Meeting Summary
The symposium reviewed the challenges in providing dialysis to patients with kidney disease and outlined data supporting the effectiveness of home haemodialysis for improving clinical outcomes and patient quality of life. Prof Maria Auxiliadora Bajo opened the symposium by introducing the NxStage® System One™ for home-based dialysis. Dr Natalie Borman discussed the growing global burden of dialysis treatments and the ability of new technologies, such as the NxStage System One, to meet this need. Dr Sunita Nair then reviewed outcomes for patients using home-based dialysis in the Knowledge to Improve Home Dialysis Network in Europe (KIHDNEy) cohort. Prof Eric Goffin outlined the need to manage phosphorus levels in patients undergoing dialysis and compared phosphorus measures in patients undergoing different dialysis modalities. Matthew Herbert and his wife Kay then described their personal experience of using home-based dialysis and the impact this has had on their quality of life.
Introduction
Doctor Natalie Borman
Chronic kidney disease (CKD) is estimated to affect 13.4% of the global population and is characterised by a progressive loss of kidney function.1 CKD progresses with age and is often associated with cardiovascular (CV) comorbidities, premature mortality, and decreased quality of life.1
For patients with end-stage renal disease or acutely disturbed kidney function, the primary treatment option is augmenting impaired kidney function using haemodialysis (HD), i.e. removing waste and excess fluids by filtering a patient’s blood through semi-permeable membranes integrated in an extra-corporeal circulation. However, as the incidence of CKD continues to rise worldwide, more effective and accessible dialysis treatments are required.2 Indeed, the number of patients undergoing renal replacement therapies (RRT) is predicted to more than double by 2030.3
RRT becomes a central component of the lives of patients requiring dialysis, so patient preferences significantly influence treatment choices. Self-care or home-based dialysis is the preferred treatment for 56% of renal professionals and 56% of patients and includes options such as peritoneal dialysis (PD), home HD, and HD in self-care units.4,5 Patients with impaired kidney function have more control over the timing and efficiency of their treatment and are able to clear greater volumes of water per week with home-based dialysis. More frequent HD allows patients to more closely mimic the fluid and toxin removal performed by healthy kidneys and allows them to avoid having to attend regular appointments at a dialysis unit. Home HD has offered a safe and reliable treatment option for approximately 50 years, with levels of patient survival surpassing those seen for hospital or satellite units.6
Despite the success of self-care, it is estimated that only between 2% and 4% of patients undergoing regular dialysis in the UK are engaging in home HD therapy,7,8 which is well below the National Institute for Health and Care Excellence (NICE) HD/PD target of 30%. This trend is reflected internationally, with many countries failing to grow their home HD programmes and instead experiencing flat, or even decreasing, levels of use.9
Specialist renal healthcare professionals report that the poor implementation of more frequent home HD is reflective of the barriers to implementation that exist, including installation costs, complexity, and concern regarding cannulation and risk of infection. However, considering the established benefits associated with this therapy, emerging technologies must be embraced to break through these obstacles.
The NxStage System One is a simple and portable home dialysis option, designed to fit easily into a patient’s daily life, which utilises a drop-in cartridge-based system that contains a high-flux membrane to effectively remove waste products and permit more frequent, and even nocturnal, HD treatments at home. Compared with in-centre treatments three- times per week, more frequent HD with NxStage System One reduces the risk of mortality,10-13 improves blood pressure,14-16 and decreases cardiac stress,14,17-20 while improving recovery time21,22 and the overall quality of life reported by patients.23-30 In countries that previously experienced stagnating levels of home HD modality, implementation of NxStage System One has significantly increased the penetration of home HD treatment (Figure 1).31,32 However, strict adherence to training and system guidelines is necessary to fully realise the benefits of more frequent home-based HD using NxStage System One.
