DAKI and had IgG deposition along the glomerular capillary walls with antiglomerular basement membrane disease and myeloperoxidase positivity. The role of immunosuppressants and corticosteroids have been reported in 2012 and 2013, wherein renal biopsies of a 32-year-old female and 22-year-old female with DAKI triggered the presentation of lupus membranoproliferative glomerulonephritis.44,45 Generally, glomerulonephritis triggered by dengue virus has a good outcome even though some cases require immunosuppressants. Hypotension or haemodynamic instability in which patients can enter a state of shock can be seen in DVI. During this period, there will be a release of inflammatory cytokines, activation of the complement system, endothelial injury, and plasma leakage which eventually results in loss of intravascular fluid. Haemodynamic instability and a state of shock can cause renal hypoperfusion which leads to AKI.18
One important, well-recognised complication of DAKI is the progression to CKD. A gradual decrease in glomerular filtration rate (GFR), as well as increased proteinuria, is the hallmark of the disease. Patients with AKI who required haemodialysis have a poorer outcome, including progression to end-stage renal disease.47 To date, there is only one publication that studied the short-term renal outcome following AKI among dengue patients. In this study, 72 out of 526 patients diagnosed with dengue who developed AKI were recruited using AKIN criteria. Dengue patients who developed AKI were followed up for a post-discharge period of 3 months and renal recovery was assessed. A substantial number of patients at the end of the study had estimated GFR (eGFR) values that corresponded to the KDIGO classification of CKD. Of these, 50.7% (n=36/71) of patients had residual renal impairment compatible to CKD Stage 2, while 22.5% (n=16/71) of patients had eGFR corresponding to the advanced CKD stages, such as Stage 3 and Stage 4. Extended longitudinal follow-up of these patients may result in further improvement in eGFR.48 This study highlighted the need to monitor the long-term renal function of patients who develop DAKI. Moreover, DAKI can cause prolonged hospital stays which can increase the rate of mortality and morbidity, as well as healthcare costs.
PREDICTORS OF DENGUE-ASSOCIATED ACUTE KIDNEY INJURY
Demographic
Male sex was found to be an independent predictor for developing DAKI,7,27 with an odds ratio of 2:1.34 In another retrospective study of 304 patients, DHF and being over the age of 30 were identified as risk factors for DAKI.6 The incidence of AKI was higher in those above the age of 65 years.29 An observational study that involved 43 patients with DAKI showed similar findings.33 These findings were consistent in another cross-sectional study that recruited 217 DAKI patients.50 The authors’ review of the literature has found no specific racial predilection for DAKI. Similarly, socioeconomic status is not considered a risk factor for DAKI.
Severe Dengue with Hypotension
Severity of dengue infection, specifically DHF and DSS, was a significant predictor of development of DAKI and was associated with a higher mortality rate.51 Similar findings were noted in a study involving paediatric population from Thailand.10 Another study found that those with severe DF with hypotension shock requiring inotropic support had higher incidence of DAKI.8,33 In addition, those requiring ventilatory support were at increased risk of developing DAKI.49 Two recent studies have reported third-spacing and haemoconcentration as significant risk factors for DAKI. The incidence of DAKI risk was increased by as much as 4.7-fold in the presence of haemoconcentration or when the haematocrit level exceeded 46.5, indicating severe bleeding or third-spacing.35,46 Notably, severe dengue with concurrent DAKI resulted in the worst outcome with the reported mortality rate as high as 64%.6-8
Pre-Existing Renal Disease and Autoimmune Disorder
Acute dengue infection with underlying renal parenchymal diseases predisposed the patient to develop DAKI. The associated renal diseases were glomerulonephritis, nephrotic range proteinuria, lupus nephritis, IgA nephropathy, mesangioproliferative glomerulonephritis, and systemic lupus erythematosus.6,27
Comorbidities and Organ Involvement