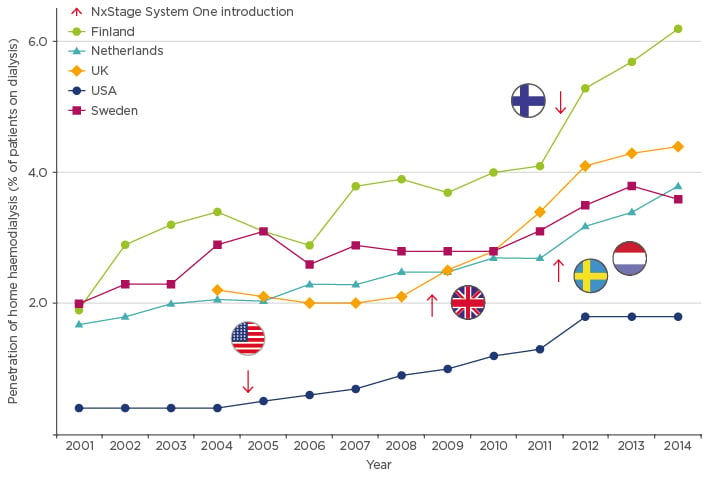
Figure 1: Penetration of home haemodialysis in countries where NxStage System One has been introduced.
This symposium discussed data supporting the use of the NxStage System One from the KIHDNEy cohort and how home HD can improve outcomes and quality of life for patients requiring dialysis.
New European Evidence with Home Haemodialysis Patients: 12-Month Follow-Up in KIHDNEy Cohort
Doctor Sunita Nair
The KIHDNEy cohort is a collaborative effort undertaken by nine home HD programmes that enrolled 182 patients using the NxStage System One to dialyse at home across five western European countries (Belgium, France, Italy, Spain, and the UK).31 Patients participating in the study had a mean age and BMI of 49.5 years (range: 15–84 years) and 26.1 (range: 13.3–50.8), respectively. Furthermore, approximately two-thirds of patients had at least one comorbid condition and the sample population had a mean comorbidity score (Charlson score) of 3.9.31
Prior to receiving home HD training, patients spent an average of 3 years on dialysis; 61% of patients were on conventional HD, 9% were on PD, 7% were kidney transplant recipients, and 17% were incident dialysis patients. The vascular access patterns of the patients showed that 74% used a fistula to dialyse, 24% used a catheter, and only 2% used a graft. Of the patients using a fistula, 76% used the button-hole technique to cannulate and 24% used the rope ladder technique.31
Compared with the previous Frequent Hemodialysis Network (FHN) study, patients in the KIHDNEy cohort took fewer sessions (18.9 versus 27.7) to complete training before being fully established to dialyse at home (Figure 2).31,33 Furthermore, patients who initiated the programme being able to cannulate independently required approximately three fewer training sessions.31
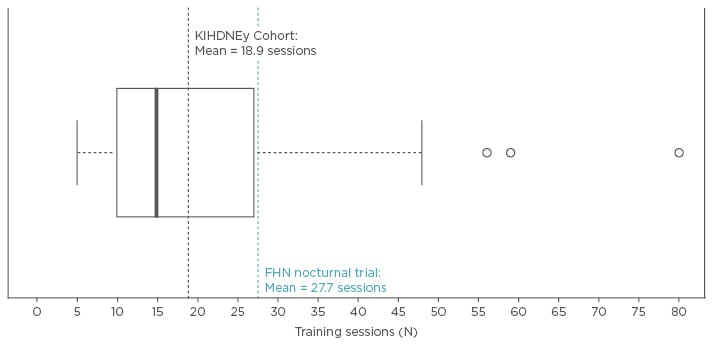
Figure 2: Short training times.
Number of training sessions before transition to home without supervision.
The majority of patients dialysed across 5–6 sessions per week with each session lasting 2–3.4 hours.31 On average, patients dialysed for 14.6 hours per week,31 which was marginally higher than the Dialysis Outcomes and Practice Patterns Study (DOPPS); however, 16% of the patients in KIHDNEy dialysed for less than the conventional 12 hours per week.34 Of the 30 patients who dialysed for <12 hours per week in this cohort, 28 had residual renal function (RRF), of whom 20 had a urine output of ≥1 L/day.31 Four patients had an output of 500–900 mL/day and 4 patients had anuria.31 Of the 4 patients with anuria, 3 weighed 40–56 kg and had a Kt/V of 2–2.6, while 1 patient weighed 67 kg and had a Kt/V of 1.2.31
The majority of patients used 20–30 L of dialysate per session or 100–175 L per week.31 When comparing prescription patterns by patient BMI, it was found that increasing BMI positively correlated with mean sessions per week, time per session, and volume of dialysate used per session, providing evidence that a personalised prescription allowing tailoring of dialysis intensity was achieved with the NxStage System One.31
Patients had a mean ultrafiltration rate (UFR) of 6.8 mL/h/kg, with 84% of patients having a UFR of <10 mL/h/kg, indicating that patients were achieving a less intensive UFR per session with the higher frequency of sessions enabled by home dialysis.31 Patients had a mean Kt/V of 2.61, which remained stable throughout the follow-up period.31 The proportion of patients requiring two or more antihypertensive agents to maintain appropriate blood pressure significantly decreased from baseline.31 Bicarbonate levels rapidly and significantly improved during the first 6 months and then stabilised, whereas potassium levels were stable throughout the 12 months of follow-up.31 Haemoglobin also remained stable and adequate at 11.4 g/dL and did not require any incremental dose in erythropoiesis-stimulating agent (ESA).31 However, there was a significant reduction in the use of anticoagulants.31
Of the 98 patients who provided data for RRF, 54 patients had RRF. Of these, 18% were rendered anuric at 12 months, compared with 50–67% of patients reported to be anuric in other studies of patients undergoing continuous ambulatory PD, automated PD, or intensive HD.31,35,36 Finally, the cumulative proportion of patients who remained on home-based HD after 3 years was 40%, while 19% of patients were stopping the modality, 31% had received a kidney transplant, and the mortality rate was 10%.31
In summary, the KIHDNEy cohort demonstrated the effectiveness of the NxStage System One in delivering dialysis to a wide spectrum of patients, including patients with comorbid conditions. The NxStage System One provided patients with individualised prescriptions, which resulted in less intensive UFRs and reduced use of antihypertensive agents. Patients maintained stable biochemistry and haematinics, with no change in ESA dosing and a reduced use of anticoagulants. RRF was preserved in 80% of patients and overall excellent clinical outcomes, including a high rate of continuation on home-based HD, were achieved. NxStage System One was able to offer the right treatment, to the right patients at the right place, with good clinical efficacy.
Mineral and Bone Disorders and Serum Phosphorus: We Can Free Diet Without Increasing Pill Burden
Professor Eric Goffin
Phosphorus homeostasis is important in CKD, and mineral and bone disease (MBD) remains an ongoing issue in dialysis populations. Patients undergoing HD typically fail to achieve adequate phosphorus control,37 and evidence suggests that increased phosphorus concentrations are toxic for endothelial cells, inducing arterial stiffness and vascular calcification. These changes are significant risk factors for CV mortality in patients undergoing HD.37,38
Dietary and pharmacological interventions can help lower phosphorus intake and serum phosphorus concentrations.37 Dietary counselling helps educate patients about phosphorus sources in food, such as food additives, and about how to reduce the phosphorus content when preparing meals. Supplemental phosphorus binders can be used to remove the excess of dietary phosphorus, but this therapy has limitations, including gastrointestinal side effects and a high pill burden, which can lead to poor adherence.39 In addition, phosphorus-binding medication is associated with significant economic costs.40,41
Typically, conventional HD has limited effectiveness in eliminating phosphorus, despite its low molecular weight, with a rebound in serum phosphorus levels generally following conventional three-times per week in-centre dialysis sessions.42 DOPPS found serum phosphorus levels in a population undergoing HD remained relatively unchanged over a 5-year period (2010–2015), and >36% of patients undergoing HD had serum phosphorus levels persistently above the target range.43