A retrospective cohort study reported that co-existing viral hepatitis put DVI patients at risk of kidney failure. The study also emphasised the elevation of serum level of alanine aminotransferase (ALT) and alkaline phosphatase (ALP) as additional risk factors for development of AKI.8 Several other studies also reported that transaminitis was positively associated with the development of DAKI. The presence of multiple organ failure was also reported in the literature as a common predictor of DAKI where capillary leakage was a common feature.8,27,34,51 Rhabdomyolysis with raised creatinine kinase levels could also lead to renal damage by infrarenal vasoconstriction, direct tubular injury, and tubular obstruction.17,37 Other precipitating factors included concurrent sepsis or bacteraemia, low levels of albumin and bicarbonate, metabolic acidosis, prolonged activated partial thromboplastin time, thrombocytopenia, jaundice, gastrointestinal bleeding, elevated blood urea nitrogen, hyperglycaemia, and neurological involvement.6-8,27,33,49 A higher baseline SCr in dengue patients was associated with higher risk of DAKI.49 A small study that involved 75 patients showed that obesity with a weight standard deviation score of more than two was associated with an increased risk of DAKI.10 Despite hyperglycaemia being a risk factor, diabetes was not associated with DAKI.34,46 Length of hospital stay and intensive care unit admission were identified as independent risk factors for DAKI.49
Nephrotoxic drugs
The nephrotoxic drugs used in DVI were predominantly antibiotics, especially the potent antibiotics with systemic effect, such as vancomycin and carbapenems. Other drugs included amphotericin B and various antiviral medications. Additionally, a study in elderly dengue patients with comorbidities and multiple organ dysfunction found that the use of nephrotoxic drugs in dengue patients during hospitalisation had poorer renal outcomes.34
BIOMARKERS
The quest for AKI biomarkers is a field of intense contemporary research. Conventional biomarkers, such as urinary casts and fractional excretion of sodium, were non-specific in diagnosing the early stages of AKI. With the advent of functional genomics and proteomics, several studies have attempted to utilise molecular biomarkers in an attempt for early detection, risk stratification, and prognostication of DAKI. Several studies predicted an early phase of AKI based on differential expression of plasma and urine neutrophil gelatinase-associated lipocalin and cystatin C. This powerful biomarker acted as emerging independent predictors of AKI with an outstanding area under curve of 0.998 and 0.910 for 2-hour urine and plasma respectively, hence making it the most sensitive biomarker for AKI.52 Other promising sequential biomarkers were IL-18, kidney injury molecule, liver-type fatty acid binding protein, and NF-κB. To date, none of these biomarkers have been tested in DAKI.52
DISCUSSION
The authors’ review of the literature revealed wide variation of DAKI incidence, which was due to the difference in criteria selected to define AKI, namely the AKIN, RIFLES, and KDIGO classifications, and the heterogeneity of the studied population. There is no international consensus to date. Potential biomarkers like neutrophil gelatinase-associated lipocalin were being studied to diagnose AKI. There were no specific or standardised recommendations for medical treatment of patients with DAKI. Renal replacement therapy was still used conventionally as there were no updates on the techniques, timing to initiate dialysis, or dosing and modality in dengue patients. Fluid balance and replacement remained the main methods of treatment in addition to targeted therapy tailored specifically to the vast spectrum of dengue complications.
CONCLUSION
In summary, multiple retrospective studies and case series have demonstrated that DAKI is a frequent and highly significant complication of EDS. The morbidity and mortality rates are greatly increased when there is a delay in recognition and consequent lack of prompt intervention. Hence, there should be an increased awareness among clinicians involved in the management of DVI. In addition, DAKI is associated with prolonged hospital stay and possible poorer renal outcome, thus indirectly adding to the healthcare burden. Presence of male sex, DHF, multiple organ dysfunction, rhabdomyolysis, diabetes, nephrotoxic drug use, and late hospitalisation may increase the incidence of DAKI. These findings clearly demonstrate the need for a high index of suspicion and vigilant monitoring in these groups of patients from the onset. The authors strongly believe a future prospective, multicentre study will likely aid clinicians regarding early predictors and clinicopathogenesis of DAKI, which will provide a better opportunity for timely intervention and a more positive outcome of the disease.