More frequent HD sessions, or longer sessions, offer improved phosphorus control. Furthermore, nocturnal HD improves the efficiency of phosphorus removal, reduces the phosphorus binder pill burden and is systematically better than conventional HD for CKD and MBD parameters.44-47 The FHN Daily Trial demonstrated better control of phosphorus with intensive therapy compared with conventional therapy, with a 0.46 mg/dL decrease (95% confidence interval [CI]: 0.13–0.78 mg/dL) in serum phosphorus levels in the intensive group (1.5–2.75 hours of HD, six-times per week) compared with the conventional group (2.5–4.0 hours of HD, three-times per week).45 In addition, patients treated with intensive daily dialysis experienced a 1.35 g/day reduction (95% CI: 0.20–2.50 g/day) in equivalent phosphorus binder dose at 12 months.45
The FHN Nocturnal Trial found that nocturnal HD sessions of 6–8 hours, six-times per week, were associated with a 1.24 mg/dL decrease (95% CI: 0.68–1.79 mg/dL) in serum phosphorus levels compared with the conventional HD group. Also, 73% of these patients did not require phosphorus binding medication at Month 12 compared with 8% of patients assigned to three sessions per week (p<0.001).45
Finally, preliminary results from the KIHDNEy cohort indicated that patients treated frequently with the NxStage One System maintain stable levels of phosphorus within the first 6 months.48 Further examination of the KIHDNEy cohort data revealed a trend towards decreased MBD parameters in patients undergoing more frequent home-based HD, but changes were not statistically significant.31 Even patients with high baseline phosphorus levels achieved good control of MBD parameters and a decreased phosphorus binder pill burden at 12 months, provided they were exposed to a longer duration of dialysis (≥15 hours).31 Furthermore, there was significant improvement in the proportion of patients achieving target phosphorus ranges across groups with or without RRF.31 There were also improvements in nutritional markers, including progressive gains in post-dialysis body weight and serum albumin concentrations, indicating that patients were eating more protein (Figure 3).31
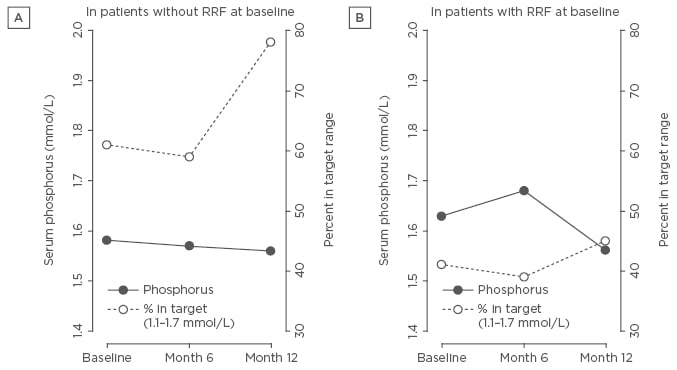
Figure 3: Serum phosphorus concentration and percentage of patients within target range at baseline, after 6 months and after 12 months on home haemodialysis.
A) Patients transitioning to System One without RRF at baseline; B) Patients transitioning to System One with RRF at baseline.
RRF: residual renal function.
My Dialysis Journey
Matthew and Kay Herbert
Coming to terms with a lifelong dependency on dialysis treatment can be an enormously daunting prospect. After being diagnosed with focal segmental glomerulosclerosis at 15 years of age, Matthew had to endure several in-centre dialysis treatments per week, which took a significant toll on his life and wellbeing. Here, his journey towards a life with greater control over dialysis treatment is described.
Initially, Matthew had to undergo two in-centre dialysis sessions every week. Initially, each session lasted 5 hours, before he progressed to three 4-hour sessions. After treatment, he often felt weak and fatigued, which made it difficult to complete daily tasks. Despite these obstacles, he continued persevering, successfully completing college, becoming a qualified chef, and meeting his wife in the process.
Over the following 10 years, Matthew had to co-ordinate life around work and family commitments and around appointments for in-centre dialysis treatment. A typical day would involve waking up at 05:30, travelling to the clinic for a 4-hour dialysis session, then heading straight to work for his morning shift at a restaurant. Matthew would then travel home to rest over his lunch break and return for the evening service. As it is not unusual for chefs to work 60–65 hour weeks, he was left exhausted.
Three years ago, at the suggestion of Dr Borman, Matthew began nocturnal home HD using NxStage System One. As he was already experienced with self-cannulation, training was straightforward and completed in ~5 days. Almost immediately, he felt a sense of rejuvenation, gaining back the energy that he felt was wasted during in-centre visits.
NxStage System One has allowed Matthew to more effectively balance his work schedule, while offering time to pursue activities that he was previously restricted from participating in, such as running, cycling, international travel, and even the occasional skydive for charity. After the birth of their son Oliver, he was able to lend a hand with night-time feedings, an activity that would have been exceedingly difficult alongside his in-centre dialysis schedule.
For Matthew, the transition from in-centre to home-based dialysis has changed his life, and he would recommend that any healthcare professional treating patients undergoing HD seriously consider this treatment option, as it has helped him a great deal both mentally and physically.
Summary
RRT is placing an increasing burden on healthcare systems around the world. Data from the KIHDNEy cohort indicate that home HD, when performed more frequently with the NxStage System One, is an effective RRT option, potentially offering improved clinical outcomes compared with other RRT.11,14 Furthermore, home HD can help improve quality of life for patients with renal disease.







